Carbonyl compounds Weekly plan 3
advertisement

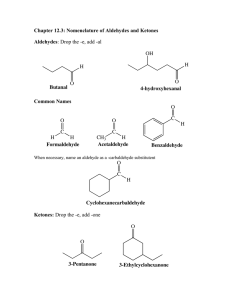
Weekly plan 3 Carbonyl compounds Student book links Specification links Link to AS specification Suggested time allowed (includes contact and non-contact time): • 1.1.9–12 • 4.1.2 (a)–(f) • 2.2.1 Alcohols Four to five hours Suggested teaching order Weekly learning outcomes Students should be able to: • Recognise and name aldehydes and ketones. • Describe the oxidation of primary alcohols to form aldehydes and carboxylic acids. • Describe the oxidation of secondary alcohols to form ketones. • Describe the oxidation of aldehydes to form carboxylic acids. • Describe the use of 2,4-dinitrophenylhydrazine (2,4-DNPH) to detect and identify a carbonyl compound. • Describe the use of Tollens’ reagent to detect the presence of an aldehyde group. • Describe the reduction of carbonyl compounds to form alcohols. • Outline the mechanism for nucleophilic addition reactions of aldehydes and ketones with hydride. Key words Aldehyde Functional group Electronegativity Carbonyl How Science Works Ketone Stem redox Suffix Reflux Recrystallisation Nucleophile 2,4-DNPH Tollens’ reagent Addition Hydride ion Oxidation Reduction Distillation Learning styles (S = Starter activities, M = Main activities, P = Plenary activities) Kinaesthetic Activities S1–3 Activities M1–3 Activities P1&3 1. The carbonyl group and the difference between aldehydes and ketones 2. The oxidation of primary alcohols to aldehydes and carboxylic acids 3. The oxidation of secondary alcohols to ketones 4. The oxidation of aldehydes to carboxylic acids 5. The reduction of aldehydes and ketones 6. The mechanism for the reaction of carbonyls with the H– ion in NaBH4 7. The use of 2,4-DNPH to detect and identify carbonyl compounds 8. The use of Tollens’ reagent to distinguish between aldehydes and ketones Interpersonal Activity S2 Activities M1–3 Activities P1–3 Auditory Activity S3 Activities M1–3 Activities P1&3 Visual Activities S1&3 Activities M1–3 Activity P1 • • HSW 1 & 4 The mechanism for nucleophilic addition HSW 5a & b Testing for and differentiating between aldehydes and ketones ICT activities • Prepare a PowerPoint presentation to show the nucleophilic addition reaction between carbonyls and the H– ion. The web links referred to here are some that the author has found personally helpful but are not intended to be a comprehensive list, many other good resources exist. © Pearson Education Ltd 2009 This document may have been altered from the original 1 Weekly plan 3 Suggested starter activities Equipment Teacher notes 1. Play What am I? Cards with names or structures on them Students must correctly identify them as aldehydes or ketones. 2. Play Pairs. Same cards as above, but each name has a matching structure on another card. Students play Snap or try to pair up the structure with the corresponding name. 3. Use molecular models to show the difference between aldehydes and ketones. Molecular model kits Can also be used to practise naming carbonyl compounds. Suggested main activities Equipment Teacher notes 1. Practical activity 2: The characteristic test for a carbonyl compound and the use of the 2,4-DNPH derivative to identify an unknown carbonyl compound See technician worksheet. See teacher worksheet. 2. Practical activity 3: Oxidation and reduction reactions of carbonyl compounds See technician worksheet. See teacher worksheet. 3. In groups, students produce a flow sheet to distinguish between aldehydes, ketones and alcohols via simple chemical tests. Carboxylic acids and esters may also be added. Suggested plenary activities Equipment Teacher notes 1. Students deliver their PowerPoint presentations. Data projector Again, the nucleophile and the carbonyl can be replaced with others. 2. Devise a key word quiz. Cards containing key words from the last three weekly plans Divide students into pairs or small groups, they then take turns to draw a card and must explain the word on the card. 3. Play Beach ball game. Large, plain beach ball Write keywords from this topic on the ball; more are added from lesson to lesson. Students sit in a circle and throw the ball to a friend – the recipient has to explain the word nearest their right thumb. © Pearson Education Ltd 2009 This document may have been altered from the original 2 Weekly plan 3 Homework suggestions • Construct a table that compares and contrasts the key properties of aldehydes and ketones. • Draw alcohol structures and their oxidation product when given a variety of alcohol names (primary, secondary and tertiary). • Compare reactions of non-polar C=C bond with the polar carbonyl bond. Cross-curriculum links • Biology – reducing sugars that are aldehydes Stretch and Challenge • Research how the 2,4-DNPH test was used to identify the alkenes in naturally-occurring rubbers via ozonolysis. • Research other nucleophilic addition reactions of aldehydes and ketones. Potential misconceptions • Students find it difficult to distinguish between aldehydes and ketones – if there is at least one hydrogen attached to the carbonyl carbon then it is an aldehyde. • In structures, aldehydes are written as –CHO and an alcohol as –CHOH. Notes © Pearson Education Ltd 2009 This document may have been altered from the original 3