File
advertisement

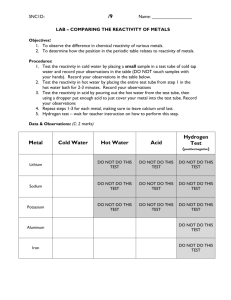
Unit 3 Periodic Table Trends: Reactivity Lab You will use the website below to complete your lab report and submit to the dropbox. You will put answer to the questions in the comments section of the dropbox. See the end of the report for details. http://www.syngentaperiodictable.co.uk/reaction-zone.php This site will open up other browsers to view the videos. IF your computer won’t allow you to view the video, the reactions are also in written form. You are given the website and the questions that need answered. It is up to you to determine HOW you will get those questions answered! I am not going to give you a list of steps to follow. I am going to give you the website you can use and some guideline questions. I would suggest creating tables to organize the data you have collected. You will need to submit this lab report to the dropbox with all of your data. This is like your lab notebook. It will contain your observations and raw data. You will answer the questions in the comments section using this data you have collected to support your answers. Part A. Reactivity Trends in Group 1 Goal A: Determine the trend in reactivity of Group 1 metals. Why does this trend occur? What evidence did you find to support this conclusion? (PT below just for reference) Video with cesium and water doesn’t work. Here is one to watch: http://www.youtube.com/watch?v=DUIorZ_2htk Part B. Reactivity Trends in Group 7: Halogens Goal B: Determine the trend in reactivity of Halogens. Why does this trend occur? What evidence did you find to support this conclusion? Fluorine with Aluminum Reaction Video: starting at 1:25 http://youtu.be/rDpM9_G3Giw?t=1m25s Part C. Reactivity Series of Metals Goal C: Determine the reactivity of Mg, Al, Zn Fe and Cu. List in order from most reactive to least reactive. What evidence did you find to support this conclusion? You need to submit your entire lab report with all of your collected data to the dropbox. In the comments section, you need to answer the following questions: 20pts Goal A: Describe the trend of reactivity for group 1? Is this group metals or non metals? Why does this trend occur? What evidence did you find to support this conclusion? 20pts Goal B: Describe the trend of reactivity for halogens? Is this group metals or non metals? Why does this trend occur? What evidence did you find to support this conclusion? 20pts Goal C: Determine the reactivity of Mg, Al, Zn Fe and Cu. List in order from most reactive to least reactive. What evidence did you find to support this conclusion? Q’s? Post them on the Padlet on the homepage!