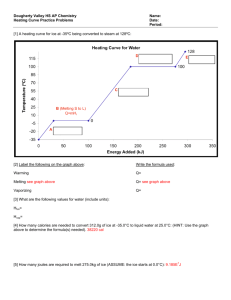
Heat in Changes of State
advertisement

Heat in Changes of State Solids, Liquids, and Solutions Introduction • When substances change state, there is a transfer of energy. Introduction • When substances change state, there is a transfer of energy. • The evaporation of sweat helps to regulate our body temperature in warm weather or when we exercise. Introduction • When substances change state, there is a transfer of energy. • The evaporation of sweat helps to regulate our body temperature in warm weather or when we exercise. Introduction • When substances change state, there is a transfer of energy. • • The evaporation of sweat helps to regulate our body temperature in warm weather or when we exercise. The solidification of a liquid results when there is a transfer of heat from the liquid to the surroundings. Introduction • When substances change state, there is a transfer of energy. • • The evaporation of sweat helps to regulate our body temperature in warm weather or when we exercise. The solidification of a liquid results when there is a transfer of heat from the liquid to the surroundings. Introduction • When substances change state, there is a transfer of energy. • • The evaporation of sweat helps to regulate our body temperature in warm weather or when we exercise. The solidification of a liquid results when there is a transfer of heat from the liquid to the surroundings. Heats of Fusion and Solidification • When we place an ice cube on a table in a warm room Heats of Fusion and Solidification • When we place an ice cube on a table in a warm room Heats of Fusion and Solidification • When we place an ice cube on a table in a warm room, it begins to melt. Heats of Fusion and Solidification • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. Heats of Fusion and Solidification • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. Heats of Fusion and Solidification • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. Heats of Fusion and Solidification • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. Heats of Fusion and Solidification • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. Heats of Fusion and Solidification • • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. There is no change in temperature until all of the solid is converted to liquid. Heats of Fusion and Solidification • • • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. There is no change in temperature until all of the solid is converted to liquid. This is an endothermic process ... Heats of Fusion and Solidification • • • • • When we place an ice cube on a table in a warm room, it begins to melt. The ice cube is the system and the table, room, and the air in the room are the surroundings. The system takes in energy from the surroundings to convert the solid ice to liquid water. There is no change in temperature until all of the solid is converted to liquid. This is an endothermic process ... • that is, ∆H is positive. Heats of Fusion and Solidification • When we make ice cream ... Heats of Fusion and Solidification • When we make ice cream ... Heats of Fusion and Solidification • When we make ice cream ... • the ice cream mix is the system and the salt/ice mixture and the ice cream maker are the surroundings Heats of Fusion and Solidification • When we make ice cream ... • • the ice cream mix is the system and the salt/ice mixture and the ice cream maker are the surroundings energy is transferred from the system to the surroundings Heats of Fusion and Solidification • When we make ice cream ... • • • the ice cream mix is the system and the salt/ice mixture and the ice cream maker are the surroundings energy is transferred from the system to the surroundings Once the liquid gets to the freezing point, there is no further change in temperature until all of the mix has frozen. Heats of Fusion and Solidification • When we make ice cream ... • • • • the ice cream mix is the system and the salt/ice mixture and the ice cream maker are the surroundings energy is transferred from the system to the surroundings Once the liquid gets to the freezing point, there is no further change in temperature until all of the mix has frozen. This is an exothermic process ... Heats of Fusion and Solidification • When we make ice cream ... • • • • the ice cream mix is the system and the salt/ice mixture and the ice cream maker are the surroundings energy is transferred from the system to the surroundings Once the liquid gets to the freezing point, there is no further change in temperature until all of the mix has frozen. This is an exothermic process ... • that is, ∆H is negative. Heats of Fusion and Solidification • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. Heats of Fusion and Solidification • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. Heats of Fusion and Solidification • • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. For any given substance, ∆Hfus = −∆Hsolid. Heats of Fusion and Solidification • • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. For any given substance, ∆Hfus = −∆Hsolid. Liquid ∆Hfus ∆Hsolid Solid Heats of Fusion and Solidification • • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. For any given substance, ∆Hfus = −∆Hsolid. • Liquid ∆Hfus ∆Hsolid Solid For example, for H2O, going from ice to water, ∆Hfus = 6.01 kJ/mol Heats of Fusion and Solidification • • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. For any given substance, ∆Hfus = −∆Hsolid. • Liquid ∆Hfus ∆Hsolid Solid • For example, for H2O, going from ice to water, ∆Hfus = 6.01 kJ/mol For H2O to go from water to ice, ∆Hsolid = −6.01 kJ/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Tinitial = Tfinal = 0°C Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Tinitial = Tfinal = 0°C MH2O = 18.0 g/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Find: mH2O = ? g Tinitial = Tfinal = 0°C MH2O = 18.0 g/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Find: mH2O = ? g Calculate: Tinitial = Tfinal = 0°C MH2O = 18.0 g/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Find: mH2O = ? g Calculate: 2.25 kJ 1 ∆H Tinitial = Tfinal = 0°C MH2O = 18.0 g/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol ∆H = 2.25 kJ/mol Find: mH2O = ? g Calculate: 2.25 kJ 1 × 1 mol 6.01 kJ ∆Hfus Tinitial = Tfinal = 0°C MH2O = 18.0 g/mol Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol Tinitial = Tfinal = 0°C ∆H = 2.25 kJ/mol Find: MH2O = 18.0 g/mol mH2O = ? g Calculate: 2.25 kJ 1 × 1 mol 6.01 kJ × 18.0 g 1 mol MH2O Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol Tinitial = Tfinal = 0°C ∆H = 2.25 kJ/mol Find: MH2O = 18.0 g/mol mH2O = ? g Calculate: 2.25 kJ 1 × 1 mol 6.01 kJ × 18.0 g 1 mol = 6.74 g mH2O Heats of Fusion and Solidification Sample Problem 17.4 (page 521) How many grams of ice at 0°C will melt if 2.25 kJ of heat are added? Known: ∆Hfus = 6.01 kJ/mol Tinitial = Tfinal = 0°C ∆H = 2.25 kJ/mol Find: MH2O = 18.0 g/mol mH2O = ? g Calculate: 2.25 kJ 1 × 1 mol 6.01 kJ × 18.0 g 1 mol = 6.74 g mH2O Heats of Vaporization and Condensation • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. Heats of Vaporization and Condensation • • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. Heats of Vaporization and Condensation • • • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. For any given substance, ∆Hvap = −∆Hcond. Heats of Vaporization and Condensation • • • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. For any given substance, ∆Hvap = −∆Hcond. Gas ∆Hvap ∆Hcond Liquid Heats of Vaporization and Condensation • • • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. For any given substance, ∆Hvap = −∆Hcond. • Gas ∆Hvap ∆Hcond Liquid For example, for H2O, going from water to steam, ∆Hvap = 40.7 kJ/mol Heats of Vaporization and Condensation • • • The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. For any given substance, ∆Hvap = −∆Hcond. • Gas ∆Hvap ∆Hcond Liquid • For example, for H2O, going from water to steam, ∆Hvap = 40.7 kJ/mol For H2O to go from steam to water, ∆Hcond = −47.0 kJ/mol Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. Heats of Vaporization and Condensation If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. Temperature • Heat Supplied Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. Temperature boiling point melting point Heat Supplied Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. Temperature boiling point ∆Hfus melting point Heat Supplied Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. Temperature boiling point ∆Hfus melting point Heat Supplied Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. ∆Hvap Temperature boiling point ∆Hfus melting point Heat Supplied Heats of Vaporization and Condensation • If we look at the change in temperature of a substance as we add heat at a constant rate to a solid until it becomes a liquid and then a gas, we get the following heating curve. ∆Hvap Temperature boiling point ∆Hfus melting point Heat Supplied Heat of Solution • During the formation of a solution, heat is either released or absorbed. Heat of Solution • During the formation of a solution, heat is either released or absorbed. • This is the molar heat of solution, ∆Hsoln. Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs and cold packs. Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs and cold packs. Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs and cold packs. The dissolution of calcium chloride in water is exothermic. • • CaCl2(s) → Ca2+(aq) + 2 Cl−(aq) ∆Hsoln = −82.8 kJ/mol Heat of Solution • During the formation of a solution, heat is either released or absorbed. • • • This is the molar heat of solution, ∆Hsoln. This is the basis of heat packs and cold packs. The dissolution of calcium chloride in water is exothermic. • • • CaCl2(s) → Ca2+(aq) + 2 Cl−(aq) ∆Hsoln = −82.8 kJ/mol The dissolution of ammonium nitrate in water is endothermic. • • NH4NO3(s) → NH4+(aq) + NO3−(aq) ∆Hsoln = +25.7 kJ/mol Heat of Solution • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Heat of Solution • • • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Knowns: n = 2.50 mol ∆Hsoln = −445.1 kJ/mol Heat of Solution • • • • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Knowns: n = 2.50 mol ∆Hsoln = −445.1 kJ/mol Find: ∆H = ? kJ Heat of Solution • • • • • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Knowns: n = 2.50 mol ∆Hsoln = −445.1 kJ/mol Find: Calculation: ∆H = ? kJ ∆H = n × ∆Hsoln Heat of Solution • • • • • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Knowns: n = 2.50 mol ∆Hsoln = −445.1 kJ/mol Find: Calculation: ∆H = ? kJ ∆H = n × ∆Hsoln = (2.50 mol)(−445.1 kJ/mol) Heat of Solution • • • • • • Sample Problem: How much heat is released when 2.50 mol of NaOH is dissolved in water. ∆Hsoln = −445.1 kJ/mol Knowns: n = 2.50 mol ∆Hsoln = −445.1 kJ/mol Find: Calculation: ∆H = ? kJ ∆H = n × ∆Hsoln = −1113 kJ Summary • • • • • The amount of heat absorbed by one mol of a solid to become one mol of a liquid is the molar heat of fusion, ∆Hfus. The amount of heat given up by one mol of a liquid to become one mol of a solid is the molar heat of solidification, ∆Hsolid. • For any given substance, ∆Hfus = −∆Hsolid. The amount of heat absorbed by one mol of a liquid to become one mol of a gas is the molar heat of vaporization, ∆Hvap. The amount of heat given up by one mol of a gas to become one mol of a liquid is the molar heat of condensation, ∆Hcond. • For any given substance, ∆Hvap = −∆Hcond. During the formation of a solution, heat is either released or absorbed. • This is the molar heat of solution, ∆Hsoln.