Chemical Engineering Problem Set: Unit Operations & Equilibrium
advertisement

CHE 425
__________________
LAST NAME, FIRST
Problem set #3
1-8 Copy the program UnitOp3.exe from the CHE 425 class distribution folder to your flash
or H drive. You can also download the program from the website:
https://www.csupomona.edu/~tknguyen/che425/homework.htm.
Run the program and choose problem 1-8 in any order. Solve the problems with the data
provided by the program, copy the problem statement to Word. The program will check your
answer and provide an answer code when you click on “Check”. Copy the answer code and
paste them after the problem statement. You need to present all your work with a diagram in
details to get full credit. Your work should look like this:
CHE425
Problem set #1
NGUYEN, THUAN
1. A vapor at the dew point and 200 kPa containing a mole fraction of 0.40 benzene (1) and
0.60 toluene (2) and 100 kmol total is brought into contact with 110 kmol of a liquid at the
boiling point containing a mole fraction of 0.30 benzene and 0.70 toluene. The two streams
are contacted in a single stage, and the outlet streams leave in equilibrium with each other.
Assume constant molar overflow, calculate the amounts and compositions of the exit
streams.
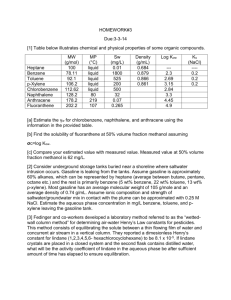
Data: Vapor pressure, Psat, data: ln Psat = A B/(T + C), where Psat is in kPa and T is in K.
Compound
Benzene (1)
Toluene (2)
A
14.1603
14.2515
B
2948.78
3242.38
C
44.5633
47.1806
Problem 1: Correct, Code =4313336481
Solution
x0 = 0.3, L0 = 110 kmol, x and y are mole fraction of benzene in the liquid and vapor phase,
respectively.
L0
V1
Equilibrium stage
L1
V2
y2 = 0.4, V2 = 100 kmol
For CMO, L1 = L0 = 110 kmol, V2 = V1 = 100 kmol. Making a balance on benzene gives
1/1
L0x0 + V2y2 = L1x1 + V1y1
110(0.30) + 100(0.40) = 110x1 + 100y1
11x1 + 10y1 = 7.3 y1 = 0.73 1.1x1
(E-1)
Since the two streams V1 and L1 are in equilibrium, we have
y1 P1sat
=
200y1 = x1exp(14.1603 2948.78/(T 44.5633))
x1 200
(E-2)
1 y1 P2sat
=
200(1 y1) = (1 x1)exp(14.2515 3242.38/(T 47.1806))
1 x1 200
(E-3)
The three equations (E-1,2,3) can be solved for T, x1, and y1 either by graphical or numerical method.
5.2 A liquid mixture containing 25 mol% benzene and 75 mol% ethyl alcohol, in which
components are miscible in all proportions, is heated at a constant pressure of 1 atm from
60oC to 90oC. Using the spline command in Matlab to plot the following T-x-y
experimental data:
y, x = mole fraction of benzene in vapor and liquid phase, respectively
T,oC 78.4 77.5 75
72.5 70
68.5 67.7 68.5 72.5
y
0
0.075 0.28 0.42 0.54 0.60 0.68 0.73 0.82
x
0
0.015 0.05 0.12 0.22 0.31 0.68 0.81 0.91
75
0.88
0.95
Note: the following Matlab codes can plot the data using spline command:
T=[78.4 77.5 75 72.5 70
68.5 67.7 68.5 72.5 75
77.5
y=[0 0.075 0.28 0.42 0.54 0.60 0.68 0.73 0.82 0.88 0.95
x=[0 0.015 0.05 0.12 0.22 0.31 0.68 0.81 0.91 0.95 0.98
ppx=spline(x,T);
ppy=spline(y,T);
xp=0:0.02:1;yp=xp;
Tx=ppval(ppx,xp);
Ty=ppval(ppy,yp);
plot(xp,Tx,yp,Ty)
grid on
Title('Txy data for benzene-ethyl alcohol at 1 atm')
xlabel('x,y: mole fraction benzene')
ylabel('T(^oC)')
77.5
0.95
0.98
80.1
1.0
1.0
80.1];
1.0];
1.0];
Determine: (a) the temperature where vaporization begins; (b) the composition of the first
bubble of vapor; (c) the composition of the residual liquid when 25 mol% has evaporated,
assuming that all vapor formed is retained in the apparatus and is in equilibrium with the
residual liquid. (d) Repeat part (c) for 90 mol% vaporized. (e) Repeat part (d) if, after 25
mol% is vaporized as in part (c), the vapor formed is removed and an additional 35 mol% is
vaporized by the same technique used in part (c).