Lecture 9

Lecture 17
– Exams in Chemistry office with M’Lis.
Please show your ID to her to pick up your exam.
– Quiz on Friday
– Enzyme mechanisms
General Acid-Base Catalysis
• General acid catalysis - a process in which partial proton transfer from a Brønstead acid (a species that can donate protons) lowers the free energy of a reaction’s transition state.
• General base catalysis - process in which partial proton abstraction by a Brønstead base (a species that can combine with a proton) lowers the free energy of a reaction’s transition state.
• General acid-base catalysis -a combination of both.
Figure 15-1a Mechanisms of keto –enol tautomerization.
( a ) Uncatalyzed.
Figure 15-1b Mechanisms of keto –enol tautomerization.
( b ) General acid catalyzed.
Figure 15-1c
Mechanisms of keto –enol tautomerization.
(
c
) General base catalyzed.
General Acid Base Catalysis
• Ex. Ester hydrolysis
O d
+ H +
O
H
H
2
O
O
H
C d
OR C d
OR C
O
+
OR
H H
O
C OH
+ ROH
- H + O
C
H
O
H
+
OR
H +
General Acid-Base Catalysis
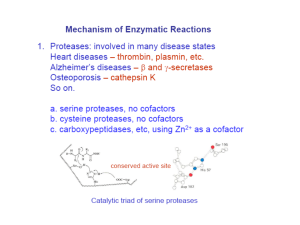
• Large number of possible amino acids
• Requires that they can accept and donate a proton
• Glu, Asp
• Lys, His, Arg
• Cys, Ser, Thr
• Also can include metal cofactors
• Example can be observed in carboxypeptidase A (both acid and base catalysis)
General Acid-Base Catalysis
• Ex. Carboxypeptidase A
Zn plays role of acid (4 th ligand is normally H
2
O, but it is displaced by peptide binding)
Glu 72
R
H-C CO
2
-
His 196
His 69
Zn ++ O
N H d d
C
O
H d d
O
H
H-C-R
NH
C O
Glu 270
C-O -
Glu acts as base catalyst to polarize water and form nucleophile
+ Arg 145
Key aas that holds molecule in place
HO-Tyr 248
Tyr also plays role as 2 nd acid catalyst
+ Arg
Study of Enzyme Mechanisms
• X-ray crystallography-crystallize the molecule with substrate in place and compare to crystal structure of the molecule without the substrate (differences in structure)
• For carboxypeptidase A they could show that
• Water is expelled by binding of substrate
• Arg145 moves 2Å closer to the carboxyl group of the substrate
• Glu270 moves 2Å towards the C=O group
• Tyr248 moves 12Å towards the amide plane of the peptide
• Also able to show what aa surround certain groups-
Tyr248 in a hydrophobic pocket.
Study of Enzyme Mechanisms
• Check the pH profile of the enzyme.
• For carboxypeptidase
• The coordination of Zn by His69 and His196 (pK 6.0)
• Tyr248 (pK 9.1)
Log (V max
/K
M
)
6.7
8.5
Example in book: RNAse
(p. 499)
6 7 pH
8 9
Lysozyme (Strain and Acid Catalysis)
• Hen Egg White (HEW) Lysozyme digests bacterial cell walls.
• Cleaves
(1 4) glycosidic linkages from Nacetylmuramic acid (NAM) to Nacetylglucosamine (NAG)
• Requires about 6 sugars for good recognition.
SugA-SugB-SugC-SugD-SugE-SugF
Lysozyme (Strain and Acid Catalysis)
• In theory
Asp 52
SugA-SugB-SugC-SugD-SugE-SugF
C
O O d O
O
E
O
C
D
O H +
OH
OH
O
OR
OH
C
OH
Glu 35
• Must distort ring into a flat, planar shape
• Supply acid catalysis
Lysozyme (Strain and Acid Catalysis)
• In practice
Figure 15-12 Interactions of lysozyme with its substrate.
Figure 15-11 Chair and halfchair conformations.
Study of Enzyme Mechanisms
• Lysozyme
• Only the D ring is strained
• Glu35 is in a hydrophobic environment
• Asp52 is in a hydrophilic environment
• Covalent modification of the active site
• Block essential groups
• May or may not act at active site
• Cd or R-As=O (trivalent arsenic)
The Aspartate Proteases
• Pepsin, Renin, HIV protease (AZT targets this)
• General acid-base catalysis
Serine hydrolases: trypsin, chymotrypsin, elastase
• Synthesized in pancreas as inactive zymogen (ex. trypsinogen)
• Generally operate by " charge relay system"
• Asp102, His57, Ser195 conserved in all 3 enzymes.
Asp 102
COO
-
His 57
Ser 195
H N NH
O
H
R
N
H
C
O
R'
1
Serine hydrolases: trypsin, chymotrypsin, elastase
Asp 102
COO H
Asp 102
COO
-
N
H N
His 57
His
NH
57
NH
Ser 195
O
R'
C
O
-
R
N
H
Ser 195
O
C
O
R'
RNH
2
2
Rate limiting step for amides
Serine hydrolases: trypsin, chymotrypsin, elastase
Asp 102
COO
-
Asp 102
COO H
H N
His 57
NH
H
Ser 195
H
O
O
C
O
R'
His 57
N NH
Ser 195
O
R'
C
O
-
H
O
3
Rate limiting step for esters
Serine hydrolases: trypsin, chymotrypsin, elastase
Asp 102
COO
-
H N
His 57
NH
Ser 195
O
H
H
O
C
O
R'
Charge-relay systems
• Relay charges between amino acid side chains in order to catalyze the reaction.
Summary: various methods to increase rate
• Increase frequency of the correct group in the correct place e.g. proximity effect
• Lower E
A by specific catalysis -acid-base catalysis, nucleophile or electrophile
• Raise energy of reactants (closer to E
A
) - ring distortion, transition state analog
• Provide alternate low E
A
• Michaelis Menten pathway - covalent catalysis.]
• Lineweavear Burk
• Eadie Hofstee
• Competitive inhibition
• Noncompetitive inhibition
Terms to review for enzymes
• Cofactor
• Coenzyme
• Prosthetic group
• Holoenzyme
• Apoenzyme
• Lock and Key
• Transition analog model
• Induced fit
• Active site, binding site, recognition site, catalytic site
Catalytic Mechanisms
• Acid-base catalysis
• Covalent catalysis
• Metal ion catalysis
• Proximity and orientation effects (ex. anhydride)
• Preferential binding of the transition state complex
General Acid-Base Catalysis
• Large number of possible amino acids
• Requires that they can accept and donate a proton
• Glu, Asp
• Lys, His, Arg
• Cys, Ser, Thr
• Also can include metal cofactors (metal ion catalysis)
• Example can be observed in RNAse
Figure 15-2 The pH dependence of V¢ max
/K¢
M in the
RNase A –catalyzed hydrolysis of cytidine-2 ¢ ,3 ¢ -cyclic phosphate.
Example in book: RNAse
(p. 499)
RNAse mechanism
• His12 acts as general basetakes proton from
RNA 2’-OH-making a nucleophile which attacks the phosphate group.
• His119 acts as a general acid to promote bond scission.
•2’,3’ cyclic intermediate is hydrolyzed through the reverse of the first step-water replaces the leaving group. His12 is the acid, His119 acts as the base
Covalent catalysis
• Rate acceleration through the transient formation of a catalyst-substrate covalent bond.
• Example-decarboxylation of acetoacetate by primary amines
• Amine nucleophilically attacks carbonyl group of acetoacetate to form a Schiff base (imine bound)
Figure 15-4 The decarboxylation of acetoacetate.
uncatalyzed e sink
Catalyzed by primary amine
Covalent catalysis
• Made up of three stages
1. The nucleophilic reaction between the catalyst and the substrate to form a covalent bond.
2. The withdrawal of electrons from the reaction center by the now electrophilic catalyst
3. The elimination of the catalyst (reverse of 1.)
• Nucleophilic catalysis - covalent bond formation is limiting.
• Electrophilic catalysis-withdrawal of electrons is rate limiting
Covalent catalysis
• Nucleophilicity is related to basicity . Instead of abstracting a proton, nucleophilically attacks to make covalent bond.
• Good covalent catalysts must have high nucleophilicity and ability to form a good leaving group.
• Polarized groups (highly mobile e-) are good covalent catalysts: imidazole, thiols.
• Lys, His, Cys, Asp, Ser
• Coenzymes: thiamine pyrophosphate, pyridoxal phosphate.
Covalent Catalysis
• Form transient, metastable intermediates that can supply bond energy into the reaction.
Side chain structures
O NH
Examples
Serine
RC -O-CH
2
-CH
(acyl ester) COO-
Chymotrypsin
Trypsin
Elastase acetylcholinesterase
Serine
Phosphoglucomutase
Alkaline phosphatase
O NH
O-P -O-CH
2
-CH
O
(phosphoryl ester)
COO-
Covalent Catalysis
Group
Cysteine
Histidine structures
O NH
RC -S-CH
2
-CH
(acyl cysteine) COO-
Examples
Papain
3-PGAL-DH
NH
CH
O
O-P -N
O
(phosphoryl imidazole)
COO-
Succinate thiokinase
Group
Lysine
Covalent Catalysis
R' structures
NH
R-C =N-(CH
2
)
4
-CH
(Schiff base) COO-
Examples
Aldolase
Transaldolase
Metal ion catalysis
• Almost 1/3 of all enzymes use metal ions for catalytic activity. 2 main types:
1. Metalloenzymes -have tightly bound metal ions, mmost commonly transition metal ions such as Fe 2+ , Fe 3+ , Cu 2+ ,
Zn 2+ , Mn 2+ , or Co 3+
2. Metal-activated enzymes -loosely bind metal ions form solution-usually alkali or alkaline earth metals-Na + , K + ,
Ca 2+
Metal ion catalysis
• Three ways for catalysis
1. Binding to substrates to orient them properly for the reaction
2. Mediating oxidation-reduction reactions through reversible changes in the metal ion’s oxidation state
3. Electrostatically stabilizing or shielding negative charges.
Serine Hydrolases (Proteases)
• Chymotrypsin, trypsin and elastase.
• All have a reactive Ser necessary for activity.
• Catalyze the hydrolysis of peptide (amide) bonds.
• Chymotrypsin can act as an esterase as well as a protease.
• Study of esterase activity provided insights into the catalytic mechanism.
CH
3
O
C O NO
2 p -Nitrophenylacetate
Chymotrypsin
H
2
O
2H +
O
CH
3
C
Acetate
O +
O NO
2 p -Nitrophenolate
Serine Hydrolases (Proteases)
• Reaction takes place in 2 phases
1.
The “burst phase”-fast generation of p- nitrophenolate in stoichiometric amounts with enzyme added
2.
The “steady-state phase”-p-nitrophenolate generated at reduced but constant rate; independent of substrate concentration.
Figure 15-18 Time course of
p
nitrophenylacetate hydrolysis as catalyzed by two different concentrations of chymotrypsin.
CH
3
O
C O NO
2 p -Nitrophenylacetate
+ Enzyme
Chymotrypsin
FAST O NO
2
O p -Nitrophenolate
SLOW
CH
3
C OEnzyme
Acyl-enzyme intermediate
O
H
2
O
2H +
CH
3
C
Acetate
O -
+ Enzyme
Chymotrypsin
• Follows a ping pong bi bi mechanism.
• Rate limiting step for ester hydrolysis is the deacylation step.
• Rate limiting step for amide hydrolysis is first step (enzyme acylation).
Identification of catalytic residues
• Identified catalytically important residues by chemical labeling studies.
(active Ser)-CH
2
OH
+
• Ser195-identified using diisopropylphosphofluoridate (DIPF)
• Irreversible!
CH(CH
3
)
2
O
F-P=O
O
CH(CH
CH(CH
O
3
)
3
Diisopropylphospho
-fluoridate (DIPF)
2
)
2
DIP-enzyme
(active Ser)-CH
2
O-P=O
O
CH(CH
3
)
2
Identification of catalytic residues
• His57 was identified through affinity labeling
• Substrate analog with a reactive group that specifically binds to the active site of the enzyme forms a stable covalent bond with a nearby susceptible group.
• Reactive substrate analogs are sometimes called “Trojan horses” of biochemistry.
• Affinity labeled groups can be identified by peptide mapping.
• For chymotrypsin, they used an analog to Phe.
Identification of catalytic residues
CH
3
O
O
S NH
CH
2
O
CH C CH
2
Cl
Tosyl-L-phenylalanine chloromethyl ketone (TPCK)
Figure 15-19 Reaction of TPCK with chymotrypsin to alkylate His 57.
Homology among enzymes
• Bovine chymotrypsin, bovine trypsin and porcine elastase are highly homologous
• ~40% identical over ~240 residues.
• All enzymes have active Ser and catalytically essential
His
• X-ray structures closely related.
• Asp102 buried in a solvent inaccessible pocket (third enzyme in the “catalytic triad”)
X-ray structures explain differences in substrate specificity
• Chymotrypsin bulky aromatic side chains (Phe, Trp, Tyr) are preferred and fit into a hydrophobic binding pocket located near catalytic residues.
• Trypsin - Residue corresponding to chymotrypsin Ser189 is Asp (anionic). The cationic side chains of Arg and Lys can form ion pairs with this residue.
• Elastase - Hydrolyzes Ala, Gly and Val rich sequences.
The specificity pocket is largely blocked by side chains of
Val and a Thr residue that replace Gly residues that line the binding pocket of chymotrypsin and trypsin.
Figure 15-20a X-Ray structure of bovine trypsin.
( a ) A drawing of the enzyme in complex.
Figure 15-20b X-Ray structure of bovine trypsin. ( b ) A ribbon diagram of trypsin.
Figure 15-20c X-Ray structure of bovine trypsin. ( c ) A drawing showing the surface of trypsin
( blue ) superimposed on its polypeptide backbone
( purple ).
Figure 15-21 The active site residues of chymotrypsin.
Figure 15-22 Relative positions of the active site residues in subtilisin, chymotrypsin, serine carboxypeptidase II, and
ClpP protease.
Figure 15-23
Catalytic mechanism of the serine proteases.