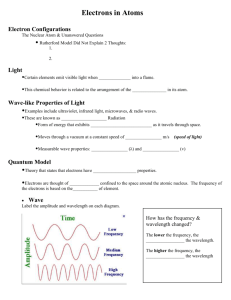
Review Unit 3 Label each part of the wave as crest, trough
advertisement

Review Unit 3 1. Label each part of the wave as crest, trough, wavelength, and amplitude. 2. Give the three main relationships between frequency, wavelength, and energy As λ ____, F ____ As E ____, F ____ As λ ___, E ___ 3. What has a higher energy of emitted energy, n=2 to n=1 or n=3 to n=1 4. What has a higher energy of emitted energy, n=1 to n=4 or n=4 to n=2 c= λf E = hf c = 3x108m/s h = 6.63x10-34 Js E=(hc)/λ 5. What is the wavelength of a photon which has a frequency of 8.6x1012hz? 6. Determine the energy in red light with a wavelength of 660nm. Vocabulary Photon Quantum Wave-particle duality Pauli exclusion Principle Principal quantum number Quantum number Orbital Sub-level Angular QN/Subshell QN Aufbau principle Ground state electron configuration Hund’s Rule Magnetic QN/Orbital QN Spin QN Bohr Model Quantum Model Describe the shape of an “S” Describe the shape of an “P” Describe the shape of an orbital orbital “D” orbital Describe the shape of an “F” orbital Use a Periodic Table to answer the following questions. 1. State the four quantum numbers and the possible values they may have. 2. Name the sublevels described by the following quantum numbers a. n = 2, L = 0 b. n = 3, L = 1 c. n = 6, L = 2 d. n = 5, L = 0 3. Give the n and L values for the following orbitals a. 1s b. 3f c. 2p d. 4d e. 5f 4. Place the following sublevels in order of increasing energy: 1s, 3s, 4s, 6s, 3d, 4f, 3p, 7s, 5d, 5p 5. What and the possible mL values for the following types of orbitals? a. s b. p c. d d. f 5. How many possible orbitals are there for n = a. 6 b. 2 6. How many electrons are in n= a. 3 b. 17 7. Give the longhand electron configuration for the following elements: a. Rh b. Gd c. P d. Cs e. Yb 8. Give the shorthand electron configuration for the following elements: a. Kr b. Ca c. Zn d. As e. Ce Identify the element whose highest energy electron would have the following four quantum numbers: 9. 3, 1, -1, +1/2___________________________________________________ 10. 4, 2, +1, +1/2___________________________________________________ 11. 6, 1, 0, -1/2____________________________________________________ 12. 4, 3, +3, -1/2___________________________________________________ 13. 2, 1, +1, -1/2___________________________________________________ Which of the following represents a permissible set of quantum numbers? (answer “yes” if permissible and “no” if no permissible) 14. 15. 16. 17. 18. 2, 2, +1, -1/2___________________________________________________ 5, 1, 0, +1/2____________________________________________________ 6, 3,-2, +1/2____________________________________________________ 7, 0, 0, -1/2____________________________________________________ 4, 1, 8, +1/2____________________________________________________ 19. Fill the boxes below with the arrow notation for electrons showing the correct ground state electron configuration for the element manganese. Give the four quantum numbers for the following elements 20. Ru 21. H 22. Pd 23. Ho 24. The 15th electron in Pb 25. The 5th electron in oxygen 26. The 45th electron in uranium How many orbitals are in these sublevels? L=0 L= 1 L=2 L=3 How many electrons are in the following subshells/orbitals? 4th principle energy level The px orbital 2nd energy level The d-1 orbital What type of orbital do the following indicate? N= 3 L=0 mL =0 N= 2 L=1 mL =1 N= 1 L=0 mL =0 N= 5 L=3 mL =-2 Review from the Past 1. Convert 103 nm to km 2. Convert 13.43 mL to L 3. Convert 195 cg to µg 4. Draw and label the following atomic models: Dalton’s Model Rutherford’s Model J.J. Thomson’s Model Bohr’s Model Quantum Model 5. How many protons, neutrons, and electrons in the following elements? a. He b. S c. Pt d. Ho 6. Using proper significant figures solve the following problems: a. A piece of metal has a mass of 25.2g and a density of 13.14 g/mL. What is its volume? (D = m/v) b. Determine the energy in orange light with a wavelength of 560nm. (E = (hc)/λ) c. A liquid has a volume of 0.152 L and a density of 2.01 g/mL. What is its mass?