the wave-mechanical model
advertisement

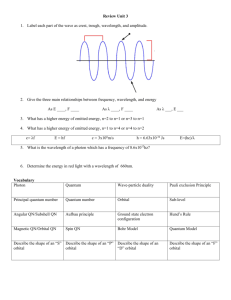
THE WAVE-MECHANICAL MODEL In the 1920's, a more mathematical model of the atom was developed through the work of such men as deBroglie, Heisenberg, and Schroedinger. Their work in quantum mechanics described a probability picture of where the electrons at each energy level might best be found. This region of greatest probability is sometimes referred to as an electron cloud. Unlike the previous model, the wave-mechanical model does not attempt to explain the path an electron travels. DeBroglie attributed wave properties to the electron, and these were confirmed later by experimentation. Quantum Numbers Each electron orbital of the atom may be described by a set of four quantum numbers in this model. They give the position with respect to the nucleus, the shape of the orbital, its spatial orientation, and the spin of the electron in the orbital. Principal quantum number 1, 2, 3, 4, 5, etc. Refers to average distance of the orbital from the nucleus. 1 is closest to the nucleus and has the least energy. These numbers correspond to the shells in the previous model. They are called energy levels. Secondary or azimuthal quantum number s, p, d, f (in order of energy) Refers to the shape of the orbital. The number of possible shapes is limited by the principal quantum number. The first energy level has only one possible shape, the s orbital. The second has two possible, the s and p orbitals. . Magnetic quantum number s = 1 space-oriented orbital p = 3 space-oriented orbitals d = 5 space-oriented orbitals f = 7 space-oriented orbitals Spin quantum number + spin -spin The s-orbital shape, is a sphere. The p orbitals have dumbbell shapes with three possible orientations. The number of spatial orientations of orbitals is referred to as the magnetic quantum number. Electrons are assigned one more quantum number, called the spin quantum number. This describes the spin in either of two possible directions. Each orbital can be filled by only two electrons, each with an opposite spin. This is referred to as the Pauli Exclusion Principle. Therefore each orbital can hold only two electrons. It is important to remember that, when there is more than one orbital at a particular energy level, such as three p orbitals or five d orbitals, only one electron will fill each orbital until each has one electron. After this, pairing will occur with the addition of one more electron to each orbital. This is called Hund's Rule of Maximum