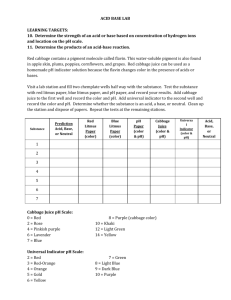
Red Cabbage PH Indicator

Red Cabbage PH
Indicator
GÜNEŞ KANAR
10C-70
Preview information;
• Red cabbage contains a pigment molecule called flavin (an anthocyanin).
• This water-soluble pigment is also found in apple skin, plums, poppies, cornflowers, and grapes. Very acidic solutions will turn anthocyanin a red color.
• Neutral solutions result in a purplish color.
• Basic solutions appear in greenish-yellow.
Therefore, it is possible to determine the pH of a solution based on the color it turns the anthocyanin pigments in red cabbage juice.
• The color of the juice changes in response to changes in its hydrogen ion concentration. pH is the -log[H+].
• Acids will donate hydrogen ions in an aqueous solution and have a low pH (pH < 7).
• Bases accept hydrogen ions and have a high pH (pH > 7).
Materials:
These are the materials that we are using during the experiment.
• red cabbage
• blender or knife
• boiling water
• filter paper
• One large glass beaker
• Six 250 mL beakers
•
• household ammonia (NH
3
) baking soda (sodium bicarbonate, NaHCO
3
)
• washing soda (sodium carbonate, Na
2
• lemon juice (citric acid, C
6
H
8
O
7
)
CO
3
)
• vinegar (acetic acid, CH
3
COOH)
• seltzer water (carbonic acid, H
2
CO
3
)
• muriatic acid or masonry's cleaner (hydrochloric acid, HCl)
• lye (potassium hydroxide, KOH or sodium hydroxide, NaOH)
Procedure;
• Chop the cabbage into small pieces .
• Place the cabbage in a large beaker and add boiling water to cover the cabbage. Allow at least ten minutes for the color to leach out of the cabbage.
• Filter out the plant material to obtain a redpurple-bluish colored liquid. This liquid is at about pH 7.
• Pour about 50 - 100 mL of your red cabbage indicator into each 250 mL beaker.
• Add various solutions to your indicator until a color change is obtained.
• Use separate containers for each household solution .
• Be amazed as the beautiful colors dazzle and delight your eyes.
Results:
• Your red cabbage juice is a simple pH tester. It reacts differently to different substances.
• Once you know what colour the juice turns in acids and alkalis, you can use it to test other liquids.
• Why It Happens:
• Red cabbage has pigments which react differently to acids and alkalis.
• When you dip the strips into a substance and wait for a few minutes, the colour will fully develop.
• The indicator strips turn red-yellow in acid, green in neutral and purple-blue in alkali.