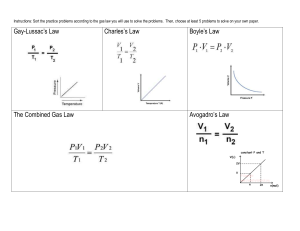
Unit 5 Test 1 Review
advertisement

Today is Thursday, December 17th, 2015 Pre-Class: We’re spending a large portion of today reviewing for the test tomorrow. Ask questions now or forever hold them or something... THINGS TO DO: -Have the Specific Heat and Thermochemistry Round Robin Review worksheets out. Today’s Agenda • Review. • Review? • Review! Before we do class questions… • Do you have any questions? Worksheet Practice • Specific Heat Worksheet – 7, 9 (only if you haven’t already) • Thermochemistry Round Robin Review – 14, 16 • Thermochemistry Problems Worksheet One – 21-23, 1-7 Review Question 1 • When do you use the equation ΔH = m*Cp*ΔT? – When temperature is changing but not phase. Review Question 2 • When do you use ΔH = mol*ΔHfus/vap? – When phase is changing but not temperature. Review Question 3 • Convert 458 calories to Joules. – 1916.272 Joules • 1 calorie = 4.184 Joules 4.184 Joules • 458 calories * 1 calorie Review Question 4 • If you are heating water from -15⁰C to 79⁰C, how many “steps” would the problem require to solve? –3 • Warming the solid • Melting the solid • Warming the liquid Review Question 5 • How much heat energy is required to raise the temperature of 74.7g of pyrex glass by 43⁰C? The specific heat capacity of pyrex is 0.837 J/g ⁰C. – 2688.53 J • ΔH = m*Cp*ΔT • ΔH = 74.7*0.837*(43) • ΔH = 2688.53 J Review Question 6 • How many torr are in 533 mm Hg? – 533 torr Review Question 7 • How many kPa are in 3.200 atm? – 324.2 kPa Review Question 8 • How many kPa are in 52.1 torr? – 6.94 kPa Review Question 9 • BONUS NON-CHEMISTRY QUESTION • You may wager any/all of your points. • Category: Geography • Montevideo is the capital of which country? – Uruguay. Review Question 10 • Given the following reaction: • 2CO2 + 2H2O C2H4 + 3O2 • ΔH = 1410.7 kJ • How much energy is needed to produce 36g of oxygen gas? – 529.01 kJ • 36 g O2 = 1.125 mol O2 • 1.125 mol * 1410.7 kJ 3 mol O2 Review Question 11 • Would it be good for a student to not study Hess’s Law for the coming test? – No. It wouldn’t. – It would also be impossible for a teacher to put a lengthy-type question as that into a review like this. – So instead, here’s a reminder to study it. Review Question 12 • What is the ΔHf0 of O2? – 0 kJ/mol. – Remember that free/diatomic (basically anything in its natural state) elements have no heat of formation. Review Question 13 • How do you calculate ΔHf0 for a reaction? – Add up the ΔHf0 values for the products and then subtract the ΔHf0 values for the reactants, making sure to adjust for mol totals.