Osmosis Diffusion Lab
advertisement

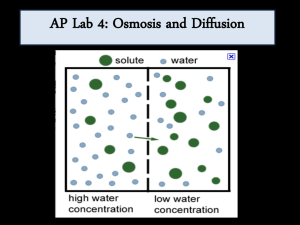
Diffusion/ Osmosis/ Water Potential Lab Notes From the teacher Day 1 Before class: Review the lab – read the background section to the end of PART A (Surface Area and Cell Size). Complete the pre-lab for PART A: o Title and date of the lab for the title, just say Osmosis/ Diffusion Lab: Part A Surface Area and Cell Size (remember to add this lab to your table of contents). o Purpose 1-2 sentences describing the overall goal of PART A only; use complete sentences o Pre-Lab Questions you need to number the question, rewrite the question, and then answer it for full credit (see the lab for a list of Pre Lab questions for Part A) o Lab Procedure Write a procedure for PART A. First list the materials that you will be using, then use your own words to describe the steps in experiment in paragraph form. o Data Table Tape this into your lab notebook (or recopy it) In class: Complete PART A along with all of the data collection, make the graph, and complete the analysis questions. If you do not have time to finish the analysis questions, finish this for homework. Day 2 Before class: Review the lab – read the background section to the end of PART B (Modeling Diffusion and Osmosis). Complete the pre-lab for PART B: o Title and date of the lab just say for the title - Osmosis/Diffusion Lab: Part B Modeling Diffusion and Osmosis o Purpose 1-2 sentences describing the overall goal of PART B; use complete sentences o Pre-Lab Questions you need to number the question, rewrite the question, and then answer it for full credit o Lab Procedure Write a procedure for PART B. First list the materials that you will be using, then use your own words to describe the steps in experiment in paragraph form. o Data Table Tape this into your lab notebook (or recopy it) In class: Complete PART B along with all of the data collection, calculations, and analysis questions. If you do not have time to finish the analysis questions, finish this for homework. Day 3 Before Class: Review the lab – read PART C (Understanding Water Potential) Complete the pre-lab for PART C: o Title and date of the lab just say for the title - Osmosis/Diffusion Lab: Part C Understanding Water Potential o Purpose 1-2 sentences describing the overall goal of PART C; use complete sentences o Pre-Lab Questions you need to number the question, rewrite the question, and then answer it for full credit In class: Review water potential with the teacher. Water Potential Problems in lab notebook Day 4 Before class: Review the lab – read PART D (Observing Osmosis in Living Cells) Complete the pre-lab for PART D: o Title and date of the lab just say for the title - Osmosis/Diffusion Lab: Part D Observing Osmosis in Living Cells o Purpose 1-2 sentences describing the overall goal of PART D; use complete sentences o Pre-Lab Questions you need to number the question, rewrite the question, and then answer it for full credit o Lab Procedure Write a procedure for PART D. First list the materials that you will be using, then use your own words to describe the steps in experiment in paragraph form. o Data Tables/ Graphs Tape or draw these into your lab notebook. In class: Complete the initial portion of PART D of the lab Day 5 Before Class: n/a In class: Finish PART D of the lab along with all of the data collection, the graph, and complete the analysis activities and the analysis questions. If you do not have time to finish the analysis questions, finish this for homework. Remember to put both graphs (one for the data part of the lab and the other for one of the analysis questions) in your lab notebook. DIFFUSION/ OSMOSIS/ WATER POTENTIAL LAB BACKGROUND Cells must move materials through membranes and throughout cytoplasm in order to maintain homeostasis. The movement is regulated because cellular membranes, including the plasma and organelle membranes, are selectively permeable. Membranes are phospholipid bilayers containing embedded proteins; the phospholipid fatty acids limit the movement of water because of their hydrophobic characteristics. The cellular environment is aqueous, meaning that the solvent in which the solutes, such as salts and organic molecules, dissolve is water. Water may pass slowly through the membrane by osmosis or through specialized protein channels called aquaporins. Aquaporins allow the water to move more quickly than it would through osmosis. Most other substances, such as ions, move through protein channels, while larger molecules, including carbohydrates, move through transport proteins. The simplest form of movement is diffusion, in which solutes move from an area of high concentration to an area of low concentration; diffusion is directly related to molecular kinetic energy. Diffusion does not require energy input by cells. The movement of a solute from an area of low concentration to an area of high concentration requires energy input in the form of ATP and protein carriers called pumps. Water moves through membranes by diffusion; the movement of water through membranes is called osmosis. Like solutes, water moves down its concentration gradient. Water moves from areas of high potential (high free water concentration) and low solute concentration to areas of low potential (low free water concentration) and high solute concentration. So, water moves towards where there is MORE TOTAL SOLUTE. Solutes decrease the concentration of free water, since water molecules surround the solute molecules. The terms hypertonic, hypotonic, and isotonic are used to describe solutions separated by selectively permeable membranes. A hypertonic solution has a higher solute concentration and a lower water potential as compared to the other solution; therefore, water will move into the hypertonic solution through the membrane by osmosis. A hypotonic solution has a lower solute concentration and a higher water potential than the solution on the other side of the membrane; water will move down its concentration gradient into the other solution. Isotonic solutions have equal water potentials. In cells without a cell wall, such as animal cells, the movement of water into and out of a cell is affected by the relative solute concentration on either side of the plasma membrane. As water moves out of the cell, the cell shrinks; if water moves into the cell, it swells and may eventually burst. In cells with a cell wall, including fungal and plant cells, osmosis is affected not only by the solute concentration, but also by the resistance to water movement in the cell by the cell wall. This resistance is called turgor pressure. The presence of a cell wall prevents the cells from bursting as water enters; however, pressure builds up inside the cell and affects the rate of osmosis. Water movement in plants is important in water transport from the roots into the shoots and leaves. You likely will explore this specialized movement called transpiration in another lab investigation later in this course. General Safety Precautions You must wear safety glasses or goggles, aprons, and gloves because you will be working with acids and caustic chemicals. The HCl and NaOH solutions will cause chemical burns, and you should use these solutions in spill-proof trays or pans. Follow your teacher’s instructions carefully. Do not work in the laboratory without your teacher’s supervision. Talk to your teacher if you have any questions or concerns about the experiments. THE INVESTIGATIONS This investigation consists of three parts. In PART A, you use artificial cells (cubes of agar) to study the relationship of surface area and volume. In PART B, you create models of living cells (by using dialysis tubing to simulate cell membranes) to explore osmosis and diffusion. Finally, in PART C, you will be observing osmosis in living cells (potato) that are submerged in different concentrations of sugar solutions. You will also be learning how to solve water potential problems during this lab. --------------------------------------------------------------------------------------------------------------------------------------- PART A: Surface Area and Cell Size Overview – Part A: Cell size and shape are important factors in determining the rate of diffusion. Think about cells with specialized functions, such as the epithelial cells that line the small intestine OR plant root hairs. How do they aid in absorbing nutrients for the plant or the animal? In this section of the lab you are going to create 3 different size “cells” and look at how fast diffusion happens in each one. The cells will be made out of agar that has been treated with a pH indicator called bromothymol blue. When this indicator comes into contact with an acid (we are using vinegar to act as the acid), it will change from the blue color to a yellowish/clear color. Depending on how fast each block changes color completely, we will be able to figure out the diffusion rate and see how cell size affects that. PreLab Questions – Part A: Read the Background information for this lab and answer the prelab questions below in your lab notebook. Don’t forget to copy the question before answering it. Your answers do NOT need to be in complete sentences, but you do need to show your work for the calculations. You may need to refer to your text or an online source to answer some of the questions. 1. How do small intestinal epithelial and root hair cells function in absorbing nutrients and how does their structure aid in this? 2. What is Bromothymol Blue and how are we using it in Part A of this lab? 3. What is diffusion? 4. What is kinetic energy? 5. Why are most cells small? 6. Calculate the surface area of this cube. 7. Calculate the volume of this cube. 8. Calculate the surface area to volume ratio of this cube (See Procedure for help with how to do these calculations!) Materials – Part A: Scalpel Metric rulers Dishes Bromothymol Blue agar preparation* Vinegar *We will be using Bromothymol Blue Agar. Bromothymol Blue is an acid indicator. It is blue and turns yellow in the presence of acid. Procedure – Part A: 1. Cut your agar into 3 different size CUBES making sure that all the sides of each cube are the same size. You should end up with a small, medium, and large size cube. Do not make any cubes where the sides are larger than 1.5 cm. 2. Measure your cubes and enter the height, length, and width for each one into the Data Table 1.1. 3. Think about how size affects diffusion rate and hypothesize which cube will turn totally yellow the fastest, the second fastest, and then slowest. Record your hypothesis in Data Table 1.1. 4. Drop all 3 cubes into a cup with vinegar in it and note the time in your data table. As the vinegar (acid) diffuses into the agar, the pH indicator in the agar (bromothymol blue) changes from blue to yellow. When each cube turns totally yellow, note the time and record it in Table 1.1. 5. While you are waiting for the vinegar to diffuse, calculate surface area and volume for each of your cubes. Show your work in your lab notebook and put your answers in the data table. Surface Area = L x W x # of sides Volume = L x W x H 6. Calculate the ratio of SA:Vol by dividing the surface area by volume for each one. This should be a DECIMAL, not a FRACTION. Show your work in your lab notebook and put your answers in the data table. 7. After all your cubes are done, figure out how many minutes it took for the vinegar to diffuse all the way through each of your cubes. 8. Calculate the distance the vinegar traveled for each of the cubes: Distance = .5 x (smallest measurement of height, length or width) For example, if my cube is 1.4 cm on each side, the distance traveled would be .7cm. 9. Calculate the rate of diffusion in each of the cubes: Rate of Diffusion = Distance / Minutes to completion Data Table 1.1: Diffusion Rate of Vinegar in Agar Cube #1 Biggest Cube #2 Medium Cube #3 Smallest Height (cm) Length (cm) Width (cm) Hypothesis Time into Vinegar Time Finished # minutes for completed diffusion Surface Area (show calculations in lab notebook) Volume (show calculations in lab notebook) SA/Volume Ratio Distance Vinegar Traveled Rate of Diffusion (cm/min) Graph – Part A: Using a ruler, create a graph of your data. Time (min) goes on the x-axis (this will be the # of minutes it took to complete diffusion) and SA/V ratio (no units) goes on the y-axis. You will have 3 data points (one for each agar cube) and you will connect them to make a line graph. Label each of your data points as “Large,” “Medium,” or “Small” for the size of the cube the point represents. Analysis Questions – Part A: 1. Looking at your graph, what can you tell me about the correlation between the size of a cell and the rate of diffusion? 2. Calculate the following data values for the cells shown below. Make sure you SHOW YOUR WORK in your lab manual and clearly identify what the numbers mean. a. Surface area, volume and ratio for the one large cell b. Surface area, volume and ratio for 1 small cell c. Total surface area of all the small cells together 3. The outer layer of a plant root may contain elongated cells called root hair cells. Explain WHY this is an advantage to plants. Also, discuss how surface area to volume differs in root hair cells verses normal epithelial cells. (You may have to do some research on the shape of epithelial cells) -------------------------------------------------------------------------------------------------------------------------------------PART B: Modeling Diffusion and Osmosis Overview – Part B: In this experiment, you will create models of living cells by using dialysis tubing. Like cell membranes, dialysis tubing is made from a material that is selectively permeable to water and some solutes. You will fill your model cells with different solutions and determine the rate of diffusion. Depending on what solution is in the “cell” (dialysis tubing) and what solution is in the liquid surrounding the cell, several things can happen. Certain molecules and water can move in, they can move out, or it can stay the same. You will be experimenting with different types of solutions to see what happens in each. PreLab Questions – Part B: (some of these questions you may have to look up in your textbook or an online resource) 1. Why is it important for an IV solution to have salts in it? 2. What would happen if you were given pure water in your IV? 3. Describe how dialysis tubing is similar to a cell membrane. 4. Which of the following solutions do you think dialysis tubing is permeable to: Water Sucrose NaCl Glucose Protein 5. Why is it important to leave room in your dialysis tubing when you knot it? 6. Calculate the percent change for the sample data provided. (See Procedure for help with how to do these calculations!) Initial weight = 12.7g Final weight = 13.6g Materials – Part B: Distilled or tap water 1 M sucrose 1 M NaCl 1 M glucose 5% ovalbumin (egg white protein) 4 x 20 cm-long dialysis tubing Cups Balances Procedure – Part B: 1. 2. 3. 4. 5. Set up 4 pairs of different solutions. One solution from each pair will be in the dialysis tubing (which represents the cell), and the other will be outside the cell in the cup. a. Set-Up #1 of the model cell will have water inside and outside; this is your control. b. Set-Up #2 of the model cell will have a glucose solution inside and water outside. c. You will choose the set-up of #3 and #4. Look at the list of solutions from the materials before choosing your solutions. IF YOU ARE CHOOSING TO DO WATER AS ONE OF YOUR SOLUTIONS, THAT ALWAYS GOES ON THE OUTSIDE! CHOOSE YOUR SOLUTIONS AND RECORD THEM IN Data Table 1.2 Before setting up your experiment, use your knowledge about solute gradients to hypothesize whether the water will diffuse INTO or OUT OF the cell for EACH set-up. Record your hypothesis for each beaker in Data Table 1.2. Make dialysis tubing cells by tying a knot in one end of four pieces of dialysis tubing. Fill each “cell” with 10 mL of the solution you chose for the inside, and knot the other end, making sure to leave enough space for water to diffuse into the cell. Make sure not to mix up your “cells” so you know which one goes with each beaker. Mass each cell, record the initial mass (Data Table 1.2), and then place it into a cup filled with the second solution for that pair (make sure you label the cups to indicate what solution is inside the cell and inside the cup and what # set-up it is). Allow the “cells” to be submerged for 30 minutes and then record the final mass in Data Table 1.2. Calculate the percent change in weight for each setup and record your results. Show your work in your lab notebook! To calculate the percent change, use the following formula: % Change = (final mass – initial mass)/initial mass X 100 Data Table 1.2: Percent Change in Dialysis Tubing Cells in Various Solutions Set-Up In Dialysis Tubing “Cell” In Surrounding Solution “Environment” Water Water Glucose Water #1 #2 #3 #4 Hypothesis Initial Weight (g) Final Weight (g) Difference Percent Change (%) Analysis Questions – Part B: 1. For each set-up, explain which way water moved and WHY you think this occurred. 2. From your results, which solutes, if any, diffused across the membrane and which, if any, were restricted? WHY do you think this occurred? (I know you don’t actually know for sure if solutes diffused, but just tell me what you think and why) 3. Explain the relationship between the size of solute molecule and its ability to diffuse across a membrane. --------------------------------------------------------------------------------------------------------------------------------------PART C: Understanding Water Potential Overview – Part C: Water potential predicts which way water diffuses through plant tissues and is abbreviated by the Greek letter psi (Ψ). Water potential is the free energy per mole of water and is calculated from two major components: (1) the solute potential (ΨS), which is dependent on solute concentration, and (2) the pressure potential (ΨP), which results from the exertion of pressure—either positive or negative (tension) — on a solution. The solute potential is also called the osmotic potential. Ψ = ΨP + ΨS Water Potential = Pressure Potential + Solute Potential Water moves from an area of higher water potential or higher free energy to an area of lower water potential or lower free energy. Water potential measures the tendency of water to diffuse from one compartment to another compartment. The water potential of pure water in an open beaker is zero (Ψ = 0) because both the solute and pressure potentials are zero (ΨS = 0; ΨP = 0). An increase in positive pressure raises the pressure potential and the water potential. The addition of solute to the water lowers the solute potential and therefore decreases the water potential. This means that a solution at atmospheric pressure (normal pressure) has a negative water potential due to the solute. Adding Pressure INCREASES Water Potential Adding Solute DECREASES Water Potential To figure out the SOLUTE POTENTIAL, use the following equation: (ΨS) = – iCRT i is the ionization constant C is the molar concentration R is the pressure constant (R = 0.0831 liter bars/mole-K) T is the temperature in K (273 + °C). To determine i, figure out if there are any ions. For example, if the molecule does not ionize in water (like glucose or sucrose), then i = 1. If the solution DOES ionize in water, such as NaCl where we have 2 ions (Na+ and Cl-), then i = 2. To determine C, find the molar concentration. So, if we have a 1.0 M solution, then C = 1. If we have a 0.6 M solution, then C = .6. R will always be .0831 liter bars/mole °K To find T, it is the temperature in Kelvin. So, determine the temperature in Celsius and add 273. For example, if the temperature is 20 °C, then T = 293. Also, water potential (or solute potential) is measured in bars. (that is the unit) When a cell’s cytoplasm is separated from pure water by a selectively permeable membrane, water moves from the surrounding area, where the water potential is higher (Ψ = 0), into the cell, where water potential is lower because of solutes in the cytoplasm (Ψ is negative). It is assumed that the solute is not diffusing (Figure 1a). The movement of water into the cell causes the cell to swell, and the cell membrane pushes against the cell wall (since the potato is a plant cell) to produce an increase in pressure. This pressure, which counteracts the diffusion of water into the cell, is called turgor pressure. Over time, enough positive turgor pressure builds up to oppose the more negative solute potential of the cell. Eventually, the water potential of the cell would equal zero and thus be the same as the water potential of the pure water outside the cell (Ψ of cell = Ψ of pure water = 0). At this point, a dynamic equilibrium is reached and therefore there is no net movement of water molecules (Figure 1b). Figures 1a-b. Plant cell in pure water. The water potential was calculated at the beginning of the experiment (a) and after water movement reached dynamic equilibrium and the net water movement was zero (b). In Figure 2, if solute is added to the water surrounding the plant cell, the water potential of the solution surrounding the cell decreases. If enough solute is added, the water potential outside the cell is equal to the water potential inside the cell, and then there will be no net movement of water. However, the solute concentrations inside and outside the cell are not equal, because the water potential inside the cell results from the combination of both the turgor pressure (ΨP) and the solute pressure (ΨS). (See Figure 2.) There is no pressure potential (ΨP) in the surrounding solution because the beaker is open to the environment. Figure 2. Plant cell in an aqueous solution. The water potential of the cell equals that of surrounding solution at dynamic equilibrium. The cell’s water potential equals the sum of the turgor pressure potential PLUS the solute potential. The solute potentials of the solution and of the cell are not equal. If more solute is added to the water surrounding the cell, water will leave the cell, moving from an area of higher water potential to an area of lower water potential. The water loss causes the cell to lose turgor pressure. A continued loss of water will cause the cell membrane to shrink away from the cell wall (plasmolysis). --------------------------------------------------------------------------------------------------------------------------------------PreLab Questions – Part C: 1. Describe the relative solute concentration and water potential in the following solutions: a. Hypertonic b. Hypotonic 2. Why do we need to consider pressure in plant cell osmosis but not animal cell osmosis? 3. What is the water potential in an open beaker of pure water? 4. Why does osmosis eventually stop in a plant cell, even if the environment is hypotonic? 5. Calculate the solute potential of a 0.1M NaCl solution at 25 °C. 6. Calculate the solute potential of a 0.3M NaCl cell at 25 °C. **Refer to the sample water potential problems to figure out #5 and #6** 7. Which way will the water move if the cell (#6) is placed in the solution (#5). 8. What must the turgor pressure (ΨP) equal if there is no net movement when the cell (#6) is placed in the solution (#5)? SAMPLE WATER POTENTIAL PROBLEMS Here are two sample water potential problems that you can look at before you try some on your own. You do not have to write these in your lab notebook, they are for your reference only. Problem #1 The value of Ψ in a leaf tissue was found to be -4.1 bars. If you take that leaf tissue and put it in a 0.2 M solution of NaCl (which dissociates into 2 particles – Na+ and Cl- in water) at 22°C in an open beaker, what is the Ψ of the solution? Which direction will the net flow of water be? (ΨS) = – iCRT Solution OUTSIDE the Cell i = 2; C = 0.2; R = 0.0831; T = 295 ΨS = -(2)(0.2)(0.0831)(295) ΨS = -9.8 Because it is in an OPEN beaker, ΨP = 0 So…total Ψ for outside Ψ = ΨP + ΨS Ψ = 0 + (-9.8) Ψ = -9.8 bars If the solution as a Ψ of -9.8 bars and the leaf cell has a Ψ of -4.1 bars, the water will move towards the lower water potential, so it will move OUT OF THE LEAF into the solution. (-9.8 is lower than -4.1) Problem #2 At 25°C, a plant cell containing 0.8M glucose is in equilibrium with its surrounding solution containing a 0.5 M glucose solution in an open container. What is the cell’s ΨP? (ΨS) = – iCRT OUTSIDE the Cell i = 1; C = 0.5; R = 0.0831; T = 298 ΨS = -(1)(0.5)(0.0831)(298) ΨS = -12.4 Because it is in an OPEN beaker, ΨP = 0 So…total Ψ for outside Ψ = ΨP + ΨS Ψ = 0 + (-12.4) Ψ = -12.4 INSIDE the Cell i = 1; C = 0.8; R = 0.0831; T = 298 ΨS = -(1)(0.8)(0.0831)(298) ΨS = -19.8 To find the ΨP of the cell, we need to keep in mind that the problem told us that the cell is in equilibrium with the surrounding solution…SO Ψ of the solution is equal to Ψ of the cell. We know that the Ψ of the solution is -12.4, and we know that ΨS of the cell is -19.8. So, we can figure out ΨP: Ψ = ΨP + Ψ S -12.4 = ΨP + -19.8 7.4 bars = ΨP Part C ACTIVITY: Water Potential Problems You can either copy each question and then answer it, or cut out the questions, tape them in your lab notebook and then answer them. Make sure to SHOW ALL WORK and label your answers clearly! 1. If a cell’s ΨP = 3 bars and its ΨS = -4.5 bars, what is the resulting Ψ? 2. The cell from question #1 is placed in a beaker of sugar water with ΨS = -4.0 bars. In which direction will the net flow of water be? 3. The value for Ψ in root tissue was found to be -3.3 bars. If you take the root tissue and place it in a 0.1 M solution of sucrose at 20°C in an open beaker, what is the Ψ of the solution, and in which direction would the net flow of water be? 4. NaCl dissociates into 2 particles in water: Na+ and Cl-. If the solution in question 3 contained 0.1M NaCl instead of 0.1M sucrose, what is the Ψ of the solution, and in which direction would the net flow of water be? 5. A plant cell with a Ψs of -7.5 bars keeps a constant volume when immersed in an openbeaker solution that has a Ψs of -4 bars. What is the cell’s ΨP? 6. At 20°C, a cell containing 0.6M glucose is in equilibrium with its surrounding solution containing 0.5M glucose in an open container. What is the cell’s ΨP? 7. At 20°C, a cell with ΨP of 3 bars is in equilibrium with the surrounding 0.4M solution of sucrose in an open beaker. What is the molar concentration of sucrose in the cell? --------------------------------------------------------------------------------------------------------------------------------------Part D Observing Osmosis in Living Cells Overview – Part D: The interactions between selectively permeable membranes, water, and solutes are important in cellular and organismal functions. For example, water and nutrients move from plant roots to the leaves and shoots because of differences in water potentials. In this part of the lab, we are going to look at how water moves into and out of the cells in a potato by putting the potato cores in several different solutions of varying osmolarity. We will use sucrose solutions that are 0.0M, 0.2M, 0.4M, 0.6M, 0.8M, and 1.0M. Each solution is color coded, and depending on whether the potato gains or loses weight after being submerged for 24 hours, and how much mass is lost/gained, you will be able to determine which solution is which color. Also, you will be able to determine the molar concentration of the potato core. PreLab Questions – Part D: 1. At what sucrose concentration would you predict the potato cores going to gain the most mass? Lose the most? 2. How do you calculate percent change in mass? Materials – Part D: Balances 6 Color-coded sucrose solutions Potato Cores Plastic Cups Graduated cylinder Knife/Scalpel Procedure – Part D: 1. Get 6 plastic cups and label them with your block number and group names. Then, label each cup with one of the following colors: blue, clear, green, purple, red, yellow 2. Go to the stock solutions and fill each of your cups with the 75 mL of the appropriate solution. 3. Get a potato and slice it into about 1-cm thick slices. Then cut those slices into rectangular cubes. Remove all the skin from your potato cores. You need 4 rectangles for each cup (total = 24 cubes). 4. Take 4 potato cores and mass them together (you want the total mass of all 4 – not the individual masses of each). Write down their initial mass (in grams) and put them in the cup labeled blue. Remember to record the initial mass in Data Table 1.3. Make sure the solution totally covers the potato cores (if it does not, add a little more solution). 5. Repeat Step 4 for each of the other colors keeping in mind to add 4 pieces of potato for each one. Record all data in Data Table 1.3. 6. Cover your cups with plastic wrap to prevent evaporation and let it stand overnight. 7. The next day, remove the cores from the cups, blot them gently on a paper towel, and determine their final mass. Record this information in the data table. Do this for each color. You may then dispose of the solutions down the drain and throw away the plastic cups and potatoes. 8. Complete the data table and then record your Percent Change in Mass on the teacher’s computer so we can create the Class Data Table. Percent Change in Mass = Final Mass – Initial Mass Initial Mass x 100 9. Once the Class Data is recorded on the computer, fill in that information in Data Table 1.4. Data Table 1.3: Potato Core Results – Individual Data Solution Color Initial Mass (g) Final Mass (g) Mass Difference Percent Change in Mass* Class Average Percent Change in Mass Blue Clear Green Purple Red Yellow *Remember to put a + or – in front of your percent change in mass to reflect whether your potatoes GAINED (+) or LOST (-) weight Data Table 1.4: Potato Core Results – Class Data Solution Percent Change in Mass of Potato Cores (+/-) Total Class Color Team Team Team Team Team Team Team Average 1 2 3 4 5 6 7 Blue Clear Green Purple Red Yellow *Remember to put a + or – in front of your percent change in mass to reflect whether your potatoes GAINED (+) or LOST (-) weight Analysis Activities – Part D: Determining the Molarity of the Solutions Determine which color solutions are which molarity of sucrose by looking at the class data and observing which colors caused the potatoes to gain/lose the most mass. The options for molarity are 0.0M, 0.2M, 0.4M, 0.6M, 0.8M, and 1.0M. Fill in Data Table 1.5 below: Data Table 1.5 – Solution Colors and Their Corresponding Molarities Color Solution Percent Change in Mass – Class Average (from Table 1.4) Molarity of Solution (M) Blue Clear Green Purple Red Yellow Using your completed Table 1.5, graph the percent change in mass at different molarities in Graph 1.1. Notice that at the beginning of the graph it is 0M, and at the end of the graph it is 1.0M. Evenly fill in the other molarities. Then, fill in values for the mass along the y-axis. Notice that 0 is in the middle of the graph to allow you to plot both positive and negative changes in mass. Plot your points (we are using the CLASS DATA here – so refer to Table 1.5 again) and then draw a STRAIGHT LINE of best fit based on your data. Graph 1.1 – Percent Change in Mass of Potato Cores at Different Molarities of Sucrose Determining the Molar Concentration of the Potato Core In order to determine the molar concentration of the potato core (this will represent “C” in your water potential equation), look at where your best-fit line crosses zero. Hint – it should be somewhere near 0.3M. Write that value in the box below: Molar Concentration of Sucrose = ___________ M Calculate the Water Potential of the Potato Cells The water potential of the sucrose solution at equilibrium will be equal to the water potential of the potato cells. The ΨP is zero, so you just need to calculate ΨS. Assume the temperature is 22°C. Show your work in your lab notebook! Recall: (ΨS) = – iCRT Analysis Questions – Part D: 1. Which color solution had the highest concentration of sucrose? How do you know this? 2. What is the water potential of the potato cell? (you calculated this above) 3. If a potato core is allowed to dehydrate by sitting in the open air, would the water potential of the potato cells decrease or increase? Why? 4. What effect does adding solute have on the solute potential component of that solution? 5. If a plant cell has a lower water potential than its surrounding environment and if pressure is equal to zero, is the cell hypertonic or hypotonic to its environment? Will the cell gain or lose water? Explain 6. (3 parts to this question!) Zucchini cores placed in sucrose solutions at 27°C resulted in the following percent changes after 24 hours: % Change Sucrose in Mass Molarity 20% 0.0 10% 0.2 -3% 0.4 -17% 0.6 -25% 0.8 -30% 1.0 You need to: A: Graph the results (just like we did for our potato cores; use the graph below) B: Calculate the Molar concentration of solutes within the zucchini cells (where does it cross the 0 line?) Remember, this will be the “C” in your water potential equation. C. Calculate the water potential of the solutes within the zucchini cores. ΨP = 0, so just find ΨS (ΨS) = – iCRT