“K” Chemistry (part 1 of 3) Chapter 13:
advertisement

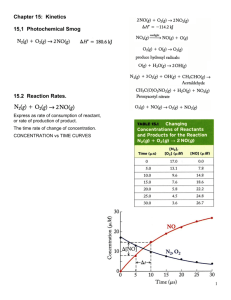
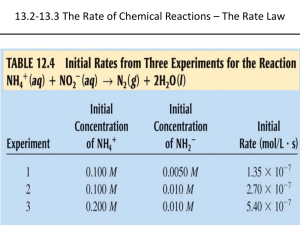
“K” Chemistry (part 1 of 3) Chapter 13: Reaction Rates and Kinetics Reaction Rates • The rate of a chemical reaction is a measure of how fast the reaction occurs • A slow reaction will have a small fraction of molecules reacting and forming products • A fast reaction will have a large fraction of molecules reaction and forming products • As the reactants react and form the products, the concentrations of each change as a function of time Rate Calculations • For a balanced chemical reaction: aA + bB cC + dD Practice • Consider the following balanced chemical equation: H2O2(aq) + 3 I-(aq) + 2 H+(aq) I3-(aq) + 2 H2O(l) • In the first 10.0 seconds of the reaction , the concentration of I- dropped from 1.000 M to 0.868 M. (a) Calculate the average rate of this reaction in this time interval (b) Predict the rate of change in the concentration of H+ (Δ[H+] / Δ t) during this time interval Practice • Using the above reaction, predict the rate of change in concentration of hydrogen peroxide and the I3-1 during the same time interval Rate Laws • The rate of a reaction depends on the concentration of one or more of the reactants • For example (a simple decomposition reaction): A Products • As long as the reverse reaction is negligibly slow (or non existent), we can write a relationship (a rate law) Rate = k [A]n Rate Laws Rate = k [A]n • k is the proportionality constant called the rate constant • n is a number called the reaction order – IT IS NOT NUMBER OF MOLES OR THE STOICHIOMETRY COEFFICIENT!!! – It MUST be determined experimentally!! Reaction Order, n • If n = 0, the reaction is zero order and the rate is independent of the concentration of A – By mathematical definition, [A]0 = 1 so the rate law (Rate = k [A]0) is equal to k regardless of the concentration of A • If n = 1, the reaction is first order and the rate is directly proportional to the concentration of A • If n = 2, the reaction is second order and the rate is proportional to the square of the concentration of A Zero-Order Reaction Rate = k [A]0 = k • For a zero order reaction, the concentration of the reactant decreases linearly with time • The rate is constant because it does not slow down as the [A] decreases First-Order Reaction Rate = k [A]1 • For a first order, the rate is directly proportional to the concentration of the reactant • Consequently, the rate slows as the concentration of the reactant decreases Second-Order Reaction Rate = k [A]2 • For a second order reaction, the rate of the reaction is proportional to the square of the concentration of the reactant • Consequently, the rate is even more sensitive to the reaction concentration Quick Tip • The “order of the reactant” refers to only the reactant of focus A Products Rate = k [A]n • This reaction is nth order with respect to “reactant A” • A “reaction order” is the sum of all the exponents Determining the Order of the Reaction • The order of the reaction can only be determined by experiment • Uses a method called the method of initial rates – The rate for a short period of time at the beginning of the reaction is measured using different initial reactant concentrations to determine the effect of [A] has on the rate Method of Initial Rates [A] (M) Initial Rate (M/s) 0.10 0.015 0.20 0.030 0.40 0.060 • Notice for this data: – When the concentration of A doubles, the rate is directly proportional… the rxn is therefore first order with respect to “reactant A” The Value of the Rate Constant, k • The rate constant, k, can be calculated using this experimentally determined data after the determination of the orders (the exponents) Rate = k [A]1 k = rate/[A] = (0.015 M*s-1) / (0.10M) = 0.15s-1 • NOTICE THAT: the rate constant for a first-order reaction is s-1 Data for Zero-Order [A] (M) Initial Rate (M/s) 0.10 0.015 0.20 0.015 0.40 0.015 • Notice for this data: – The initial rate is independent for the reactant concentration – the rate is the same at all measured initial concentrations Data for Second-Order [A] (M) Initial Rate (M/s) 0.10 0.015 0.20 0.060 0.40 0.240 • Notice for this data: – The initial rate quadruples for a doubling of a reactant concentration. The relationship between concentration and rate is quadratic. KEY THINGS!! • The rate constants for zero- and second-order reactions have different units than first-order NOTICE THAT: – the rate constant for a zero-order reaction is M*s-1 – the rate constant for a first-order reaction is s-1 – the rate constant for a second-order reaction is M-1*s-1 Reaction Order for Multiple Reactants (not a simple decomp.) aA + bB cC + dD • Again, as long as the reverse is negligibly slow (or non-existent), the rate law can be defined as: Rate = k [A]m[B]n • m is the reaction order with respect to A • n is the reaction order WRT B • The overall order is the sum of the exponents (m+n) Example • The reaction between hydrogen gas and iodine has been experimentally determined to be first order WRT both reactants… and thus second order OVERALL H2(g) + I2(g) 2HI(g) Rate =k[H2]1[I2]1 • The exponents ARE NOT THE COEFFICIENTS!! Example • The reaction between hydrogen and nitrogen monoxide has been experimentally determined to be first order WRT hydrogen and second order WRT to nitrogen monoxide 2H2(g) + 2NO(g) N2(g) + 2H2O(g) Rate = k[H2]1[NO]2 **Notice the exponents are the ‘experimentally determined’ orders… NOT THE COEFFICIENTS!! Practice Calculating Orders Consider the following reaction between NO2 and CO: NO2(g) + CO(g) NO(g) + CO2(g) The initial rate of the reaction was measured at several different concentrations if the reactants with the following results: [NO2] (M) [CO] (M) Initial Rate (M/s) 0.10 0.10 0.0021 0.20 0.10 0.0082 0.20 0.20 0.0083 0.40 0.10 0.033 • Calculate the rate law for the reaction AND the rate constant (k) [NO2] (M) [CO] (M) Initial Rate (M/s) 0.10 0.10 0.0021 0.20 0.10 0.0082 0.20 0.20 0.0083 0.40 0.10 0.033 • Look for anything held constant!! – If you compare the rates while one reactant is held constant than you can assume the reaction rate is CAUSED by the reactant that is changing!! • The [NO2] doubled while the rate quadrupled – Implies this rxn is second order WRT to NO2 • The [CO] (while the [NO2] is constant) doubles while the rate is unchanged… implying zero order [NO2] (M) [CO] (M) Initial Rate (M/s) 0.10 0.10 0.0021 0.20 0.10 0.0082 0.20 0.20 0.0083 0.40 0.10 0.033 Rate = k[NO2]2[CO]0 = k[NO2]2 • Now, calculate the k – plug in any two corresponding data points = 0.21 M-1*s-1 **Notice the UNITS!! Practice #2 • Consider the following reaction: CHCl3(g) + Cl2(g) CCl4(g) + HCl(g) [CHCl3] (M) [Cl2] (M) Initial Rate (M/s) 0.010 0.010 0.0035 0.020 0.010 0.0069 0.020 0.020 0.0127 0.040 0.040 0.027 Calculate the rate law for the reaction AND the rate constant (k) Half Life (t1/2) • Read through pages 584-587 and work out the half life problems – In a nutshell, the half life is the time it takes for half of a sample to disappear (break down / decompose) RATE LAW SUMMARY • Effects of Temperature – • Effects of Pressure (for gases) – • Activation Energy – • Catalysis – – Enzymes – • Collision Theory – • Rate Determining Step – End of Chapter 13 • WORK THROUGH THE PRACTICE PROBLEMS!!!