Friday, March 26, 2010
advertisement

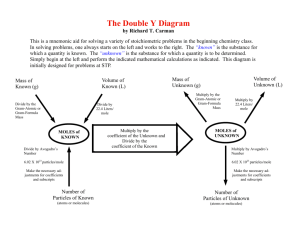
Friday, March 26, 2010 Mrs. Gall Friday, March 26, 2010 Warmup- How many cheerios are in a mole of cheerios? 2. Grade printouts. 3. Turn in today please: 4 page density worksheet, mass of 1. a score lab, mole conversion wkst (p.1 wkst, #1-5) 4. Given today, due Monday: Mole Sample Problems 1,2,3 (3 handouts!) NOTES IN CLASS: work to understand these please. Bring your best effort on Monday. Due date extensions may be given by request. How many particles in a mole? 60,200,000,000,000,000,000,000,000 Also correctly written as: 6.02 x 1023 Section: Avogadro’s Number … 1. 4.00 mol x 6.02 x 1023 = 2.41 x 10 24 2. 1.55 mol x 6.02 x 1023 = 9.33 x 10 23 1 mol 3. 2.65 x 10 25 atoms x 1 mole = 44.0 moles 6.02 x 1023 atoms 70.8 moles 4. 4.26 x 10 25 molecules x 1 mole = 6.02 x 1023 molecules 5. 1.75 x 10 26 atoms x 1 mole = 291 moles 6.02 x 1023 atoms Worksheet due in ten minutes Avogadros number and molar conversions Due at 8:53 am #1-5 Worksheet has page 1 written on the bottom