Guided Notes - Properties of Water
advertisement

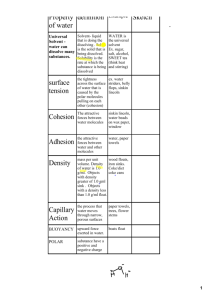
Describe Water Properties of Water Key Properties of Water • Key properties of water include: – states of matter – polarity – cohesion and adhesion – capillary action – high surface tension – universal solvent – high specific heat – density and buoyancy – high heat of vaporization The Structure of Water ● Just like all matter, water is made of ________. ● Atoms attach together, or ________, to form ______________. ● ____ hydrogen atoms bonded to an ________ atom form a water molecule. ● A short way of writing this is to use the chemical formula for water: ____. • How would you describe water to someone who had never seen it before? • You might say that pure water has no _______, no ________, and no _________. You might even say that water is a rather plain, ordinary substance. • If you asked a chemist to describe water, the chemist would say that water is very ________. Its properties ________ from those of most other familiar substances. States of Matter • Water covers about _____ of the Earth’s surface. • Water is the ______ substance on Earth that occurs ________ as a solid, liquid and gas. The Atomic Structure of Water ● This shows how the hydrogen and oxygen atoms are arranged in a water molecule. ● Each end of the molecule has a slight ____________ charge. ● The oxygen end has a slight ____________ charge. ● The hydrogen ends have a slight ____________ charge. Polarity ● _______ molecule: a molecule that has electrically charged ____________ Polarity • The positive end of one water molecule is ____________ to the negative end of another molecule. • Water molecules tend to ____________ together. ● Because water consists of polar water molecules, it is called a polar ____________. Cohesion and Adhesion • ____________: water molecule attracted to another ____________ molecule • ____________: water molecule attracted to another Cohesion and Adhesion • Cohesion causes the formation of ____________. • Adhesion causes the ____________ to stick to a ____________ _________________ Capillary Action Capillary Action • The next time you see a drink with a straw in it, look closely at the level of the liquid outside and inside the straw. You will see that the liquid rises ____________ ____________ the straw. • As water molecules are attracted to the ____________, they ____________ other water molecules up with them. Similarly, water will climb up into the ____________ of a brick or a piece of wood. • ____________ and ____________ work together to cause capillary action. • ____________ action: the ____________ force of ____________ among water molecules and with the molecules of surrounding materials – Allows water to move ____________ materials with pores inside. Capillary Action Surface Tension • Have you ever watched water striders skate across the ____________ of a pond without ____________? • They are supported by the surface ________ of the water. Surface Tension Surface Tension • Surface tension: the____________ across the surface of water that is caused by the ________ molecules pulling on one another. • The molecules at the surface are being ____________ by the molecules _________ to them and _________ them. • The pulling forces the surface of the water into a curved shape. Surface tension also causes raindrops to form ____________ beads when they fall onto a car windshield. Japanese 1 yen aluminum coin on water Universal Solvent Universal Solvent • What happens when you make a fruit drink from a powdered mix? Or stir sugar into tea? • As you stir the powder into the __________, the powder seems to disappear. When you make the fruit drink, you are making a ____________. • A ____________ is a mixture that forms when one substance dissolves another. • The substance that does the dissolving is called the ____________. Universal Solvent • Water is known as the ____________ solvent because ____________ substances dissolve in it. • Because of this, water is ____________ found in a ____________ ____________ condition. Buoyancy • ____________: ____________ force that keeps things afloat Density • Density: relationship between the ________ of the substance and how much _________ it takes up (volume). – The mass of atoms, their _________, and how they are ______________ determine the density of a substance. Specific Heat • It is a steamy summer day. The air is hot, the sidewalk is hot, and the sandy beach is hot. • But when you jump into the ocean, the water is surprisingly _________! If you go for an evening swim, however, the water is _________ than the cool air. Specific Heat Specific Heat • _________ heat: the amount of heat _________ needed to increase the temperature of a certain mass of a substance by ______. • Compared to other substances, water requires a _____ of heat to increase its temperature. • Water’s high specific heat is due to the strong __________ among water molecules. • Other substances, such as air and rocks, have _________ attractions between their molecules. – The temperature of each of these substances rises more _________ than that of water that is heated the _________ amount. Specific Heat High Heat of Vaporization A large body of water can act as a heat sink -_________ heat from sunlight during the day and summer and _________ heat during the night and winter as the water gradually cools. As a result: • Heat of vaporization: the amount of energy required to _________ one gram of a liquid substance to a gas (__________________) • Due to strong ____________, water can absorb a lot of _________ before molecules evaporate. • Water, which covers three-fourths of the planet, keeps temperature _________ within a range suitable for life. • Coastal areas have _________ climates than inland. • The marine environment has a relatively _________ temperature. Benefits of High Heat of Vaporization • _________ the earth's climate. – _________ cooling: solar heat absorbed by tropical seas dissipates when surface water evaporates – As moist tropical air moves toward the poles, water vapor releases _________ as it condenses into rain. Benefits of High Heat of Vaporization • Evaporative cooling also – _____________ temperature in aquatic ecosystems – Helps organisms _________ overheating (when you sweat to cool off) Key Properties of Water • Review: – states of matter – polarity – cohesion and adhesion – capillary action – high surface tension – universal solvent – high specific heat – density and buoyancy – high heat of vaporization