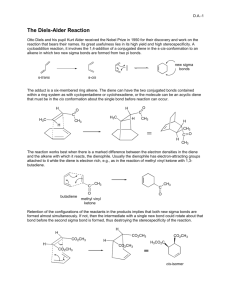
(1S, 3S)-3-methylcyclohexanol (1R, 3S)-1-bromo
advertisement

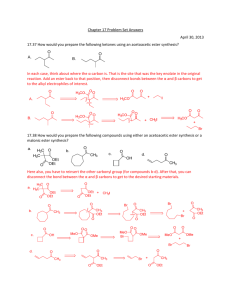
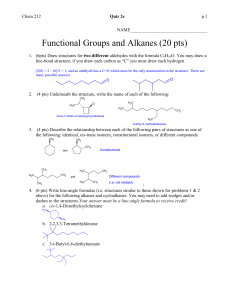
key Name ___________________ Chem 331 Exam #2 Friday, October 19, 2007 This is an open-book, open notes exam. Please show your work in detail. 1. (20 points) Draw the structures of B and C, and give IUPAC names for A, B, and C. You do not have to show mechanisms, but you do need to show stereochemistry clearly. PBr3 OH B Mg; C7H13Br CH3CN C C9H16O A (1S, 3S)-3-methylcyclohexanol Br O (1R, 3S)-1-bromo-3-methylcyclohexane 1-((1R, 3S)-3-methylcyclohexyl)ethanone 2. (20 points) Indicate the expected major product. Explain your reasoning in detail. O Br 1. ONa O O 2. KOH Br O HO O benzylic Br more reactive O O O O Br O O O O O inverted Br same O inverted 3. (20 points) Outline the synthesis steps to convert D into E. In addition to D, you may use any piece that contributes three or fewer carbons to the final product. HO D CrO3 O TsCl, pyridine MgCl MCPBA TsO CH3MgBr O OH 4. (20 points) Deduce the stucture of F, and draw an arrow-pushing mechanism for the transformation. H+ O H+ 13C C9H10O H O CH3 OH H CH3 1H NMR 14.6, q 53.0, d 127.5, d 128.3, d (2) 129.0, d (2) 137.8, s 200.8, d F H3C NMR 1.42, d, J = 7.3 Hz, 3H 3.63, dq, J = 4.2, 7.3 Hz, 1H 7.22, dd, J = 8.1, 2.4 Hz, 2H 7.31, dd, J = 8.1, 8.2 Hz, 2H 7.38, td, J = 8.2, 2.4 Hz, 1 H 9.65, d, J = 4.2 Hz, 1H H3C H O H H O F 5. (20 points) Draw a detailed arrow-pushing mechanism for the following transformation. 5/20 points for correctly showing the mapping of the starting material onto the product. b a c CO2CH3 + d S e f O f O O eO f O DMSO CO2CH3 O S O CO2CH3 O CO2CH3 O O O S S O CO2CH3 H+ O O CO2CH3 O O transfer S S O d CO2CH3 S O O O O bb bf O-e a-d f-H f-S a-f e-f O-f d-H c + S O b a