What is Matter?
advertisement

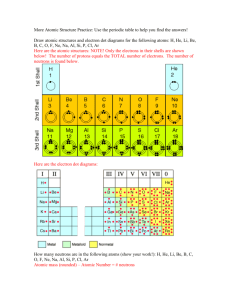
Chemistry Atomic Structure, Bonding, Periodic Table, Acid/Base More free powerpoints at www.worldofteaching.com What is Matter? Matter = any material substance with Mass & Volume What is matter made of? ATOMS – basic building blocks of matter Anything that has mass is made of atoms. Atoms are made up of particles – electrons, protons, and neutrons What does an atom look like? The center of the atom is called the nucleus. It is made up of protons and neutrons. Electrons orbit the nucleus. What are protons, neutrons, and electrons? PROTONS (+) – positively charged particles. They determine the atomic number of the atom and are found in the nucleus. NEUTRONS (0) – particles with no charge (neutral). Found in the nucleus of the atom. ELECTRONS (-) – negatively charged particles. They orbit the nucleus of the atom. They are attracted to the positive charge of the nucleus. Atoms DO NOT have a charge. They are neutral. The number of negative electrons equals the number of positive protons. Atomic Mass Unit A unit of mass used to express the mass of atoms and molecules. 1 atomic mass unit (amu) equals the mass of a proton or a neutron. 1 gram = 600 000 000 000 000 000 000 000 amu Atomic Mass Each element has a unique atomic mass. It is determined by adding the number of protons to the number of neutrons. Complete the following chart: ELEMENT SYMBOL ATOMIC ATOMIC NUMBER NUMBER NUMBER MASS PROTONS NEUTRONS NUMBER ELECTRONS OXYGEN O 8 16 8 8 8 NITROGEN N 7 14 7 7 7 HYDROGEN H 1 1 1 0 1 CALCIUM Ca 20 40 20 20 20 FLUORINE F 9 19 9 10 9 CARBON C 6 12 6 6 6 SODIUM Na 11 23 11 12 11 MAGNESIUM Mg 12 24 12 12 12 ARGON Ar 18 40 18 22 18 SULFUR S 16 32 16 16 16 How are electrons arranged in an atom? Electrons orbiting the nucleus fill in orbitals. Orbitals, or shells, each hold a set number of electrons. Orbital or shell #1 can hold 2 electrons Orbital or shell #2 can hold 8 electrons Orbital or shell #3 can hold 18 electrons Orbital or shell #4 can hold 32 electrons etc. etc. etc. Bohr models can show electron arrangements 1. Tell the number of protons (P) and neutrons (N) in the nucleus 2. Show the electron arrangement using dots on the outer shells Hydrogen Beryllium 1H 1 9Be 4 Helium Boron 4He 2 11B 5 Lithium Carbon 7Li 3 12C 6 Sodium 23Na 11 Aluminum Potassium Argon 27Al 13 Sulfur Calcium 32S 16 Bonding CHEMICAL BONDING is caused by the either the sharing or the donating of electrons between two separate atoms. A BOND is an attraction between two atoms that allows for the formation of molecules. Why do atoms form bonds? Electrostatic attraction – one atom is positively charged and one atom is negatively charged. Atoms prefer to have full shells. (8 electrons make a full outer shell) How do atoms form bonds? By either sharing or donating electrons Electron Dot Diagram This diagram is used to represent how many electrons are available to either share or donate. What are the two ways atoms can bond? Covalent Bond – forms when two atoms SHARE pairs of electrons Ionic Bond – forms when two atoms are electrostatically attracted to each other. Basically, one atoms loses an electron and the other gains an electron. Ion – an atom with charge (positive or negative) Positive Ion – an atom that has lost an electron Negative Ion – an atom that has gained an electron What is the Periodic Table of Elements? •a list of all known elements •organized by increasing atomic number •divided into two main groups – metals and nonmetals How is the Periodic Table organized? VERTICAL COLUMNS are called GROUPS or FAMILIES. Elements in the group share similar properties and behave the same way. HORIZONTAL ROWS are called PERIODS. They determine the number of shells an element has. What are Metals? Metals are good conductors of heat and electricity. Most are shiny, malleable, and ductile – meaning they can be bent or shaped. Metals are found on the left hand side of the Periodic Table. ALKALI METALS are extremely reactive and are never found in nature in their pure form. They are silver colored and shiny. They are soft enough to be cut with a knife. What are Non-Metals? Poor conductors of heat and electricity, most non-metals exist as a gas at room temperature. In solid form, they are usually dull and brittle, unlike metals. They are found on the right side of the Periodic Table, except for Hydrogen. What are Metalloids? These elements are found along the zigzag line on the Periodic Table. Metalloids have properties of both Metals and Nonmetals. Physically, they can look like metals but are brittle like nonmetals. What are Noble Gases? These elements have full outer shells and are not reactive. They are colorless, odorless gases at room temperature. They are found in Group 18, the rightmost column of the Periodic Table. What are Transition Elements? These metals have a wide range of properties. In general, they are shiny and good conductors of heat and electricity. They are found in Groups 3-12 What are Rare Earth Elements? A set of seventeen chemical elements in the Periodic Table, specifically the fifteen Lanthanides plus Scandium and Yttrium. They are located at the bottom of the Periodic Table. They are not actually rare, and can be quite common. They are not found in ore deposits like copper. LANTHANIDE – Transition metals. They are shiny and reactive. ACTINIDE – Also transition metals. They are radioactive and unstable. Elements 95 through 103 do not exist in nature but have been manufactured in a lab. What are Halogens? All nonmetals. Very reactive. Poor conductors of heat and electricity. Tend to form salts with metals. Found in Group 17. Ex: NaCl – sodium chloride, also known as “table salt” Matter comes in 3 phases Solid Gas Liquid Solid Definite Shape Definite Volume Liquid Indefinite Shape – takes the shape of the container Definite Volume Gas Indefinite Shape – takes the shape of the container Indefinite Volume – can expand and be compressed Compounds Compounds - 2 or more elements chemically combined to form a new substance with new properties Properties – The way a chemical substance looks and behaves Compounds Compounds – are made of 2 or more different atoms combined to form Molecules O H+O H2O = H H Chemical formula lists the number of different atoms in a single molecule Structural formula shows the arrangement of the atoms in a single molecule Molecules Glucose Sugar H C C6H12O6 H OH Chemical formula H C C H OH C H OH O H C C OH Structural formula H OH