Aldehydes & Ketones
advertisement

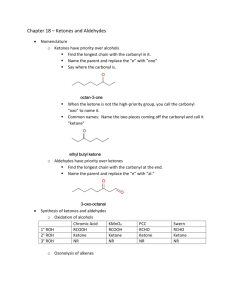
O Fragrant odors Basic building block of housing materials Hormones Digestion Vision 2 Carbonyl group › C=O › Aldehydes RCH=O Formyl › Ketones RC=OR’ 3 Aldehydes › IUPAC end in “al” › Common end in “aldeyde” › Carbonyl C is always #1 › Cyclic cpds Carbaldehyde is ending for most 4 Aldehydes H2C methanal (formaldehyde) O H3C ethanal (acetaldehyde) O H3C propanal (propionaldehyde) O H3C O butanal (n-butyraldehyde) 5 Aldehydes H3C 3-methylbutanal H3C O H2C 3-butenal O HO OH 2,3-dihydropropanal (glyceraldehyde) O 6 Aldehydes cyclopentanecarbaldehyde (formylcyclopentane) O O O OH benzenecarbaldehyde (benzaldehyde) 2-hydroxybenzenecarbaldehyde (salicylaldehyde) 7 Ketones › IUPAC end in “one” › Common end in “ketone” › Carbonyl C is never #1, but always gets low number preference › Cyclic cpds Carbaldehyde is ending for most 8 Ketones H3C CH3 O propanone (acetone) H3C 2-butanone (ethyl methyl ketone) O CH3 H3C O CH3 3-pentanone (diethyl ketone) 9 Ketones cyclohexanone O O 2-methylcyclopentanone CH3 H2C CH3 O 3- buten-2-one (methyl vinyl ketone) 10 Ketones O acetaphenone (methyl phenyl ketone) CH3 O benzophenone (diphenyl ketone) O dicyclopropyl ketone 11 Formaldehyde › Simplest aldehyde › Manufactured on large scale (8 billion lbs per annum) from catalyzed oxidation of methanol CH3OH CH2=O + H2 › Gas at RT (bp = -21˚C) but cannot be stored in free state due to polymerization › Normally 37% soln called formalin (preservative) › Most used in making of plastics, insulation, particle board, and plywood 12 Acetaldehyde › Boils close to RT (bp = 20˚C) › Made by catalyzed oxidation of ethylene 2 CH2=CH2 + O2 2 CH3CH=O › ~1/2 is oxidized to acetic acid › Remainder used for production of 1- butanol and others. 13 Acetone › Simplest ketone › Large scale production like formaldehyde › Produced from oxidation of propene, isopropyl alcohol, or isopropylbenzene H3C H3C CH3 OOH OH H3C O2 dilute H2SO4 O + H3C H2O › ~30% used directly, great solvent, H2O miscible › Rest used to make stuff like epoxy resins 14 CH3 Quinones › Cyclic conjugate diketones › Simplest is 1,4-benzoquinone › All are colored and are thus used often as dyes › Alizarin…used to dye the red coats of the British Army during American Revolution › Vitamin K is required for normal clotting of blood 15 O OH OH O O 1,4-benzoquinone O alizarin OH CH3 CH3 OH H3C H3C H3C H3C Vitamin K vitamin K 16 Oxidation › 1˚ ROH gives aldehyde › 2˚ ROH gives ketone › Cr reagents (PCC) are common oxidizing H3C OH agent H2C O 17 Friedel-Crafts Acylation › Recall the rxn? › Makes aromatic ketones Cl + AlCl3 + O benzyl chloride O benzophenone 18 HCl Hydration of terminal alkynes › Gives methyl ketones › Catalyzed by acid and mercuric ion H3C CH H+, H2O ++ Hg CH3 H3C O 19 Many have pleasant odors Used in the perfume industry Extremely expensive to gather from natural producers Chanel No. 5 (my mom’s fave perfume) was first perfume to use synthetic organic chemicals in 1921 20 O CH3 O HO O O benzaldehyde cinnamaldehyde vanillin 21 C atom is sp2 hybridized Bond angles? C=O bond length is 1.24Å (compared to 1.43Å for C-O in ROH and ROR O is more EN than C › Makes a polar bond 22 Most carbonyl reactions are nucleophilic attacks on the carbonyl C C=C usually is attacked by an electrophile Due to polarization, physical properties differ from HC’s and ROH’s › bp’s are higher than HC’s, lower than ROH’s 23 C=O is permanently polarized › Positive part of one molecule is attracted to negative part of another molecule › Dipole-dipole forces, weaker than H-bonds, stronger than LDF 24 C=O’s with low MW are soluble in water › Can form H-bonds with water or ammonia 25 Why does the attack occur? If rxn occurs in hyroxylic solvent (water or ROH), a proton is usually added to the O 26 Carbonyl cpds are weak Lewis bases due to lone pairs on O Acids can catalyze the addition of weak nucleophiles to carbonyl cpds through protonation 27 Nucleophiles add reversibly › Good leaving groups, CB of SA Nucleophiles add irreversibly › Poor LG, CB of WA In general, ketones are less reactive than aldehydes › Steric…sp2 v. sp3, R v. H › Electronic…alkyl groups are electrondonating…ketones have two 28 Alcohols are oxygen nucleophiles › OR goes to C, and H goes to O Because ROH’s are weak nucleophiles, acid catalyst must be used Product is a hemiacetal › Contains both alcohol and ether on same C Addition is reversible 29 Mechanism of hemiacetal formation has 3 steps › Carbonyl O is protonated by acid catalyst › ROH’s O then attacks carbonyl C › Proton is then lost from resulting +O Each step is reversible 30 Write an equation for the formation of a hemiacetal from acetaldehyde, ethanol, and an acid catalyst. Show each step in the rxn mechanism. 31 Excess ROH means hemiacetals react further to produce acetals Hydroxyl group of hemiacetal is replaced by an alkoxyl group. Acetals have two ether groups on same C 32 Mechanism of acetal formation 33 Mechanism of acetal formation 34 Aldehydes that have appropriately located hydroxyl group can exist in equilibrium with a cyclic hemiacetal…5-hydroxypetanal 35 Aldehydes that have appropriately located hydroxyl group can exist in equilibrium with a cyclic hemiacetal…5-hydroxypetanal 36 Cpds with hydroxyl group 4 or 5 C’s from the aldehyde group tend to form cyclic hemiacetals and acetals due to lack of strain Carbohydrates 37 Ketones also form acetals If a glycol is used, product is cyclic 38 Summary › Aldehyde or ketone reacts with ROH › Hemiacetal is formed › Further ROH makes acetal 39 Water is an oxygen nucleophile, like ROH’s Can add reversibly 40 Aside from formaldehyde hydrate most other hydrates cannot by isolated because they lost water…Keq<1 One exception is trichloroacetaldehyde (chloral) › Forms a stable crystalline hydrate, chloral hydrate, CCl3CH(OH)2 › Used as a sedative 41 Grignard reagents act as carbon nucleophiles toward carbonyl cpds › Grignard reagent adds irreversibly to the carbonyl carbon, forming a new C-C bond › Favorable because product (an alkoxide) is a much weaker base than the starting carbanion › The alkoxide can be protonated to give an ROH 42 Useful route to alcohols › Type of carbonyl determines class of ROH › Formaldehyde gives 1˚ ROH’s 43 Other aldehydes give 2˚ ROH’s 44 Ketones give 3˚ ROH’s 45 Other organometallic cpds like organolithium cpds and aceylides react with carbonyl cpds similarly to Grignard reagents 46 HCN adds reversibly to carbonyl group of aldehydes and ketones to make cyanohydrins › Hydroxyl and cyano group attached to same C › Basic catalyst is needed 47 Acetone reacts as follows: 48 Cyanohydrins play important role in the defense system of the millipede › Two-chambered gland like the bombadier beetle › Benzaldehyde cyanohydrin is stored and then converted to a mixture of benzadehyde and hydrogen cyanide and secreted 49 Write an equation for the addition of HCN to benzaldehyde. 50 Ammonia, amines, and other related cpds have a lone pair on the N and thus act as a nucleophile toward a carbonyl C 51 Aldehydes and Ketones are easily reduced to 1˚ and 2˚ alcohols, respectively Metal hydrides used to reduce › Irreversible nucleophilic attack › LiAlH4 or NaBH4 52 The original product is an aluminum alkoxide Then hydrolyzed by water and acid to give ROH Net result is addition of H across the C=O 1. LiAlH4 O H OH 2. H+, H2O 53 Aldehydes are more easily oxidized than are ketones Oxidation of an aldehyde gives an acid with the same number of C’s Oxidizing agents include KMnO4, CrO3, Ag2O CrO3, H+ O H3C O H3C Jones reagent OH OH O Ag2O O 54 Tollens Silver Test › Silver-ammonia complex ion is reduced by aldehydes but not by ketones › If test tube is clean, a mirror forms by the metallic Ag › Used to “silver” glass using formaldehyde (cheap) 55 Aldehydes and Ketones may exist as an equilibrium mixture of two forms › Keto and enol forms › Differ in the location of a proton and a double bond Tautomerism (Greek…same part) › Structural isomers › Not resonance contributors 56 In order for an enol form to exist carbonyl C must have an H attached to the carbon adjacent to the carbonyl group › Known as the -hydrogen and is attached to the -carbon 57 Most simple aldehydes and ketones exist primarily in the keto form › Keto more stable › Acetone, 99.9997% keto form Phenols have mainly enol form OH O H H 58 Carbonyl cpds that do not have an hydrogen cannot form enols and exist only in keto form O O O H H H 59 -Hydrogen is more acidic than normal H attached to a C › Carbonyl C carries a partial + charge, attracting bonding electrons away from the -H…make it easy for a base to remove the -H › Resulting anion is stabilized by resonance…enolate anion 60 Enolate anions may act as carbon nucleophiles Enolate can add reversibly to the carbonyl group of another aldehyde or ketone…known as aldol condensation Simplest is the combination of two acetaldhyde molecules due to treatment with an aqueous base 61 62 Step 1…base removes -H to form enolate anion Step 2…enolate anion adds to the carbonyl carbon of another acetaldehyde molecule, making a new C-C bond Step 3…alkoxide ion form in step 2 accepts a proton from the solvent, thus regenerating the OH- needed for the first step 63 3-hydroxyaldehydes are always formed Since -C acts as a nucleophile, the product always has just one C between the aldehyde and alcohol C’s Does not matter how long the C chain is in the starting aldehyde 64 Enolate anion of one carbonyl cpd can be made to add to the carbonyl carbon of another cpd Consider acetaldehyde and benzaldehyde (has no -H) when treated with base 65