Catalyst: Welcome back!
advertisement

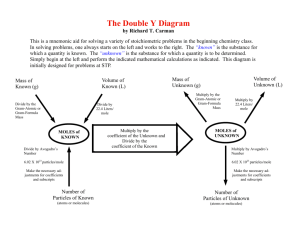
Catalyst Write a paragraph (at least 5 sentences) answering at least 1 of the following questions: How do you use chemicals in your daily life? When you hear the words “chemical reaction” what do you think of? • HW Due Today: Read textbook pp. 230, 236-240 • pp. 233 #6(b, c, d), 7(c), 8(b) • Valence electron review worksheet! • Tonight’s HW: Study for Unit 2 Exam. Lesson Objective I can explain what the different kinds of chemical bonds are and how they are formed. Agenda • • • • Catalyst HW Review Unit 2 Stations Exit Slip HW: pp. 233 #6(b, c, d), 7(c), 8(b) 6 – converting moles to particles – conversion factor is 6.022 x 1023 particles 1.00 mole 6(b) Answer: 2.7 x 1024 molecules BCl3 6(c) Answer: 1.5 x 1023 ions K+ 6(d) Answer: 3.626 x 1024 molecules O2 7(c) Answer: 3.08 x 1024 Na+ ions 8(b) convert atoms to moles – Answer: 0.1661 mol C What is the mole? (Notes) • A mole is the number of atoms in 12 grams of carbon-12. • There are 6.022 x 1023 atoms in 12 grams of carbon-12. • 1 mole (mol) = 6.022 x 1023 particles. 1 mole (mol) = 6.02 x 1023 This number is a constant. The number of particles in 1 mole is called Avogadro’s Number! 6.02 x 1023 Avocado Avogadro Review- The mole - MEMORIZE 1 mole = 6.02 x 1023 particles 6.02 x 1023 is called Avogadro’s number The periodic table is based on the molar mass of carbon (C) Stations! Instructions • There are 8 stations. There are 2 stations per table. • Groups of 3 or 4. • Begin at the station closest to your seat. • The directions are at your station. • Show all your work on a separate sheet of paper. Label your paper with the station number. • I will collect your work at the end of class. Exit Slip 1. Draw the Lewis Dot structure for the molecule: HBr 2. How many molecules of HBr are in 2.00 mole of HBr? 3. Which has more atoms: A mole Platinum or a mole of tin? Random Mole Facts! • Since the Earth has a radius of about 6400 km, its volume is approximately 1021 m3. Since about 500 large grapefruit will fit in one cubic meter, it therefore follows that a mole of grapefruit would have approximately the same volume as the Earth. • If you had exactly one mole of sheets of paper, you could make one million equal stacks from sea level on the earth that would pass the sun. • If you had a mole of pennies, you could give out enough money to everyone in the world so that they could spend a million dollars every hour, day and night, for the rest of their lives.