Heat Lecture Notes
advertisement

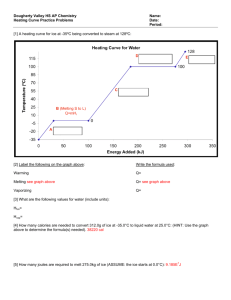
Heat Lecture Notes (Chapter 13 and 17 of Prentice Hall Chemistry) What Is Heat? Heat is a form of ENERGY Remember that ENERGY is the ability to cause change or do WORK (we will talk more about work later on in Physics) Temperature is just a measure of the AVERAGE kinetic energy of a sample. (that’s why all liquids evaporate (have a vapor pressure); some molecules will have enough energy to leave the system as a gas) (Now is a good time to look at the diagram on page 388 of the prentice hall chemistry book) The units of heat are calories or Joules ; or kcal or kJ (we still sometimes use B.T.U.’s here in the USA) One calorie is the energy necessary to change one gram of water one degree Celsius 1 calorie = 4.184 joules Heat capacity is different for different things. Larger things have more heat capacity than smaller pieces of the same stuff. Water is a material that can hold a lot of heat, it has a very large heat capacity. The specific heat of water is 1 calorie per gram oC The heat Equation is: (page 508 and 509 of text) Q = m C T Where: Q = the amount of heat m = the mass of the material C = the specific heat of the material T = the change in temperature of the material So…. The amount of heat is equal to the mass times the specific heat times the change in temperature For a change of phase (state). The heat equation is: Q = m Hfus Where: or Q = m Hvap Q = the amount of heat m = the mass of the material Hfus = the heat of fusion of the material Hvap = the heat of vaporization of the material You need to know these constants for water Hfus = the heat of fusion of water is 80 calories/gram or 6.01 kJ/mol Hvap = the heat of vaporization of water is 540 calories/gram or 40.7 kJ/mol You will need to know (or look up). C = the specific heat of the material Cwater = 4.18 J/g K or 1.00 cal. /g K This one MUST be memorized Cice = 2.1 J/g K or 0.50 cal. /g K Csteam = 1.7 J/g K or 0.40 cal. /g K Specific Heat Practice problems (w/data chart on front) Remember on the front answers are given (check your answers) Problem # 7 and #8 on the back are multi stepped ad you will need more calculating space than is given: #7 you will need to use the equations (heat ice, melt ice, heat water, boil water, heat steam) Q = mCT three times (ice, water and steam) see above Q = mH once for the ice/water change, Hfus = 80 calories/gram and Q = mH once for the water/vapor change; Hvap = 540 calories/gram Specific Heat Capacities Table J/kg oC or J/kg K cal/g oC or cal/g K Water (0 oC to 100 oC) 4186 1.000 Methyl Alcohol 2549 0.609 Ice (below 0 oC) 2093 0.500 Steam (above 100 oC) 2009 0.480 Benzene 1750 0.418 Wood (typical) 1674 0.400 Air ( @ 50 oC) 1046 0.250 Aluminum 900 0.215 Marble 858 0.205 Glass (typical) 837 0.200 Iron/Steel 452 0.108 Copper 387 0.0924 Silver 236 0.0564 Mercury 138 0.0330 Gold 130 0.0310 Lead 128 0.0305 Zinc 388 0.0926 Substance Thermodynamics: (Chapter 17 of text) Enthalpy (H) is kind of like heat at constant pressure. By definition: H = q + w (enthalpy equals heat and work, often heat expands a gas which drives a piston in an engine) Some reactions are endothermic and can be written like: Energy + CO2 + H20 C6H12O11 And would have a negative change in enthalpy H = - Some are exothermic C6H12O11 Energy + CO2 + H20 And would have a positive change in enthalpy H = +