13.2-13.3 The Rate of Chemical Reactions – The Rate Law
advertisement

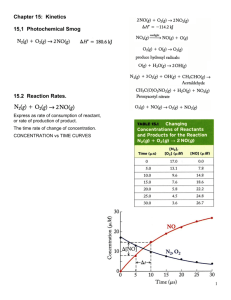
13.2-13.3 The Rate of Chemical Reactions – The Rate Law The Rate of a Chemical Reaction • Chemical Kinetics – The study of how things that happen on a molecular level over time, affect the macroscopic world. • Reaction Rate – A measure of how fast a reaction occurs. – A fast rate = a large fraction of molecules react to form products in a given period of time. – A slow rate = a small amount of molecules react to form products in a given period of time. – The change in reactant concentration decreases over time, because reactants are consumed in a chemical reaction. – The product concentration increases over time, because products are formed. – Reaction rates are reported as positive quantities. (Example on next slide ) A Few Formulas Showing Reaction Rates The following is a synthesis (combination) reaction: H2(g) + I2(g) 2HI(g) The rate of reactant concentrations over time: Δ[H2] [H2]t2 – [H2]t1 Rate = – --------- = – --------------------Δt t 2 – t1 *The brackets symbolize concentration since the concentration of the reactants decrease over time, a negative sign is placed in the formula to keep the reaction rate positive. I2 would be written the same way. The rate of product concentration over time: The factor of one half is based on the stoichiometric relationship in this particular reaction. In order to have a single rate for the entire reaction, the concentration of HI must be multiplied by a factor of ½. Δ[HI] [HI]t2 – [HI]t1 Rate = + ½ --------- = + ½ --------------------Δt t 2 – t1 The Average Rate of Reactions • The average rate of the reaction between any two time intervals can be calculated using the previous equations. • What you would notice is that over time, as the concentration of the reactant decrease, the rate of the reaction slows down. This is important, because this describes one of the factors that affect reaction rate. Instantaneous Reaction Rate • The instantaneous reaction rate could also be calculated by looking at a reaction rate curve, and calculating the slope tangent to the curve at a specific point of interest. • The instantaneous rates are the same whether one reactant or product is used for the calculation. Reaction Rate Generalization for a Generic Reaction aA +bB cC + dD 1 Δ[A] 1 Δ[B] 1 Δ[C] 1 Δ[D] Rate = – --- -------- = – ---- ------ = + --- ------ = + --- -----a Δt b Δt c Δt d Δt Let’s try a practice problem! For the following reaction, predict the rate of change in concentration of H2O2 (Δ[H2O2]/ Δt) and I3(Δ[I3-]/ Δt) during the first 10 s. (Given: the average rate of the reaction is 4.40X10-3 M/s) this was calculated based on the concentration of I- dropping from 1.00 M to 0.868 M in the first 10 s. H2O2(aq) + 3I-(aq) + 2H+(aq) I3-(aq) + 2H2O(l) The change in concentration for both H2O2 and I3- would be exactly the same: 4.40X10-3 M/s. Let’s Try Another! For the reaction A + 2B C under a given set of conditions, the initial rate is 0.100 M/s. What is Δ[B]/Δt under the same conditions. (a) -0.0500 M/s (b) -0.100 M/s (c) -0.200 M/s (c) -0.200 M/s The Rate Law: The Effect of Concentration on Reaction Rate • The rate of a reaction often depends on the concentration of one or more reactants. • In a simple decomposition reaction where A products, the rate law (the relationship between the concentration of reactants and the rate of the reaction) can be expressed using: Rate = k[A]n where K is the rate constant, and n is the reaction order. The value of n (usually an integer), determines how the rate depends on the concentration of the reactants. Defining Rate Order • If n = 0, the reaction is zero (or zeroth) order and the rate is independent of the concentration of A. • Rate = k[A]0 = k • Here the slope of the line decreases linearly with time (constant rate). • If n = 1, the reaction is first order and the rate is directly proportional to the concentration of A. • Rate = k[A]1 • If n = 2, the reaction is second order and the rate is proportional to the square of the concentration of A. • Rate = k[A]2 • There are fractional rate orders that do occur, but are rare. Let’s Try a Practice Problem! Consider the equation: NO2(g) + CO(g) NO(g) + CO2(g) The initial rate of reaction is measured at several different concentrations of the reactants with the following results: [NO2] M [CO] M Initial Rate (M/s) 0.10 0.10 0.0021 0.20 0.10 0.0082 0.20 0.20 0.0083 0.40 0.10 0.033 From the data, determine: a.) the rate law for the reaction b.) the rate constant (k) for the reaction a.) Rate = k[NO2]m[CO]n = k[NO2]2[CO]0 = K[NO2]2 b.) k = Rate / [NO2]2 = ( (0.0021 M/s) / (0.10 M)2) = 0.21 M-1s-1 Determining the Order of a Reaction • The order of a reaction can be determined only by experiment, or analysis of experimental data. Yes, you will most likely have to analyze data to determine the rate order, and the rate constant of a reaction. • Reaction Order for Multiple Reactants: • For the generic reaction: aA +bB cC + dD where there is more than one reactant, the rate law can be determined by the following formula: Rate = k[A]m[B]n m is the reaction order with respect to A and n is the reaction order with respect to B. The sum of the exponents m and n is the overall order. 13.2-13.3 pgs.638-639 #’s 26, 28, 32, and 43 Read 13.4 pgs. 607-614