8th grade ch. 6 lp
advertisement

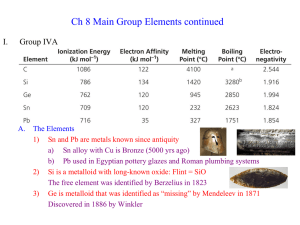
Describing Chemical Reactions Ch. 6 What are chemical equations • Chemical equations use chemical formulas and other symbols instead of words to summarize a reaction. Formulas in the equation • All chemical equations use formulas to represent substances involved in a reaction. • Ex. CO2 is a formula for carbon dioxide. Structure of an equation • Reactants ----> products • The number of reactants and products can vary. • CaCO3 ------> CaO + CO2 Conservation of Mass • Matter is not created or destroyed. • The principal of conservations of mass states that in a chemical reaction, the total mass of the reactants must equal the total mass of the products. (the same on each side) Steps for balancing equations • • • • 1. Write the equation 2. Count the atoms 3. Use coefficients to balance atoms 4. Look back and check • H2 + O2 -----> H2O Balance the equations • Fe2O3 + C ----> Fe + CO2 • SO2 + O2 ----> SO3 • Fe + HCL ---> FeCl2 + H2 • N2 + O2 ----> N2O3 • MgO + HBr ----> MgBr2 + H2O