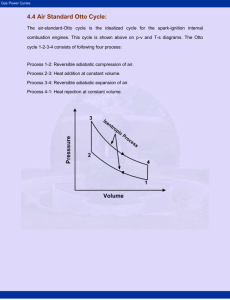
Air Standard Cycles
advertisement

Lecture-7 Prepared under QIP-CD Cell Project Internal Combustion Engines Ujjwal K Saha, Ph.D. Department of Mechanical Engineering Indian Institute of Technology Guwahati 1 Air Standard Cycles 1. 2. 3. 4. Carnot Otto Diesel Brayton - maximum cycle efficiency - spark-ignition (SI) engine - compression-ignition (CI) engine - gas turbine 2 Air Standard Cycles • Air standard cycles are idealized cycles based on the following approximations: – the working fluid is air (ideal gas) – all the processes are internally reversible – the combustion process is replaced by heat input from an external source – heat rejection is used to restore fluid to initial state 3 Thermodynamic Cycles • Air-standard analysis is used to perform elementary analyses of IC engine cycles. • Simplifications to the real cycle include: 1) Fixed amount of air (ideal gas) for working fluid 2) Combustion process not considered 3) Intake and exhaust processes not considered 4) Engine friction and heat losses not considered 5) Specific heats independent of temperature 4 SI Engine Cycle vs Thermodynamic Otto Cycle FUEL A Ignition I R Fuel/Air Mixture Combustion Products Actual Cycle Intake Stroke Compression Stroke Power Stroke Qin Otto Cycle Air Exhaust Stroke Qout TC BC Compression Process Const volume heat addition Process Expansion Process Const volume heat rejection Process 5 Air-Standard Otto cycle Process 1Æ 2 Process 2 Æ 3 Process 3 Æ 4 Process 4 Æ 1 Isentropic compression Constant volume heat addition Isentropic expansion Constant volume heat rejection Compression ratio: r= v1 v4 = v2 v3 Qin Qout v2 TC v1 BC TC BC 6 In Otto cycle, the combustion is so rapid that the piston does not move during the process, and therefore, combustion is assumed to take place at constant volume. Otto cycle efficiency T4 − T1 T1(T4 / T1 − 1) wnet qout η= =1− =1− =1− T2 (T3 / T2 − 1) T3 − T2 qin qin 7 Otto Cycle (Contd.) For isentropic process: pvk = constant with k=cp/cv For process 1-2: p1 v1k = p2 v2k RT2 T2 v1 v1k p2 v2 = = = T1 v 2 v k2 p1 RT1 v1 v1k v 2 T2 = k v 2 v1 T1 T2 v = T1 v k −1 1 k −1 2 ⎛v ⎞ = ⎜⎜ 1 ⎟⎟ ⎝ v2 ⎠ k −1 8 Since m = constant: k −1 T2 ⎛ v1 ⎞ =⎜ ⎟ T1 ⎝ v2 ⎠ k −1 ⎛ V1 ⎞ =⎜ ⎟ ⎝ V2 ⎠ k −1 ⎛ VBDC ⎞ = ⎜⎜ ⎟⎟ ⎝ VTDC ⎠ = r k −1 For process 3-4, using the same analysis: T3 ⎛ V4 ⎞ = ⎜⎜ ⎟⎟ T4 ⎝ V3 ⎠ Then k −1 ⎛V ⎞ = ⎜⎜ BDC ⎟⎟ ⎝ VTDC ⎠ T2 T3 = T1 T4 η =1− k −1 or = r k −1 T3 T4 = T1 T2 1 r k −1 9 Increasing Compression Ratio Increases the Efficiency Typical Compression Ratios for Gasoline Engines 10 Higher Compression Ratios? • Higher compression ratio leads to auto-ignition (without spark) • Causes knock • Engine damage • Thus, there is an upper limit of high compression ratio 11 CI Engine Cycle and the Thermodynamic Diesel Cycle Fuel injected at TC A I R Combustion Products Air Actual Cycle Intake Stroke Compression Stroke Power Stroke Qin Diesel Cycle Exhaust Stroke Qout Air BC Compression Process Const pressure heat addition Process Expansion Process Const volume heat rejection Process 12 Air-Standard Diesel cycle Process 1Æ 2 Process 2 Æ 3 Process 3 Æ 4 Process 4 Æ 1 Isentropic compression Constant pressure heat addition Isentropic expansion Constant volume heat rejection Cut-off ratio: Qin rc = v3 v2 Qout v2 TC v1 BC TC BC 13 Due to ignition delay and finite time required for fuel injection, combustion process continues till the beginning of power stroke. This keeps the cylinder pressure at peak levels for a longer period. Therefore, the combustion process can be approximated as constant pressure heat addition. Remaining processes are similar to that of Otto cycle. • Cycle efficiency, wnet qout η= =1− qin qin 14 V3 Cutoff Ratio, rc = V2 V1 Compression Ratio, r = V2 V4 Expansion Ratio, re = V3 Cutoff Ratio Χ Expansion Ratio = Compression Ratio 15 assuming constant specific heats: η =1− c v (T4 − T1 ) (T4 − T1 ) T (T4 / T1 − 1) =1− =1− 1 cp (T3 − T2 ) k(T3 − T2 ) T2 k(T3 / T2 − 1) for isentropic process 1-2: T1 ⎛ v 2 ⎞ =⎜ ⎟ T2 ⎝ v1 ⎠ k −1 for constant pressure process 2-3: p2 = p3 ideal gas law: RT2 RT3 = v2 v3 => T3 v3 = = rc T2 v2 16 for isentropic process 3-4: T3 ⎛ v 4 ⎞ = ⎜⎜ ⎟⎟ T4 ⎝ v 3 ⎠ => k −1 ⎛ v1 ⎞ = ⎜⎜ ⎟⎟ ⎝ v3 ⎠ T ⎛v ⎞ T4 == 3 ⎜⎜ 3 ⎟⎟ T1 T2 ⎝ v 2 ⎠ then, η = 1 − sin ce k −1 k −1 1 r k −1 = v1k −1 = k −1 v3 v3 v2 T2 k −1 v2 k −1 T2 ⎛ v 2 ⎞ T1 ⎜⎜ ⎟⎟ = = k −1 v3 T1 ⎝ v 3 ⎠ ⎛ v3 ⎞ ⎜⎜ ⎟⎟ ⎝ v2 ⎠ k −1 k ⎛v ⎞ = ⎜⎜ 3 ⎟⎟ = rck ⎝ v2 ⎠ rck − 1 k(rc − 1) rck − 1 ≥ 1, for given r k(rc − 1) ηdiesel ≤ ηOtto but diesel cycle has higher r! 17 Thermal Efficiency η Diesel Recall, k ⎡ r 1 1 ( c − 1) ⎤ ⎥ = 1 − k −1 ⎢ ⋅ r ⎢ k ( rc − 1) ⎥ ⎣ ⎦ ηOtto = 1 − 1 r k −1 Note that the term in the square bracket is always larger than one so for the same compression ratio (r), the Diesel cycle has a lower thermal efficiency than the Otto cycle. Note: CI needs higher r compared to SI to ignite fuel 18 Remark When rc (= v3/v2)Æ1 the Diesel cycle efficiency approaches the efficiency of the Otto cycle Compression ratio = 10-22 (Diesel) Compression ratio = 6-10 (Otto) Thus, efficiency of Diesel Cycle is greater than Otto Cycle. Higher efficiency and low cost fuel makes diesel engine suitable for larger power units such as larger ships, heavy trucks, power generating units, locomotives etc. 19 Diesel Cycle Otto Cycle The only difference is in process 2-3 20 Remark Both Otto cycle (Constant volume heat addition) and Diesel cycle (Constant pressure heat addition) are over-simplistic and unrealistic. In actual case, combustion takes place neither at constant volume (time required for chemical reactions), nor at constant pressure (rapid uncontrolled combustion). Dual cycle is used to model the combustion process. It is a compromise between Otto and Diesel cycles, where heat addition takes place partly at constant volume and partly at constant pressure. This cycle is also known as mixed cycle. In fact, Otto and Diesel cycles are special cases of Dual cycle. 21 Modern CI Engine Cycle and the Thermodynamic Dual Cycle Fuel injected at 15o bTC A I R Air Combustion Products Actual Cycle Intake Stroke Compression Stroke Power Stroke Qin Dual Cycle Air Exhaust Stroke Qin Qout TC BC Compression Process Const volume heat addition Process Const pressure heat addition Process Expansion Process Const volume heat rejection 22 Process Dual Cycle Process 1 Æ 2 Isentropic compression Process 2 Æ 2.5 Constant volume heat addition Process 2.5 Æ 3 Constant pressure heat addition Process 3 Æ 4 Isentropic expansion Process 4 Æ 1 Constant volume heat rejection 2.5 Qin 3 3 2 Qin 4 2.5 4 2 1 1 Qout 23 Thermal Efficiency η Dual = 1 − cycle Qout m u4 − u1 = 1− Qin m (u2.5 − u2 ) + (h3 − h2.5 ) η Dual const cv ⎤ αrck − 1 1 ⎡ = 1 − k −1 ⎢ r ⎣ (α − 1) + αk (rc − 1)⎥⎦ where rc = v3 v2.5 and α = P2.5 P2 Note, the Otto cycle (rc=1) and the Diesel cycle (α=1) are special cases: ηOtto = 1 − 1 r k −1 η Diesel const cV ( ( ) ) 1 ⎡ 1 rck − 1 ⎤ = 1 − k −1 ⎢ ⋅ ⎥ r ⎣ k rc − 1 ⎦ 24 The use of the Dual cycle requires information about either: i) the fractions of constant volume and constant pressure heat addition (common assumption is to equally split the heat addition), or ii) maximum pressure P3. For the same inlet conditions P1, V1 and the same compression ratio: ηOtto > η Dual > η Diesel For the same inlet conditions P1, V1 and the same peak pressure P3 (actual design limitation in engines): η Diesel > η Dual > ηotto 25 For the same inlet conditions P1, V1 and the same compression ratio P2/P1: For the same inlet conditions P1, V1 and the same peak pressure P3: Pressure, P Pmax Pressure, P “x” →“2.5” Po Po Specific Volume Specific Volume Entropy Temperature, T Temperature, T tto O al Du sel Die Tmax el Dies al Du to Ot Entropy 26 References Crouse WH, and Anglin DL, DL (1985), Automotive Engines, Tata McGraw Hill. 2. Eastop TD, and McConkey A, (1993), Applied Thermodynamics for Engg. Technologists, Addison Wisley. 3. Fergusan CR, and Kirkpatrick AT, (2001), Internal Combustion Engines, John Wiley & Sons. 4. Ganesan V, (2003), Internal Combustion Engines, Tata McGraw Hill. 5. Gill PW, Smith JH, and Ziurys EJ, (1959), Fundamentals of I. C. Engines, Oxford and IBH Pub Ltd. 6. Heisler H, (1999), Vehicle and Engine Technology, Arnold Publishers. 7. Heywood JB, (1989), Internal Combustion Engine Fundamentals, McGraw Hill. 8. Heywood JB, and Sher E, (1999), The Two-Stroke Cycle Engine, Taylor & Francis. 9. Joel R, (1996), Basic Engineering Thermodynamics, Addison-Wesley. 10. Mathur ML, and Sharma RP, (1994), A Course in Internal Combustion Engines, Dhanpat Rai & Sons, New Delhi. 11. Pulkrabek WW, (1997), Engineering Fundamentals of the I. C. Engine, Prentice Hall. 12. Rogers GFC, and Mayhew YR, YR (1992), Engineering Thermodynamics, Addison 1. Wisley. 13. Srinivasan S, (2001), Automotive Engines, Tata McGraw Hill. 14. Stone R, (1992), Internal Combustion Engines, The Macmillan Press Limited, London. 15. Taylor CF, (1985), The Internal-Combustion Engine in Theory and Practice, Vol.1 & 2, The MIT Press, Cambridge, Massachusetts. 27 Web Resources 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. http://www.mne.psu.edu/simpson/courses http://me.queensu.ca/courses http://www.eng.fsu.edu http://www.personal.utulsa.edu http://www.glenroseffa.org/ http://www.howstuffworks.com http://www.me.psu.edu http://www.uic.edu/classes/me/ me429/lecture-air-cyc-web%5B1%5D.ppt http://www.osti.gov/fcvt/HETE2004/Stable.pdf http://www.rmi.org/sitepages/pid457.php http://www.tpub.com/content/engine/14081/css http://webpages.csus.edu http://www.nebo.edu/misc/learning_resources/ ppt/6-12 http://netlogo.modelingcomplexity.org/Small_engines.ppt http://www.ku.edu/~kunrotc/academics/180/Lesson%2008%20Diesel.ppt http://navsci.berkeley.edu/NS10/PPT/ http://www.career-center.org/ secondary/powerpoint/sge-parts.ppt http://mcdetflw.tecom.usmc.mil http://ferl.becta.org.uk/display.cfm http://www.eng.fsu.edu/ME_senior_design/2002/folder14/ccd/Combustion http://www.me.udel.edu http://online.physics.uiuc.edu/courses/phys140 http://widget.ecn.purdue.edu/~yanchen/ME200/ME200-8.ppt 28