Chapter 1-
advertisement

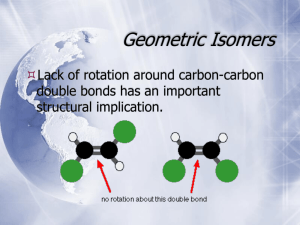
Chapter 5 Stereochemistry: Chiral Molecules 1 Chapter 5 Stereochemistry: Chiral Molecules Isomerism: Constitutional Isomers and Stereoisomers Isomers are different structures with the same molecular formula. Constitutional isomers, already examined, are isomers with different bond connectivities. Examples of Constitutional Isomers formula constitutional isomers C3H8O CH3CH2CH2OH C4H10 CH3CH2CH2CH3 OH CH3CHCH3 CH3CHCH3 CH3 Stereoisomers Stereoisomers are not constitutional isomers. They have the same general atom connectivity, but they differ in the arrangement of atoms in space. The cis-trans isomers of alkenes and cycloalkanes are examples of stereoisomers. Examples of Stereoisomers 2-butene H3C CH3 H cis H H3C H H trans CH3 1,2-dichlorocyclopropane H H H Cl Cl Cl Cl H cis trans Enantiomers and Diastereomers Stereoisomers are divided into two general categories: enantiomers and diastereomers. Enantiomers are stereoisomers that are mirror image related, and are not superimposable upon one another. Diastereomers are stereoisomers that are not enantiomers. They are not mirror images of one another. Isomerism: Constitutional Isomers and Stereoisomers • Stereoisomers are isomers with the same molecular formula and same connectivity of atoms but different arrangement of atoms in space 5 • Enantiomers: stereoisomers whose molecules are nonsuperposable mirror images (R, S) • Diastereomers: stereoisomers whose molecules are not mirror images of each other (R/S, cis/trans) • Example: cis and trans double bond isomers • Example: cis and trans cycloalkane isomers 6 Enantiomers and Chiral Molecules Enantiomers are possible only with molecules that are chiral. If a molecule has two distinct stereoisomeric forms that are mirror image related, it is chiral. These stereoisomeric forms are called enantiomers. Enantiomers and Chiral Molecules • Chiral molecule – Not superposable on its mirror image – Can exist as a pair of enantiomers • Pair of enantiomers – A chiral molecule and its mirror image • Achiral molecule – Superposable on its mirror image 7 • Example: 2-butanol – I and II are mirror images of each other (figures a and b) – I and II are not superposable and so are enantiomers (figure c) – 2-butanol is a chiral molecule 8 Achiral Molecules Achiral molecules do not exist in two unique stereoisomeric forms that are mirror image related (enantiomers). The mirror image of an achiral molecule can be superimposed upon the original structure. • Example: 2-propanol – Not chiral 9 Stereocenters A stereocenter is an atom at which the exchange of any two groups around the atom interconverts stereoisomers. Tetrahedral Carbons as Stereocenters A tetrahedral carbon with four different groups attached can exist in enantiomeric forms. It is a stereocenter because exchange of any two groups around the tetrahedral center interconverts stereoisomers. X W C* Y Z a stereocenter with four different groups The asterisk indicates a stereocenter. • Chiral molecule – A molecule with a single tetrahedral carbon bonded to four different groups will always be chiral – A molecule with more than one tetrahedral carbon bonded to four different groups is not always chiral – Switching two groups at the tetrahedral center leads to the enantiomeric molecule in a molecule with one tetrahedral carbon • Stereogenic center – An atom bearing groups of such nature that an interchange of any two groups will produce a stereoisomer – Carbons at a tetrahedral stereogenic center are designated with an asterisk (*) • Example: 2-butanol 11 Stereocenters in Alkenes The cis and trans isomers of an alkene, such as 1,2-chloroethene, are stereoisomers. Since they are not related as enantiomers, they are diastereomers. Cl Cl Cl a stereocenter trans cis Cl Since exchange of the Cl and H around a carbon interconverts the cis and trans stereoisomers, the carbons are stereocenters. Find the Stereocenter CH3 CH3CCH2CH3 H Cl CH3CCH2CH3 H CH3 CH3CH2CCH2CH2CH3 H achirall chirall chirall 2-methylbutane 2-chlorobutane 3-methylhexane The Importance of Chirality in Biological Systems Chirality or handedness is common in nature. On the macroscopic level, helical seashells generally spiral like a right-handed screw and are chiral. Climbing vines wind in a specific direction. On the molecular level, most biologically important molecules are chiral. The biological activity of chiral molecules is specifically associated with a specific enantiomer. This specificity results from reaction between a chiral molecule and a chiral receptor that only accomodates one enantiomer. This enantioselectivity is a key factor in drug design. The Biological Importance of Chirality • The binding specificity of a chiral receptor site for a chiral molecule is usually only favorable in one way 14 The History of Stereochemistry In 1874 two young chemists, J.H. Van't Hoff (Utrech) and J.A. Le Bel (Sorbonne) deduced a tetrahedral geometry for tetravalent carbon from their observations of substituted methanes. The Van't Hoff and Le Bel Argument At that time, it was known that only one compound of the general formula CH3Y had ever been found. For example, there is only one compound with the formula CH3Br. This observation means that all 4 H in CH4 are equivalent. Possible Geometries for CH4 H H H C H H square planar H C H H H H pyramidal tetrahedral C H H Disubstituted Methanes Also, for the general formula CH2XY, there is only one known compound. For example, only one compound exists with the formula CH2BrCl. This observation rules out the square planar and pyramidal geometries for methane (and tetravalent carbon, in general) because stereoisomers should exist for CH2XY with those geometries. replace H H C Y X H H C H Stereoisomers X H square planar (nonequivalent H) X replace H H C H Y replace H Y C X H H H C X H H pyrimidal (nonequivalent H) Stereoisomers replace H H C X Y H Stereochemical Properties of a Tetrahedral Carbon Atom A tetrahedral geometry for methane is consistent with the stereochemical observations on substituted methanes. H H X X C C C H H CH4 4 equivalent H H H H H X X C C H A Y H Y CH2XY 2 equivalent H CH3X 3 equivalent H CH2XY is achiral. B may be superimposed on A. H Y H B H The same stereostructure. Two Stereoisomeric Forms of CWXYZ Introduction of one more group, Z, leads to stereoisomers. X H C H Y replace H replace H X X Z C H A Y not superimposable H C Z B Y A and B are stereoisomers that are mirror image related. They are enantiomers. This analyis by Van't Hoff and Le Bel that led to their proposal of a tetrahedral geometry for tetravalent carbon was not immediately accepted. In fact, some of the leading organic chemists of that period ridiculed the work of these two young scientists. Kolbe's Lament An excerpt of a letter written by Hermann Kolbe (University of Leipzig) in 1877 in response to the work of the young J.H. van't Hoff. "Not long ago, I expressed the view that the lack of general education and of thorough training in chemistry was one of the causes of the deterioration of chemical research in Germany. Will anyone to whom my worries seem exaggerated please read , if he can, a recent memoir by a Herr van't Hoff on 'The Arrangements of Atoms in Space,' a document crammed to the hilt with outpourings of childish fantasy...... This Dr. J.H. van't Hoff, employed by the veterinary college at Utrech, has, so it seems, no taste for accurate chemicalresearch. He finds it more convenient to mount his Pegasus (evidently taken from the stables of the veterinary college) and to announce how, on his bold flight to Mount Parnassus, he saw atoms arranged in space." Despite such opposition, within 10 years of publishing their work, the ideas of van't Hoff and Le Bel were verified by abundant evidence and accepted by the chemical community. Tests for Chirality: Planes of Symmetry • • The absolute test for molecular chirality is the superimposability test. Determine if a molecule and its mirror image are superimposable on one another. If they are not, the molecule is chiral. A molecule is chiral if it possesses a single tetrahedral stereocenter as in 2-chlorobutane. * CH3CHClCH 2CH3 • If the molecule possesses a plane of symmetry that bisects the structure, it is achiral. The presence of a plane of symmetry means the two halves of the structure are mirror images of each other. OH 4-heptanol CH3CH2CH2 C H CH2CH2CH3 plane of symmetry achiral Tests for Chirality: Planes of Symmetry • Plane of symmetry – An imaginary plane that bisects a molecule in such a way that the two halves of the molecule are mirror images of each other – A molecule with a plane of symmetry cannot be chiral • Example – 2-Chloropropane (a) has a plane of symmetry but 2-chlorobutane (b) does not 21 Nomenclature of Enantiomers: The (R,S) System Since chiral molecules can exist in two different stereoisomeric forms (enantiomers), it is necessary to have a naming system that unambiguously identifies each. In 1956, three prominent chemists (R.S. Cahn, C.K. Ingold and V. Prelog) devised a naming system for stereocenters. A stereocenter is labeled either R (Rectus) or S (Sinister) according to the priority order of the atoms or groups around the stereocenter. (1) Each of the four atoms or groups attached to the stereocenter is assigned a priority from A (highest) to D (lowest). Priority is first determined by the atomic number of the atom attached to the stereocenter. If H is attached, it automatically has the lowest priority (D). Example: 2-butanol (B or C) (A) HO CH3 C (D) H CH2CH3 (B or C) Nomenclature of Enantiomers: The R,S System • Also called the Cahn-Ingold-Prelog system • The four groups attached to the stereogenic carbon are assigned priorities from highest (a) to lowest (d) • Priorities are assigned as follows – Atoms directly attached to the stereogenic center are compared – Atoms with higher atomic number are given higher priority • If priority cannot be assigned based on directly attached atoms, the next layer of atoms (further out) is examined • Example 23 • The molecule is rotated to put the lowest priority group back • If the groups descend in priority (a,b then c) in clockwise direction the enantiomer is R • If the groups descend in priority in counterclockwise direction the enantiomer is S 24 • Groups with double or triple bonds are assigned priorities as if their atoms were duplicated or triplicated 25 Example: An Enantiomer of 3-Chloro-3-methyl-1-pentene (D) CH3 Assign an (R,S) label to this stereoisomer: (B) CH2=CH C (A) Cl CH2CH3 (C) Step 1: Assign Priorities Step 2: Visualize along the axis with the lowest priority group away from the viewer. (B) CH=CH2 (D) CH3 C (A) Cl CH2CH3 This stereoisomer is (S). counterclockwise (C) Step 3: Trace out the sequence A---->C. Quiz Chapter 5 Section 7 Provide complete IUPAC names for the compounds below. (A) Cl (A) OH (D) H C CH2CH3 CH3 (B) (C) (D) H C CH3 H2C=CHCH2 (C) (B) (R)-2-chlorobutane (S)-4-penten-2-ol • Problem: Are A and B identical or enantiomers? • Manipulate B to see if it will become superposable with A • Exchange 2 groups to try to convert B into A – One exchange of groups leads to the enantiomer of B – Two exchanges of groups leads back to B 28 Properties of Enantiomers: Optical Activity Enantiomers are distinct stereoisomers. One structure cannot be superimposed on the other. However, the two stereoisomers are identical in all respects, except that they are mirror images of each other. It is not surprising that enantiomers have identical properties, except when they are in a chiral environment. Physical Properties of (R) and (S)-2-Butanol boiling point (R) 99.5oC (S) 99.5oC density (g/mL, 20oC) 0.808 0.808 Optical Activity However, when plane-polarized light is passed through a solution of (R)2-butanol, the plane is rotated in one direction and when planepolarized light is passed through a solution of (S)-2-butanol, the plane is rotated in the opposite direction. Some Early Studies of Optical Activity In the early 19th century, experiments revealed the extraordinary interaction between light and minerals. In 1808 the French scientist Etienne Louis Malus discovered that light transmitted by a crystal of iceland spar (CaCO3) was polarized in a single plane. (Described below) A few years later, Jean Baptiste Biot (1774-1862, College de France) found that a crystal of quartz (SiO2) rotated the plane of the transmitted plane-polarized light. Biot further observed that some quartz crystals rotate the plane in one direction, and other crystals rotate the plane in the opposite direction. This property of rotation was related to the hemihedral form of the quartz crystals that exists in mirror image forms. Hemihedral Quartz Crystals Optically Active Materials in Nature Biot showed in 1815 that certain naturally occurring organic compounds rotate plane-polarized light in either the pure liquid or dissolved state as a solution. Oil of turpentine, solutions of table sugar, camphor, and tartaric acid all had this property. Biot correctly concluded that this "optical activity" was inherent in the molecules. Experiments with Tartaric Acid Tartaric acid is a byproduct of winemaking. It is present in grapes as its potassium salt. general structures - + COOH CHOH CHOH COOH CO2 K CHOH CHOH COOH tartaric acid potassium tartrate As the sugar in grape juice ferments to ethanol, the potassium tartrate precipitates. Acidification of the salt yields tartaric acid. The Experiments of Louis Pasteur Louis Pasteur (1822-1895) was a student of Biot. In 1848, the young scientist was working with tartaric acid supplied to him by a local chemical company when he noticed something that had escaped earlier workers. The various salts of tartaric acid all showed evidence of crystallizing in a hemihedral crystal of the same sense. Solutions of these salts were optically active. But in experiments with sodium ammonium tartrate (the double salt of tartaric acid), he made a dramatic discovery. This tartrate double salt showed no optical activity in solution, and it crystallized in two hemihedral forms similar to the two hemihedral forms of quartz. Pasteur carefully separated the two hemihedral forms with a tweezers. Solutions of each hemihedral crystal were optically active in the opposite sense. When equal weights of each hemihedral crystal were mixed, the resulting solution was optically inactive. Pasteur correctly speculated that the two forms may differ in arrangement of atoms in space, and envisioned arrangements of opposite senses. Louis Pasteur anticipated the importance of "stereochemistry" as a feature of molecular structures Jean Baptiste Biot studies the crystals prepared by Louis Pasteur. Plane Polarized Light Light is a moving wave of electric and magnetic fields called electromagnetic radiation. The strengths of the electric and magnetic fields oscillate in a repeating pattern as the wave moves through space. Electric and Magnetic Fields (electric and magnetic fields are perpendicular) X Oscillating electric and magnetic fields moving in the X-direction. Head-on view of oscillating electric or magnetic field vector. Ordinary and Plane-Polarized Light Ordinary light is a bundle of waves with electromagnetic field vectors moving in all directions around the propagating axis. Plane-polarized light is a wave with only a single oscillating electromagnetic field vector head-on view head-on view Plane-polarized light is obtained by passing ordinary light through a polarizer (material such as Iceland spar). ordinary light polarizer plane-polarized light The Interaction of Plane-Polarized Light with Materials When plane-polarized light is passed through certain materials (gases, liquids or crystalline solids), the plane of the polarized light rotates. material plane-polarized light rotated plane of polarized light Solutions of certain organic compounds have this capability. The rotatory power is characteristic of the compound and is measured in a polarimeter. Components of a Polarimeter light source polarizer view polarizer for analysis The second polarizer is rotated to match the degree of rotation of the plane-polarized light. When a match is achieved, light passes through the polarizer. The direction of rotation is either: counterclockwise "Levorotatory" "l" or (-) rotated plane of light () sample cell clockwise "Dextrorotatory" "d" or (+) • The Polarimeter 38 Specific Rotation: A Measurement of Rotatory Power The measured rotation, a, depends on how many molecules the plane-polarized light interacts with in passing through the cell, and experimental variables such as the wavelength of the light, solvent and temperature. In order to adjust for these experimental variables, the intrinsic rotatory power of a compound is described by its specific rotation, []: [] where temp l = LxC is the observed rotation L is the cell path length in C decimeters is the concentration in g/mL Some Examples A sample of a compound A in chloroform (0.500 g/mL) at 25.0oC shows a rotation of +2.5o in a 1.0 decimeter cell. What is the specific rotation? [] temp l = LxC = +2.5o 1.0 dm x 0.5 (g/mL) = +5.0o dm-1 (g/mL)-1 What is the observed rotation of A in a 0.5 dm cell? = x L x C = 5.0o dm-1 (g/mL)-1 x 0.5 dm x 0.5 g/mL = +1.25o What is the observed rotation if C = 0.050 g/mL? = x L x C = 5.0o dm-1 (g/mL)-1 x 1.0 dm x 0.050 g/mL = +0.25o • The specific rotation of the two pure enantiomers of 2butanol are equal but opposite • There is no straightforward correlation between the R,S designation of an enantiomer and the direction [(+) or (-)]in which it rotates plane polarized light • Racemic mixture • A 1:1 mixture of enantiomers • No net optical rotation • Often designated as (+) 41 Some Examples of Specific Rotations CH3 CH3 HO C H CH2CH3 H C OH CH2CH3 (S)-2-butanol (R)-2-butanol D25 = -13.52 o 25 = +13.52 o D CH 3 ClCH 2 CH 3 C H CH 2CH 3 H (R)-(-)-1-chloro-2-methylbutane D25 = -1.64 The subscript "D" refers to the sodium D-line at 589.3 nm. o C CH 2Cl CH 2CH 3 (S)-(+)-1-chloro-2-methylbutane 25 D = +1.64 o NOTE: there is no direct correlation between (R,S) and the sign (+ or -) of the rotation. Racemic Forms and Enantiomeric Excess • Often a mixture of enantiomers will be enriched in one enantiomer • One can measure the enantiomeric excess (ee) • Example : The optical rotation of a sample of 2butanol is +6.76o. What is the enantiomeric excess? 43 Optical Purity Racemic Form or Racemate A mixture of equal amounts of the two enantiomers of a chiral compound is called a racemic form (or mixture) or simply racemate. Because the rotatory power is balanced by the equal numbers of the enantiomers, a racemic form shows no net rotation of plane-polarized light. Optical Purity and Enantiomeric Excess (ee) If the specific rotation of a single enantiomer of a chiral compound is known, it is possible to determine the enantiomeric mixture of samples of the compound from polarimetric experiments. A sample of a chiral compound that contains only a single enantiomer is enantiomerically pure, and is said to have an enantiomeric excess (ee) of 100%. Example: (S)-(+)-1-chloro-2-methylbutane A sample that is 100% of this enantiomer has a specific rotation of [a]25 = +1.64o. CH3 H D C CH2Cl CH2CH3 As the levorotatory (R) enantiomer is added to the sample, it cancels the dextrorotatory power of an equal number of (S) molecules. The rotatory power of the sample is due only to the enantiomers that are in excess. Enantiomeric Excess (ee) The enantiomeric excess of a sample is (moles of one enantiomer -moles of other enantiomer) %(ee) = x 100 (total moles of both enantiomers) and is directly calculated from the specific rotations by (observed specific rotation) %(ee) = (specific rotation of pure enantiomer) x 100 Optical Purity of (R)- and (S)-1-Chloro-2-methylbutane observed specific rotation in degrees +1.64 optical purity (ee) in % %S %R 100 100 0 +0.82 50 75 25 +0.41 25 62.5 37.5 50 0 50 0 0 -0.41 25 37.5 62.5 - 0.82 50 25 75 -1.64 100 0 100 Quiz Chapter 5 Section 9 A sample of 100% ee (R)-2-butanol shows [] = -13.5o. What is the enantiomeric mixture of a sample of 2-butanol that shows [] = +1.35o ? %ee = +1.35o +13.5o x 100 = 10% excess of S This sample of 2-butanol is 90% racemic form and 10% excess S, or S = (45 + 10) = 55% and R = 45%. The Synthesis of Chiral Molecules • Most chemical reactions which produce chiral molecules produce them in racemic form 48 Symmetrical Reaction Pathways Racemic forms of chiral products result whenever a reaction goes through an achiral stage: starting point or chemical intermediate. An achiral stage introduces symmetrical reaction pathways that necessarily lead to 50% (S) and 50% (R) chiral products. Example: Reduction of 2-Butanone = O CH3CH2CCH3 + H2 CH3 CH3CH2 C=O O=C H-H H-H CH3 CH2CH3 These and other mirror image modes of reaction occur with equal probability. CH3 CH3CH2 OH C H (R) 50% Racemic Form CH3 HO C CH2CH3 H (S) 50% Enantioselective Reactions A reaction that produces a stereocenter where one enantiomer is favored over the other is enantioselective. This preference requires some external chiral influence such as a chiral reagent, a chiral solvent, or a chiral catalyst. Enantioselectivity in Nature There are many enantioselective reactions in natural systems where the chiral influence is an enzyme. These biopolymers of amino acids provide chiral reaction sites where one enantiomer generally reacts much faster than the other because of stereochemical and electrostatic factors. H = O enzymatic reduction ClCH2CH2CH2CCH3 alcohol dehydrogenase 5-chloro-2-pentanone (achiral) OH C ClCH2CH2CH2 CH3 (S)-5-chloro-2-pentanol (98% ee) Enantioselectivity in the Laboratory Synthetic chemists are designing chiral catalysts that mimic the enantioselectivity of enzyme-catalyzed reactions. O HO H H O O (achiral) (ii) acid workup + CH2=C (i) chiral catalyst OSi(CH3)3 OCH3 (achiral) methyl 5-phenyl pentan-3-ol-oate (98% ee) (S) Chiral Drugs The driving force behind the effort to design enantioselective reactions is the pharmaceutical industry. Typically, only one enantiomer of a chiral drug is active. One example is the antiinflammatory drug ibuprofen (sold as advil, motrin, nuprin). The (S) enantiomer is active while the (R) enantiomer is inactive. H CH3 HO H CH3 HO O (S) (active) O (R) (inactive) Molecules with More than One Stereocenter A compound with one stereocenter can exist in two stereoisomeric forms (enantiomers) called (R) and (S). If there are two stereocenters, each center may be (R) or (S). The various stereocenter combinations are shown in the table. Stereocenters Possible Combinations R 1 R 2 R Stereoisomers S R S S S 2 S 4 R The maximum number of stereoisomers is 2n, where n is the number of stereocenters. The Stereoisomers of 2,3--Dibromopentane * * two stereocenters 2n = 4 stereoisomers CH3CHCHCH2CH3 Br Br What are the four stereoisomers and how are they related to each other? Possible Combinations of the Stereocenters 1 2 3 4 5 CH3-CHBr-CHBr-CH2CH3 (2R) (2R) (2S) (2S) (3R) (3S) (3R) (3S) The Four Stereoisomers of 2,3--Dibromopentane CH3 CH3 H H C Br C Br CH2CH3 I (2S,3R) Br C H C Br H CH2CH3 II (2R,3S) CH3 CH3 Br H C H C Br CH2CH3 III (2R,3R) H Br C Br C H CH2CH3 IV (2S,3S) (1) Use eclipsed conformations for easier assignment of (R) or (S). (2) Add the groups around the stereocenters in one stereoisomer and assign labels to the stereocenters. (3) Draw the mirror image of I. That gives II. Each stereocenter changes configuration. (4) Change the configuration only at C-2 in I from S to R. That gives III. (5) Draw the mirror image of III. That gives IV. The Relationships Among the Four Stereoisomers of 2,3-Dibromopentane CH3 H C H C Br CH3 Br C H C H Br Br CH2CH3 CH2CH3 2S, 3R 2R, 3S I II CH3 CH3 C H H C Br H C Br Br C H CH2CH3 CH2CH3 2R, 3R 2S, 3S III IV Br Stereoisomer pairs I/II and III/IV are mirror image related. They are enantiomers. Note that all stereocenters change in going from one enantiomer to the other. CH3 H C H C Br Br Br CH2CH3 2S, 3R I CH3 CH3 C H H C Br CH2CH3 H C Br Br C H CH2CH3 CH3 Br C H C H CH2CH3 2R, 3S II enantiomers Br 2R, 3R III 2S, 3S IV enantiomers Stereoisomers that are not Enantiomers are Diastereomers of Each Other Among the following four stereoisomers CH3 H C H C Br CH3 Br C H C H Br Br CH2CH3 CH2CH3 2S, 3R 2R, 3S I II the diastereomeric pairs are: CH3 CH3 C H H C Br H C Br Br C H CH2CH3 CH2CH3 2R, 3R 2S, 3S III IV Br I/III, I/IV, II/III, II/IV Note: In diastereomers, all the stereocenters are not mirror image related as in enantiomers. Unlike the mirror-image related enantiomers, diastereomers have different physical and chemical properties. The diastereomeric relationship between structures is an important and fundamental principle in simple organic and biological systems. Meso Compounds: A Special Stereochemical Situation The maximum number of stereoisomers when there are two stereocenters is 22= 4. But structural symmetry influences this analysis. Example: 2,3-dibromobutane * There are two stereocenters in this compound. * CH3-CH-CH-CH3 Br Br The four possible stereoisomers of 2,3-dibromobutane are: CH 3 H C Br H C Br CH 3 2S, 3R I CH 3 CH 3 Br C H Br C H Br C H H C Br CH 3 2R, 3S II CH 3 2R, 3R III CH 3 H C Br Br C H CH 3 2S, 3S IV At first view, it may appear that this compound exists in four unique stereoisomeric forms as found in 2,3-dibromopentane, but.............. Structural Symmetry: The Meso Diastereomer Symmetry reduces the number of stereoisomers from four to three. CH3 H C H C CH3 Br Br Br Br CH3 2S, 3R I C H C H CH3 2R, 3S II CH3 C H H C Br CH3 2R, 3R III Br CH3 H C Br Br C H CH3 2S, 3S IV A more careful inspection of the four possible stereoisomers reveals that I and II are not different because one structure may be superimposed on the other. They are the same stereoisomer. Thus, 2,3-dibromobutane exists in only three unique stereoisomeric forms: I=II, and the enantiomeric pair III and IV. The Diastereomers of 2,3-Dibromobutane Stereoisomer I=II is achiral and is called a meso diastereomer. The meso diastereomer has an internal plane of symmetry that necessarily makes it achiral. Br H C CH3 R Br C H S CH3 The meso diastereomer of 2,3-dibromobutane is achiral and does not exist in enantiomeric forms. The Chiral Diastereomer of 2,3-Dibromobutane CH3 Stereoisomers III and IV are the two enantiomers of the chrial diastereomer of 2,3-dibromobutane. C H H C Br CH3 2R, 3R III Br CH3 H C Br Br C H CH3 2S, 3S IV . This diastereomer is sometimes called the ( +- ) or (d,l) diastereomer, meaning it exists in enantiomeric forms, to distinguish it from the meso-diastereomer. Meso Compounds • Sometimes molecules with 2 or more stereogenic centers will have less than the maximum amount of stereoisomers 62 • Meso compound: achiral despite the presence of stereogenic centers • Not optically active • Superposable on its mirror image • Has a plane of symmetry 63 Quiz Chapter 5 Section 12 How many stereoisomers are possible for each structure below? Identify each stereoisomer with its complete name and identify enantiomeric and diastereomeric relationships. CH3CHClCH2CHClCH3 2,4-dichloropentane (S,S)-2,4-dichloropentane enantiomers (R,R)-2,4-dichloropentane diastereomers (S,R)-2,4-dichloropentane CH3CHClCHClCH2CH3 2,3-dichloropentane (2S,3S)-2,3-dichloropentane (2R,3R)-2,3-dichloropentane enantiomers diastereomers (2S,3R)-2,3-dichloropentane (2R,3S)-2,3-dichloropentane enantiomers Naming Compounds with More than One Stereogenic Center • The molecule is manipulated to allow assignment of each stereogenic center separately – This compound is (2R, 3R)-2,3-dibromobutane 65 Fischer Projection Formulas Emil Fischer (1852-1919), one of the most prominent German chemists of the late 19th and early 20th centuries, devised a convention for representing three-dimensional structural information in two dimensions. These "Fischer formulas" are especially helpful in keeping track of stereochemical details in structures with more than one stereocenter. Example: (R)-2-chlorobutane CH2CH3 CH2CH3 H C H Cl Cl CH3 CH3 The Two-Dimensional Fischer Formula Draw vertical and horizontal crossing lines. The point of intersection is the stereocenter. The horizontal line represents bonds projected towards the viewer. The vertical line represents bonds projected away from the viewer. How to Use Fischer Formulas: (S,R)-2,3-Dibromobutane * * CH3-CH-CH-CH3 Br Br By convention, Fischer formulas are written with the main chain extending top to bottom. (S,R)-2,3-Dibromobutane (meso) CH3 CH3 H C H C Br Br CH3 3-dimensional representation H Br H Br CH3 Fischer Formula Note: A plane of symmetry identifies this stereoisomer as achiral in both representations . The superimposability test may be used with Fischer formulas to determine whether structures are identical or different. Rules for the Superimposability Test with Fischer Formulas (1) Fischer formulas only have meaning for structures with stereocenters. o (2) Fischer formulas may be rotated 180 in the plane of the paper, but no other angle. (3) Any one exchange of groups around a stereocenter produces the other configuration (R--->S or S--->R). Any even number of exchanges produces the original configuration. (4) If any of the above manipulations allows one Fischer formula to be superimposed on a second, they are the same stereoisomer. Example: Compare A and B as Fischer formulas CH3 H C Br C H Br CH3 A CH3 CH3 Br H H Br H Br Br H CH3 CH3 I II I and II are not superimposable as drawn. Rotate II by 180o and check again. CH3 H C Br C H Br CH3 B rotate in plane 180o CH3 I and III are also not H Br superimposable, so I and II/III are different stereoisomers. Br H Since they are mirror image CH3 related, they are enantiomers. III Fischer Projection Formulas • Vertical lines represent bonds that project behind the plane of the paper • Horizontal lines represent bonds that project out of the plane of the paper 70 Quiz Chapter 5 Section 13 Complete the Fischer projection formula for (R)-2-butanol. CH3 HO H CH2CH3 solution H CH3 C OH CH2CH3 (R) H CH3 CH3 OH CH2CH3 HO H CH2CH3 two exchanges of groups around stereocenter retains configuration Stereoisomerism in Cyclic Compounds The stereochemical features of cyclic systems can be examined using the (sometimes hypothetical) planar structures. 1,2-Dimethylcyclopropane CH3 CH3 II I CH3 III CH3 trans Stereoisomers I and II are the enantiomers of the chiral trans diastereomer. H3C CH3 cis Stereoisomer III is the meso diastereomer (achiral) with a plane of symmetry. Stereoisomerism of Cyclic Compounds • 1,4-dimethylcyclohexane • Neither the cis not trans isomers is optically active • Each has a plane of symmetry 73 1,3-Dimethylcyclopentane CH3 CH3 CH3 II CH3 I CH3 trans Stereoisomers I and II are the two enantiomers of the chiral trans diastereomer. CH3 III cis Stereoisomer III is the meso cis diastereomer. Even though cyclopentane has a nonplanar conformation, the analyses of the planar geometries lead to correct stereochemical conclusions. Stereochemical Features of Dimethylcyclohexanes An examination of hypothetical planar structures does provide quick and correct conclusions about the stereochemical features of the dimethylcyclohexanes. A thorough evaluation requires an examination of the chair conformations. 1,3-Dimethylcyclohexane I I III cis (meso) trans (chiral) II II III This quick analysis indicates that the trans diastereomer is chiral because it exists in two non-superimposable mirror image stereoisomeric forms (I and II). The cis diastereomer is achiral and is meso because it has an internal plane of symmetry. • 1,3-dimethylcyclohexane • The trans and cis compounds each have two stereogenic centers • The cis compound has a plane of symmetry and is meso • The trans compound exists as a pair of enantiomers 76 A Conformational Analysis of the 1,3-Dimethylcyclohexanes Draw a chair conformation of the trans diastereomer (axial-equatorial) and its mirror image (enantiomer). These are stereoisomers I and II below. Chair-chair interconversion leads to I' and II' that are also mirror image related (enantiomers). I' I mirror II mirror II' After rotations, I superimposes on I' and II superimposes on II'. Therefore, chair-chair interconversion gives back the same stereoisomer. I/I' and II/II' are not superimposable. They are mirror image isomers. They are the two enantiomers of the chiral trans diastereomer. cis-1,3-Dimethylcyclohexane: The Meso Diastereomer Analysis of the (hypothetical) planar structure quickly reveals a plane of symmetry in the cis diastereomer, which means this compound is achiral. This conclusion is confirmed from an examination of the two chair conformations. III chair 1 diaxial IV chair 2 diequatorial (highly favored) Although III and IV are different stereoisomers (more specifically conformational stereoisomers), they are both meso, since each has a plane of symmetry. Therefore, cis-1,3-dimethylcyclohexane exists in two interconverting chair conformations that preserve the symmetry features revealed in the planar structure. Relative Configurations at Stereocenters For many years before the development of x-ray crystallography, configurations at stereocenters were assigned relatively. The stereocenter in one chiral compound was related to one in another compound through a stereochemically well-defined chemical transformation. CH3 H C* bond-breaking is remote from stereocenter CH3 + CH2OH C2H5 HCl (S)-(-)-2-methyl-1-butanol o 25 []D = -5.756 heat H C* CH2Cl + H2O C2H5 (S)-(+)-1-chloro-2-methylbutane o 25 []D = +1.64 Because the bonding changes are remote from the stereocenter, the configuration of groups around the stereocenter in the product is the same as in the reactant. This reaction proceeds with retention of configuration. Relative Configurations: (D)- and (L)-Glyceraldehyde In the late 19th century, Emil Fischer developed a method for assigning configurations at stereocenters relative to the enantiomers of glyceraldehyde. For the next 50 or 60 years, configurations at stereocenters were labeled relative to the stereocenters in the stereoisomers of glyceraldehyde. The Stereoisomers of Glyceraldehyde O O CH CH O HO H OH glyceraldehyde H C OH CH2OH (R) (+) (D) HO C H CH2OH (S) (-) (L) Over 100 years ago, Fischer assigned the dextrorotatory (+) stereoisomer, the configuration we call (R), and the levorotatory (-) stereoisomer was assigned the (S) configuration. The labels Fischer assigned were called (D) and (L). These assignments were a guess. Relating Configurations through Reactions in which No Bonds to the Stereogenic Carbon are Broken • A reaction which takes place in a way that no bonds to the stereogenic carbon are broken is said to proceed with retention of configuration 81 An Example: Relating (-)-Lactic Acid to (+)-Glyceraldehyde O CH H C* OH HgO oxidation HNO2 COH H C* OH Retention CH2OH (+)-glyceraldehyde O O H2O Retention CH2OH (-)-glyceric acid COH H C* OH CH2NH2 (+)-isoserine This transformation shows that (+)-isoserine has the same absolute configuration as (+)-glyceraldehyde. HNO2 HBr Retention O O COH H C* OH CH3 (-)-lactic acid Zn, H+ Retention COH H C* OH CH2Br (-)-3-bromo-2-hydroxypropanoic acid This transformation shows that (+)-isoserine has the same absolute configuration as (-)-lactic acid. Absolute Configurational Assignments The series of chemical reactions involving retention of configuration at the stereocenters configurationally link (+)-glyceraldehyde and (-)-lactic acid. O CH H C * OH O configurationally the same CH 2OH (+)-gly ceraldehy de COH H C * OH CH 3 (-)-lact ic acid Before 1951 the absolute configurations were not known. Only these relative configurations were known from carefully designed chemical transformations linking the assignments to the configurations of the glyceraldehydes assumed by Emil Fischer. Absolute Configurations In 1951 J.M. Bijvoet demonstrated the absolute configuration of (+)-tartaric acid by X-ray analysis. Earlier work showed that this compound was configurationally linked to (-)-glyceraldehyde. The assumed assignments of Emil Fischer were shown to be correct. COOH O CH configurationally linked * HO C H CH2OH (S)-(-)-glyceraldehyde The work of Bijvoet allows the absolute assignments of configurations in all chiral compounds that had been chemically linked to the glyceraldehydes. In more recent years, X-ray analysis has been widely used to assign configurations in other compounds with stereocenters. H C* OH * HO C H CH2OH (+)-tartaric acid structure confirmed by X-ray Absolute Configurations O O CH CH * HO C H CH2OH (S)-(-)-glyceraldehyde "L" * H C OH CH2OH (S)-(-)-glyceraldehyde "D" The Separation of Enantiomers: Resolution Because enantiomers have identical physical properties, they are not separable by simple direct methods such as distillation, chromatography or crystallization. They may be separated in the presence of a chiral influence that introduces diastereomeric relationships. The separation of the enantiomers of a racemic form is called resolution. Resolution Scheme - R S (identical properties) R S R R R separate + reaction Racemic Form R R R R R Diastereomers (different properties) S pure forms R - R S is a resolving agent. It is a single enantiomer (such as R) of a chiral compound. The racemic form (R,S) is reacted with a single enantiomer (R) of a resolving agent to produce diastereomers (R,R and S,R) that are separable by physical means. The resolving agent is then removed producing the pure enantiomers R and S. Resolving Agents Potential resolving agents are optically active acids and bases. Nature provides a group of optically active amines (bases) called alkaloids in plants. Many form crystalline salts when reacted with chiral organic acids. The two diastereomeric salts produced from a racemic form of the organic acid may be separated. Removal of the resolving agent yields the pure enantiomers of the organic acid. Examples of optically active alkaloids are (-)-quinine, (-)-strychnine and (-)-brucine. CH3O HO H H N H N H CH3O H O= H quinine (primary alkaloid from various species of Cinchona) H H N N H N O strychnine (abundant in seeds of Strychnos nux-vomica L.) CH3O H N H O= brucine H O (from Strychnos seeds) Resolution of a Carboxylic Acid CH3 C6H5*CCOOH H (+)(-)-Salt + (-)-alkaloid (-)(-)-Salt (basic) diastereomers (+,-)-2-phenylpropanoic acid (racemic form) (+)(-)-Salt H3O+ separate by fractional crystallization (-)(-)-Salt H3O+ water phase organic phase water phase organic phase CH3 CH3 *CCOOH (-)-alkaloid as (-)-alkaloid as C H (-)* 6 5 (+)- C6H5CCOOH ammonium salt ammonium salt H H Stereocenters other than Carbon Any tetrahedral atom with four different groups is a stereocenter, similar to carbon, with the potential to exist in two stereoisomeric forms. R1 R1 R4 R2 R4 + R2 Si N R3 R 3 quaternary ammonium ion silane Chiral Molecules without a Stereocenter: Molecular Chirality A molecule is chiral if it is not superimposable on its mirror image. It is not required that there be a stereocenter in the structure. A chiral structure without a stereocenter has molecular chirality. Examples of Molecular Chirality H H CH3 C C C CH3 an allene COOH NO2 NO2 COOH a biphenyl Chiral Molecules that Do Not Possess a Tetrahedral Atom with Four Different Groups • Atropoisomer: conformational isomers that are stable • Allenes: contain two consecutive double bonds 90