There is a theory which states that if ever for... Universe is for and why it is here it will...
advertisement

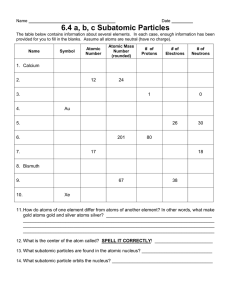
There is a theory which states that if ever for any reason anyone discovers what exactly the Universe is for and why it is here it will instantly disappear and be replaced by something even more bizarre and inexplicable. There is another that states that this has already happened. - Douglas Adams Atoms are the basic building blocks of matter that make up everyday objects. - A desk, the air, even you are made up of atoms. There are 90 naturally occurring kinds of atoms. - Scientists in labs have been able to make about 25 more. Living things are mostly made up of 4 elements - Oxygen - Carbon - Hydrogen - Nitrogen Atoms are ageless – Matter is conserved; it is neither created nor destroyed Define: Subatomic Particle_____________________________________________________________________ _____________________________________________________________________________ _____________________________________________________________________________ Element- _____________________________________________________________________ _____________________________________________________________________________ _____________________________________________________________________________ WebNotes: Go to the website http://web.jjay.cuny.edu/~acarpi/NSC/2-matter.htm and answer the following questions: I. Matter and Energy a) Everything in the universe is made of two things: _________________ and ____________________ b) Atoms are particles of elements, ______________________________________________________ c) Compound: ________________________________________________________________________ d) The difference between states of matter is due to __________________________________________ (skip Piza, click arrow at bottom right to move on to next page) II. Atomic Structure a) Atoms are made of 3 different particles (include description): i) ii) iii) Draw a picture of an atom with the three subatomic particles in the correct arrangement and label: b) What keeps the electron and proton from crashing into each other? What is a good example of this? (In your own words) c) Describe how far away an electron orbits from the nucleus in your own words d) It is not accurate to show an electron spinning around the nucleus like a ball in a circle because . . . e) Draw a more accurate way to describe the electron: f) In a neutral atom, the positively charged protons are balanced by _____________________________ Atomic Number = _________________________________________________________________________ Atomic Mass = ____________________________________________________________________________ Practice – use the information given to complete the chart. Element Oxygen Gold Potassium Sulfur Hydrogen Iron Iodine Uranium Krypton Silicon Erbium # # # protons electrons neutrons 8 8 8 79 79 118 19 20 16 0 26 30 53 92 146 Atomic Number 8 79 16 1 127 36 14 Atomic Mass 16 197 39 32 1 83 28 68 167