forces - haganchemistry
advertisement

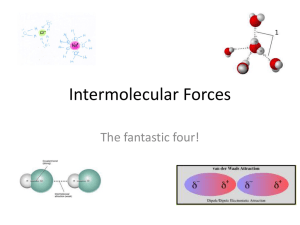
Intermolecular Forces UNIT 3 DAY 5 Do Now/Catalyst (8 min) Entrance Slip: Write your name and period at the top. If you did not do Day 2, Day 3 or Day 4 Questions, do those now Answer the questions under Day 5. You may use your notes. SHOW: Calculation to find the number of bonds (2 pts) Correct Lewis Structure (2 pts) How are you doing? Unit 3 Day 4: Ionic Bonds Class Average Mastery 90 85 80 87 75 70 82 81 82 75 73 65 Period 1 Period 5 Period 7 Period 2 Period 4 Period 6 Period 8 Announcements Edmodo – see class codes on board ACT tutoring Sat 9am-12pm Behavior Grade every two weeks - login Office hours: Tuesday, Wednesday and Thursday New homework policy – NO late work accepted BRING YOUR Due NOTES PACKET Homework ExamEVERYDAY! Unit 3 Friday 11/9 ODD Tuesday 11/13 EVEN Wednesday 11/14 ODD Thursday 11/15 EVEN Objectives: Intermolecular Forces SWBAT define Van der Waals, Dispersion, Dipole-Dipole, and Hydrogen Bonding forces and compare their relative strengths. Real World! Water Striders can walk on water because of hydrogen bonding and strong attractions in the water molecules! Intermolecular Forces Intermolecular Forces are forces between individual atoms or compounds that hold molecules together. They are weaker than the bonds between atoms in a molecule, which are called intramolecular forces. Reminder: dipole results from differences in electronegativity, which produce small charges on atoms in a molecule. Intermolecular Forces Intermolecular forces come in three types, depending on polarity. 1. 2. 3. Dispersion Forces Dipole-Dipole Forces Hydrogen Bonding Dispersion Forces Result from interactions between molecules, temporary charges between non-polar molecules. They are very weak. Results from Strength Attractive force Weakest of between all the forces neighboring molecules, caused by motion of electrons Diagram Dispersion Forces: Diagram Dipole-Dipole Interactions Result from permanent partial charges. These forces are also polar, and thus slightly stronger than dispersion forces. Note: Both Dispersion Forces and Dipole-Dipole Interactions are sometimes called van der waals forces. Results from Strength Negative region of one polar molecule attracted to positive region of other polar molecule Medium strength Diagram Dipole-Dipole Interactions: Diagram Hydrogen Bonding Very similar to intramolecular forces. These are strong and permanent forces (“bonds”) that result from a special type of polar attraction. Form between H and either N, O, F or Cl ONLY. Results from Strength Slightly positive H bonded to one atom is also weakly attracted to another EN atom Strongest of the forces ONLY N, O, F or Cl Diagram Hydrogen Bonding: Diagram We Do: CFU #1 Order these forces from strongest to weakest: Hydrogen Bonding, Dispersion Forces, DipoleDipole Interactions, Covalent Bonds. Covalent bonds, hydrogen bonding, dipoledipole interactions, dispersion forces We Do: CFU #2 True or False: Intermolecular Forces hold the atoms in a molecule together False We Do: CFU #3 True or False: These forces rely on differences in polarity, which produces small charges on the molecules. True We Do: CFU #4 Do you think it would be harder to pull a molecule away from a compound that was held together using Hydrogen Bonds or Dipole-Dipole Interactions? Why? Hydrogen bonds, they have stronger interactions We Do: CFU #5 Do forces get stronger or weaker as differences in electronegativity increase (as polarity increases)? Stronger You Do: Forces Poster (20 min) Fold paper into 3 sections, one for dipole interactions, dispersion forces and hydrogen bonding. On your posters include the following information for each force (use notes and p.250-51 of text): Definition 2 facts One real world example (except for Dipole) Drawing/diagram Chemistry Interim Test Counts as a grade so do your best! Mark all answers on your answer sheet 25 questions Closing Essential Question: What holds the individual molecules in a compound together?