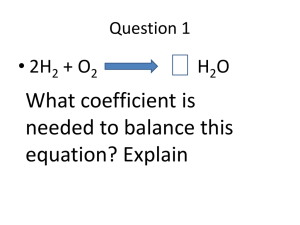
Atomic Theory Introduction: Atoms, Protons, Electrons
advertisement

Introduction to Atomic Theory - Atoms are the building blocks of all matter. From stars to humans to rocks to air, all of these things are composed of atoms. - Atoms are not complicated; they have only 3 parts… o Electrons which have a negative charge. o Protons which have a positive charge. o Neutrons which have no charge. - The idea of an atom has been around for a long time. Even around the 4th century BCE, the Greeks had a concept of the atom. - While the Greeks had an accurate concept of the atom, they had no experimental evidence (no tests) to support its existence. - Almost all the experimental evidence that supports “atomic theory” has accumulated over the last 200 years. - No one person deserves all the credit for “atomic theory,” many people, over a number of years, building on each others work, gave us the theory we have today. - This is because science is cumulative.