Heating Curve 1
advertisement

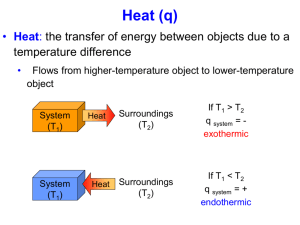
Heat Calculations Agenda • Review specific heat • Look at lab sections • M&M and Results • Learn about heating curve What is specific heat? The energy to raise one gram of a substance one degree Celsius. What has a higher specific heat: the ocean or the beach? The OCEAN – it takes a lot of energy to warm it up! What has a higher specific heat: aluminum foil or the glass pan? The GLASS PAN – holds the heat for a much longer period of time Define energy. The ability to do work! When a reaction releases energy in the form of heat, it is said to be… EXOTHERMIC This means it’s negative! When a reaction absorbs energy in the form of heat, it is said to be… ENDOTHERMIC This means it’s positive. What is the formula for finding heat? q = m*s*DT What are the different units used to measure energy? Joule and Calorie Related? 1 cal = 4.184 J What happens if there is a phase change????? This is where the heating curve comes in… Heat Calculations Date: T E M P E R A T U R E E N E R G Y T E M P CONDENSE E 100OC VAPORIZE R A T 0OC FREEZE MELT U R E E N E R G Y T E M P CONDENSE E 100OC VAPORIZE R A T 0OC FREEZE MELT U ENDOTHERMIC g R EXOTHERMIC f E E N E R G Y T E M q = m*DHvap P DHvap = 540cal/g CONDENSE E 100OC R A T 0O C U q = m*DHfusion VAPORIZE DHfusion = 80 cal/g FREEZE MELT ENDOTHERMIC g R EXOTHERMIC f E E N E R G Y Solving Problems Read question and determine number of “lines” involved. Calculate the amount of heat required for each “line” Determine the sum of all “lines” used. IF ENERGY IS RELEASED, you MUST make DHfus and DHvap negative!!!! Calculate the amount of heat necessary to raise 20g H2O from -10oC to 45oC Determine lines: -10oC = solid, 45oC = liquid so 3 lines used. Solid: q = m*sice*DT q = (20g)(0.5cal/goC)(0oC – (-10oC)) = Melt: q = m*DHfus q = (20g)(80cal/g) = Liquid: q = m*sliquid*DT q = (20g)(1cal/goC)(45oC – 0oC) = TOTAL AMOUNT OF HEAT = Calculate the amount of heat released when 30g H2O are cooled from 115oC to 65oC Determine lines: 65oC = liquid, 115oC = gas so 3 lines used. Gas: q = m*sgas*DT q = (30g)(0.4cal/goC)(100oC – 115oC) = Condensed: q = m*DHfus q = (30g)(-540cal/g) = Liquid: q = m*sliquid*DT q = (30g)(1cal/goC)(65oC – 100oC) = TOTAL AMOUNT OF RELEASED HEAT = Suppose the heat needed to melt 38.0g of ice is absorbed from water contained in a glass. If this water has a mass of 210. g and an initial temperature of 21.0oC, what is the final temperature of the water? Remember the ice melts and then warms up as a liquid. Check homework Lab M&M: – how was calorimeter constructed – when were temperature and mass readings made – not what but when – don’t list supplies – does it make sense – can you picture the lab like a movie Results: Charts – Data used for calculations – Table 1 – Temperature over time GRAPH!!!!! Graph 1 – Show a sample calculation (q = msDT) Metal sample Mass of metal Volume of water Mass of water T I metal (Boiling water) T I water/calorimeter T f system DT metal DT water/calorimeter s water q water q metal s metal Homework Heat Calculations worksheet Make corrections to MM and Results – Put data into spreadsheet from wiki Work on conclusions