Moles and the Ideal Gas equation - science
advertisement

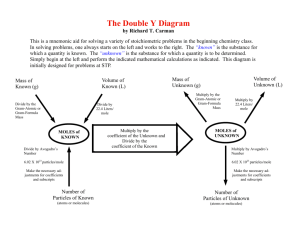
Moles and the Ideal Gas equation Objectives Be able to state that one mole of any substance contains 6.02 × 1023 particles and that 6.02 × 1023 mol-1 is the Avogadro constant NA. Be able to select and solve problems using the ideal gas equation expressed as pV = nRT, where N is the number of atoms and n is the number of moles. OUTCOMES ALL MUST • Be able to state that one mole of any substance contains 6.02 × 1023 particles and that 6.02 × 1023 mol-1 is the Avogadro constant NA. • Be able to state that R is the molar gas constant and be able to select and use it correctly from a data book. MOST SHOULD • Be able to show that R has the Units J mol-1 K-1. • Be able to select and solve problems using the ideal gas equation expressed as pV = nRT, where N is the number of atoms and n is the number of moles. The mole • The mole (symbol: mol) is the SI base unit that measures an amount of substance. One mole contains Avogadro's number (approximately 6.022×1023) things. • A mole is like "a dozen" in that both are absolute numbers (having no units) and can describe any type of elementary object, although the mole's use is usually limited to measurement of subatomic, atomic, and molecular structures. • A mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram (or 12 grams) of carbon-12. • It is useful because we can take any element and 1 mole of it will equal its atomic mass in grams. n= mass of sample / molar mass of substance The Ideal Gas Equation PV = nRT The gas constant • R is called the gas constant and has the value 8.314 Jmol-1K-1 • since R=PV/nT • an analysis of the units for • PV/nT ≡ Nm-2 m3 /mol K ≡ Nm/mol K ≡ J mol-1 K-1 OUTCOMES ALL MUST • Be able to state that one mole of any substance contains 6.02 × 1023 particles and that 6.02 × 1023 mol-1 is the Avogadro constant NA. • Be able to state that R is the molar gas constant and be able to select and use it correctly from a data book. MOST SHOULD • Be able to show that R has the Units J mol-1 K-1. • Be able to select and solve problems using the ideal gas equation expressed as pV = nRT, where N is the number of atoms and n is the number of moles.