FIB notes- Water structure and properties
advertisement

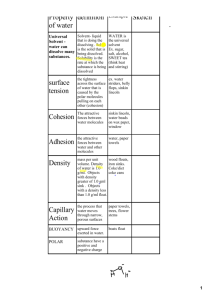
Structure & Properties of Water Structure 1 molecule of water is made up of _________________________________ bonded with ___________________________. The bond that forms water is a __________________________(between the oxygen and hydrogen atoms). Chemical formula: __________ ________________________ of molecules of water make up _________________________. Polar Molecule A molecule that has ____________________________ areas. Also called a _______________________ or a polar ____________________. ___________________________ substances do NOT have _______________ ends. The positive ______________________ends of 1 molecule are attracted to the ________________________ of the ________________ of another molecule. The properties of: cohesion, adhesion, specific heat, universal solvent, capillary action, and surface tension all happen because water is a ________________________________! Capillary Action The combines force of ______________________________________ ____________________ and with the molecules of the ___________ _______________________. _________________________ + __________________________ Surface Tension The _______________________ across the surface of water that is caused by the ______________________________ pulling on one another. Makes the surface act like a _________________. Universal Solvent This is the ____________________ given to water. Water dissolves LOTS of things. Remember: SOLVENT: _____________________________________ SOLUTE: ________________________________________________ Other ______________ molecules can be dissolved by water. Non-polar substances _________________ dissolve in water. Why is water so good at dissolving things? __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ ______________________________________. Cohesion Because water is a polar molecule, it is ________________________ _______________________. Cohesion- the attractive force ________________________________ Water molecules like to _____________________________________. Rubbing alcohol doesn’t like to _______________________________ As much as water. Adhesion Because water is a polar molecule, it is attracted to _______________ substances. Adhesion- ________________________________________________ ________________________________________________________. Picture of adhesion: Adhesion is going on between the _____________ and the _________________. _________________________ ________________________ Water molecules like to Water molecules like to stick stick to each other. to other things. Ex: Ex: Specific heat Specific heat- the amount of energy _______________ to ___________________ the temperature of something ____________. Water has a _________________________ specific heat. That means it takes a lot of energy for water to ___________________ _________________________________. This is because of the ______________________________ between water molecules. It’s POLAR! Density of ice vs. water Ice is ________________ than liquid water.