The Extraordinary Properties of Water
advertisement

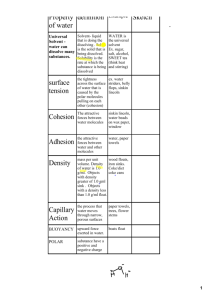
The Properties of Water Adapted from Biology Junction Water A water molecule (H2O), is made up of three atoms. one oxygen and two hydrogen H O H Water is Polar Water is an overall neutral molecule because it has an equal number of electrons and protons. The oxygen end “acts” negative The hydrogen end “acts” positive This is why water is described as POLAR Interaction Between Water Molecules The negative oxygen end of one water molecule is attracted to the positive hydrogen end of another water molecule. This is why water molecules are attracted to each other. This is called COHESION. Cohesion results in SURFACE TENSION -a measure of the strength of water’s surface Produces a surface film on water that allows insects to walk on the surface of water. In summary… A water molecule has a positive and negative end (polar), so… water molecules are attracted to water molecules (cohesion) and… this creates surface tension. Jesus Christ Lizard Adhesion is the attraction between two different substances. Water molecules will make bonds with other surfaces such as glass, soil, plant tissues, and cotton. Adhesion causes Capillary Action Capillary Action occurs when water molecules “tow” each other along when in a thin tube. Transpiration is a form of capillary action where plants and trees remove water from the soil, and paper towels soak up water. In summary… Water molecules have the ability to “climb” structures (adhesion) and this can result in capillary action. Adhesion also causes water to… Hold onto plant leaves (the droplets form due to cohesion) Attach to a silken spider web Meniscus Turgor Pressure Celery Turgor pressure is the pressure that happens when the water located inside the cells begins to expand, pushing on the cell membranes or walls. Turgor pressure is important to the plant cells because it is what gives them the rigidness to stand straight and continue normal cellular functions. If a plant is not able to access enough water to maintain the turgid state then it will begin to wilt, which interferes with function. Water is called the UNIVERSAL SOLVENT More substances dissolve in water than in any other chemical. This is also caused by water’s polarity. The positive part of an ionic compound is attracted to the oxygen side of a water molecule, and the the negative part of an ionic compound is attracted to the hydrogen side. Water is called the UNIVERSAL SOLVENT Water is the solvent for biochemical processes. Hemoglobin, carbonates, various proteins, and many other molecules in the body use water as a solvent. Water has High Heat Capacity Specific Heat Capacity is the amount of heat needed to raise or lower 1g of a substance 1°C Water resists temperature change, both for heating and cooling. This allows water to act as a buffer for body temperature. Water is Less Dense as a Solid Ice is less dense as a solid than as a liquid. Frozen water (ice) floats on liquid water. Frozen water forms a crystal-like lattice where molecules are set at fixed distances. Water is Less Dense as a Solid Which is ice and which is water? Water is Less Dense as a Solid Water Ice This is important for organisms that live underwater. If frozen water sunk, small bodies of water would be more likely to freeze completely in the winter, which would be bad for all the organisms living there. A layer of ice effectively insulates the underlying water, allowing many aquatic organisms to survive through the winter. Homeostasis the ability to maintain a steady state despite changing conditions Water is important to this process because: Human Tissue % Water Blood 83.0 Heart 79.2 Muscle 75.6 Brain -Water is a good insulator -Water resists temperature change Skin Bone -Water is a universal solvent -Ice protects against temperature extremes (insulates frozen lakes) 74.8 72.0 22.0