Intermolecular Forces: Chemistry Presentation
advertisement

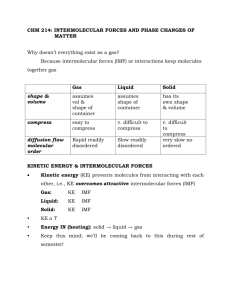
Intermolecular Forces Irresistible attraction… ATTRACTIVE FORCES Always electrostatic in nature Intramolecular forces bonding forces These forces exist within each molecule. They influence the chemical properties of the substance. Intermolecular forces nonbonding forces These forces exist between molecules. They influence the physical properties of the substance. Why Are Molecules Attracted to Each Other? • Intermolecular attractions are due to attractive forces between opposite charges – + ion to − ion – + end of polar molecule to − end of polar molecule • H-bonding especially strong – even nonpolar molecules will have temporary charges • Larger charge = stronger attraction • Longer distance = weaker attraction • However, these attractive forces are small relative to the bonding forces between atoms – generally smaller charges – generally over much larger distances Dipole–Dipole Attractions • Polar molecules have a permanent dipole – because of bond polarity and shape – dipole moment – as well as the always present induced dipole • The permanent dipole adds to the attractive forces between the molecules – raising the boiling and melting points relative to nonpolar molecules of similar size and shape Polar molecules and dipole-dipole forces. solid liquid Hydrogen Bonding • When a very electronegative atom is bonded to hydrogen, it strongly pulls the bonding electrons toward it – O─H, N─H, or F─H • Because hydrogen has no other electrons, when its electron is pulled away, the nucleus becomes deshielded – exposing the H proton • The exposed proton acts as a very strong center of positive charge, attracting all the electron clouds from neighboring molecules H-Bonding HF Drawing Hydrogen Bonds Between Molecules of a Substance PROBLEM: Which of the following substances exhibits H bonding? For those that do, draw two molecules of the substance with the H bonds between them. O (a) C2H6 (b) CH3OH (c) CH3C NH2 Find molecules in which H is bonded to N, O or F. Draw H bonds in the format -B: H-A-. SOLUTION: (a) C2H6 has no H bonding sites. H H O (b) (c) O H N CH3C CH C H C O H 3 H H N H O CH3C N H H O C H H H H H H N CH3C O Dispersion Force Dispersion forces among nonpolar molecules. separated Cl2 molecules instantaneous dipoles Predicting the Type and Relative Strength of Intermolecular Forces PROBLEM: For each pair of substances, identify the dominant intermolecular forces in each substance, and select the substance with the higher boiling point. (a) MgCl2 or PCl3 (b) CH3NH2 or CH3F (c) CH3OH or CH3CH2OH CH3 (d) Hexane (CH3CH2CH2CH2CH2CH3) CH3CCH2CH3 or 2,2-dimethylbutane CH3 Use the formula, structure •Bonding forces are stronger than nonbonding(intermolecular) forces. •Hydrogen bonding is a strong type of dipole-dipole force. •Dispersion forces are decisive when the difference is molar mass or molecular shape. Predicting the Type and Relative Strength of Intermolecular Forces SOLUTION : Mg2+ and Cl- are held together by ionic bonds while PCl is covalently (a) 3 bonded and the molecules are held together by dipole-dipole interactions. Ionic bonds are stronger than dipole interactions and so MgCl2 has the higher boiling point. (b) CH3NH2 and CH3F are both covalent compounds and have bonds which are polar. The dipole in CH3NH2 can H bond while that in CH3F cannot. Therefore CH3NH2 has the stronger interactions and the higher boiling point. (c) Both CH3OH and CH3CH2OH can H bond but CH3CH2OH has more CH for more dispersion force interaction. Therefore CH3CH2OH has the higher boiling point. (d) Hexane and 2,2-dimethylbutane are both nonpolar with only dispersion forces to hold the molecules together. Hexane has the larger surface area, thereby the greater dispersion forces and the higher boiling point. Summary diagram for analyzing the intermolecular forces in a sample. INTERACTING PARTICLES (atoms, molecules, ions) ions present ions only IONIC BONDING (Section 9.2) ions not present polar molecules only DIPOLE-DIPOLE FORCES ion + polar molecule ION-DIPOLE FORCES H bonded to N, O, or F HYDROGEN BONDING nonpolar molecules only DISPERSION FORCES only polar + nonpolar molecules DIPOLEINDUCED DIPOLE FORCES DISPERSION FORCES ALSO PRESENT