Honors Quiz Study Guide - Kawameeh Middle School
advertisement

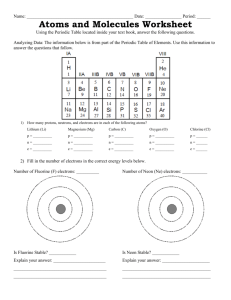
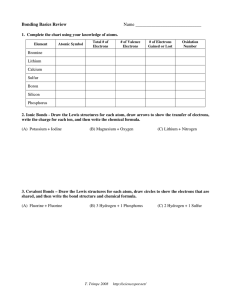
Name __________________________________________ Period _______ Date _____________________ Honors: Chemical Bonds Study Guide Chapters 7-9 1. Below is a Bohr’s Model of Lithium Fluoride. When Lithium and Fluorine bind Lithium loses an electron and Fluorine gains one. a) b) c) d) What charge is Lithium? _____________ What charge is Fluorine? ___________ Which electrons were involved in this bond?_______________ Why did Fluorine gain one electron? ____________________ 2. Two or more substances that are physically blended but are not chemically bonded are called ______________. 3. List the correct method of separating the following mixtures. a. Iron filings and sulfur _______________________________________ b. Salt and water _____________________________________________ c. Sand and water ____________________________________________ 4. Which one of the choices above is a solution (homogenous mixture)? ________________________ 5. Identify whether the following characteristics are related to ionic or covalent bonds. a) b) c) d) e) Takes place between 2 nonmetals ________________________________ Electrons are transferred _______________________________________ Electrons are charged (+/-) _____________________________________ Electrons are shared ___________________________________________ Takes place between metal and nonmetal __________________________ 6. Does the following formula follow the law of conservation of mass? Why or why not? Show your work here! _______________________________________________________________________ 7. Create an ionic bond for the following: a) Boron + 3 Chlorine Calcium + 2 Fluorine 8. Create a covalent bond for the following a) Carbon + 4 Hydrogen Sulfur + Sulfur 9. Identify the following as either physical or chemical changes a. gasoline burning ____________________________ b. whipping egg whites ____________________________ c. dissolving sugar in coffee ____________________________ d. milk souring (sour-tasting lactic acid is produced) __________________ e. Iron rusting ____________________________ f. Formation of bubbles ___________________________ 10. Label the pH scale below with the following labels: acids, bases, and neutral. 11. A) Define Endothermic Change __________________________________________________________ B) What happens to the temperature in an endothermic reaction? _________________ 12. A) Define Exothermic Change ___________________________________________________________ B) What happens to the temperature in an exothermic reaction? ___________________ 13. The last known Tasmanian tiger died at Hobart Zoo on Sept. 7, 1936. Since then Tasmanian tigers have been _____________________________. 14. What are some factors which may cause animals to become extinct? _______________________________________________________________________ 15. Webbed feet are a(n) __________________ for ducks because it allows them to swim. 16. Adaptations allow animals to be able to ______________________________________. Balance the following equations. 17. K + Cl2 18. NaF + Br2 KCl NaBr + F2