Chem104gw3
advertisement

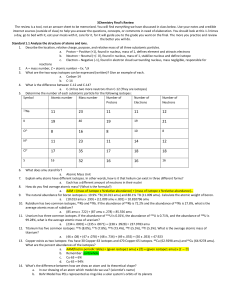
Chem 104 Unit 3 Group Work (September 12th) Name_______________________________ 1. For each of the following isotopes, determine the number of protons, number of neutrons, mass number, atomic number, electron configuration and. Please show the shorthand configuration for the last two. Protons (p) Mass # (p +n, A) Neutrons (n) Atomic # (p, Z ) Electron configuration orbital diagram Carbon-13 Iron-55 119Sn 190Os 2. For each of the elements in Problem 1, determine the valence orbitals and number of valence electrons. Valence orbitals Valence electrons Carbon-13 Iron-55 119 Sn 190 Os 3. How many unpaired electrons are in each of the following elements? Element symbol / Element # / # unpaired electrons Li / Al / P/ Cl / Ti / Cr / Fe / Ni / Zn / Element symbol / Element # / # unpaired electrons Be / Si / S/ Sc / V/ Mn / Co / Cu / 4. Magnesium has three naturally occurring isotopes. The most abundant, 24Mg, has an exact mass of 23.9850 amu and makes 78.99% of a sample of magnesium. Determine the natural abundance of the 25Mg (24.9858 amu) and 26Mg (25.9826 amu) isotopes. Chem104 Unit 3 Group Work [Type text] Page 1 of 1