Science Lab

Science Lab
Unit 3: Properties of Matter
07-PS1-2
1. I can identify and classify matter using their physical properties.
2. I can identify and describe the evidence of chemical and physical changes in matter.
3. I can determine if a reaction has occurred by analyzing properties of a substance.
• Properties of Matter
• Mass
• Volume
• Denstiy
• Solubility
• Melting & Boiling Point
• State of Matter
• Metallic Properties
• Length
• Other: Shape, Size, and Color
Mass
• Definition
– the amount of matter in an object
• How do we Measure?
– Triple Beam Balance
– Digital Scale
• Mass of a Liquid
– Use a graduated cylinder and take the mass of the empty cylinder
– Pour liquid in and then measure the graduate cylinder again
– Take the difference= mass of the liquid
• Mass of a solid
– Simply place object on balance or digital scale.
• Unit
– Grams
• 1000mg=1 gram
• 1000g=1 kg
( Mini-Lab Mass of a solid, Mass of Liquid)
Volume
• Definition
– The amount of space an object takes up
• How do we Measure?
– Ruler, graduated cylinder
• Volume of a liquid
– Simply read the graduated cylinder
– Unit- mL (milliliter)
• Volume of a Regular shaped object
– Use a ruler and measure the length, width, and height of the object
– L x W x H
– Unit- cmᶟ
• Volume of an irregular sized object
– Water Displacement Test
• Fill a graduated cylinder up to a desired amount. Usually 100 mL
• Place the object into the water and take a reading on graduated cylinder
• Subtract 100mL from the reading = volume of an irregular sized object
– Unit- cmᶟ
(Lab- How to Measure Volume)
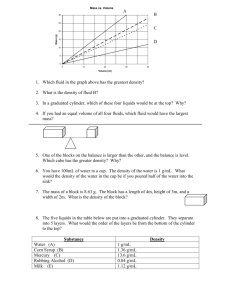
Density
• Definition
– the amount of mass a material has in a given volume
– D=M/V
• How do we measure?
– Ruler, graduated cylinder, scale or triple beam balance
• Density of a solid and liquid
– Take the mass of the object on the scale.
– Take the volume of the object.
• Refer to Volume notes on how to do so.
– Mass divided by volume
– Unit- g/cmᶟ or cubic mL
• Comparing densities of different liquids
– Build a density column
(Cover Density PPT activity and Liquid Layer Lab and Density for Solids Lab)
Solubility
• Definition
– The number of grams of one substance will dissolve in 100g of another substance at a given temperature.
• How do we measure?
– Hot plate, stirrer, beaker, thermometer
(
Dissolving Rates and Solutions Lab)
Melting Point & Boiling Point
• Definition
– Melting point- the temperature at which a solid changes to liquid
• MP of Water= 0°C
– Boiling Point- the temperature at which a liquid changes to gas.
• BP of water=100°C
• It does NOT depend on the size of the sample the MP and BP are ALWAYS the same.