University of Babylon /College Of Engineering Electrochemical Engineering Dept.
advertisement
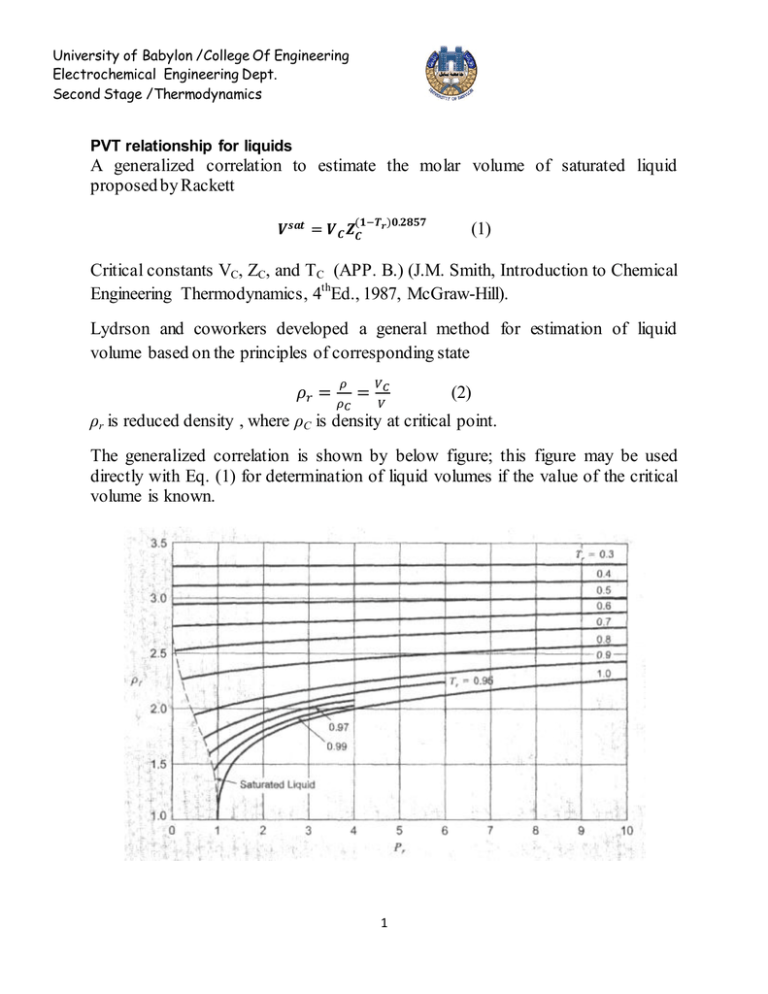
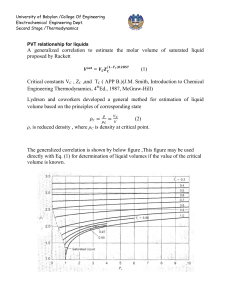
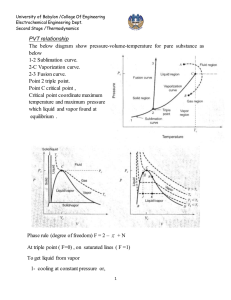
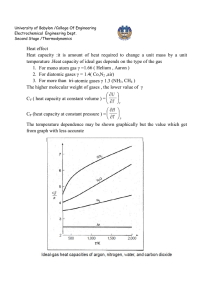
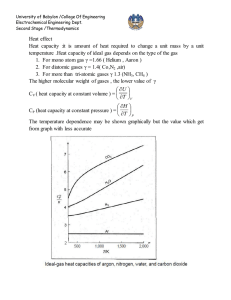
University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics PVT relationship for liquids A generalized correlation to estimate the molar volume of saturated liquid proposed by Rackett (1) Critical constants VC, ZC, and TC (APP. B.) (J.M. Smith, Introduction to Chemical Engineering Thermodynamics, 4thEd., 1987, McGraw-Hill). Lydrson and coworkers developed a general method for estimation of liquid volume based on the principles of corresponding state (2) ρr is reduced density , where ρC is density at critical point. The generalized correlation is shown by below figure; this figure may be used directly with Eq. (1) for determination of liquid volumes if the value of the critical volume is known. 1 University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics A better procedure is to make use of a single known liquid volume (state 1) by the identity, V2 V1 r1 r 2 V2 = required volume V1 = known volume r1,r2 = reduced densities read from figure Example: a) Estimate the density of saturated liquid ammonia at 310K. b) Estimate the density of liquid ammonia at 310 K and 100bar. 310 Tr 0.7643 405.6 VC 72.5 and Z C 0.242 ( from App.B)we get V Sat VC Z C(1Tr ) errer % 0.2857 (72.5)(0.242) ( 0.2357) 0.2857 28.35 cm 3 mol 1 theoratica l 29.14 28.35 2.71% exp erimental 29.14 b) The reduced conditions 100 Tr 0.7643 , and Pr 0.887 112.8 From figure we have ρr = 2.38 V VC r 72.5 30.5 cm 3 mol 1 2.38 30.5 28.6 6.64% 28.6 For saturated liquid at Tr = 0.764, we find from figure that ρr = 2.34. Substituation of known value into below equation errer % V2 V1 r1 2.34 3 1 (29.14) 28.65 cm mol r 2 2.38 2