Water Properties PPT Guided Notes
advertisement

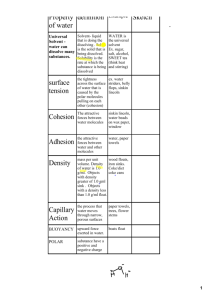
Name: ____________________________ Date: ________________________ Properties of Water Guided Notes 1. #1Polarity: Water molecules have a slightly _____________charge near the ______________ atom and a slightly ___________________ charge around the _____________________ atom. 2. #2 Universal Solvent: Water _____________ more substances than sulfuric acid because of its _________________ characteristic. It can _________________ molecules of a substance and pull it apart. 3. #3 Cohesion : Water is attracted to other ________________________ molecules. 4. #4 Adhesion: Water can also be attracted to other different ____________________. 5. #5 Density: Ice is less dense than __________________ which is why ice floats. It _____________________ as it freezes allowing fish to live in water during winter 6. #6 High Specific Heat Capacity: Water can take a lot of heat without its________________ rising much. Our ____________________ are about 85% water and this allows us to withstand temperature changes when we go outside. 7. #7 Neutral pH: Water has a pH of approximately _____________which means it is neither ____________________ or ______________________. 8. #8 Surface Tension: __________________of water molecules at the surface of a body of water causes the water to pull itself into a shape with the _________________ amount of surface area (sphere). _____________ _______________allows water striders to 'skate' across the top of a pond. (Cohesion) 9. #9 Capillary Action: Placing a straw into a glass of water, it ________________ up the straw. Water molecules are attracted to the _________________ molecules (adhesion). When one ________________ molecule moves, the other water molecules _________________ (cohesion) . Capillary action is limited by ________________ and the _______________ of the straw. Plants take advantage of capillary action to pull _______________ into their _________________. 10. Cohesion or Adhesion? Explain the picture.