Lecture Notes Chemistry 342-2008 Mukund P. Sibi Lecture 31
advertisement

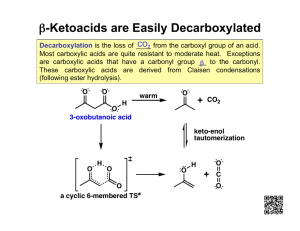
Lecture Notes Chemistry 342-2008 Lecture 31 Mukund P. Sibi Reactions at the Alpha-Carbon of Carbonyl Compounds Enolates are nucleophilic and undergo reaction with electrophiles. For example, one can do halogenation under basic conditions. Even a weak base is sufficient for the reaction. As soon as the enolate is formed, it reacts immediately with the electrophile forming the product. It is not necessary to form the enolate completely. O C O NaOH H O Br2 C H2O Br C + Br- carbon nucleophile It is difficult to control halogenation of ketones under basic conditions. The product halo compound is more reactive than the starting material. Thus one obtains products with multiple halogen atoms. The final product in this reaction is a carboxylic acid and a trihalomethane. The reaction is called the haloform reaction. One can use chlorine, bromine and iodine as the halogen. The Haloform Reaction O O NaOH H C H H O Br2 C H2O O Br H C H Br Br C Br O HO Br Br C Br OH O O H + O Br Br C Br Br + H C Br Bromoform Br O Enolate Alkylation: Formation of C-C bonds One of the important modes of reactivity for the enolate is the formation of C-C bonds by alkylation. The enolate nucleophile can react with alkyl halides in an SN2 reaction. Here the alkyl halides function as electrophiles. Enolate Alkylation O C Enolate Ion + C X SN2 O C C + X- Alkylating Agent Since this is an SN2 reaction, there are only a few alkylating agents that can be used. They need to be primary, allylic or benzylic. Reactions with secondary halides are not Lecture Notes Chemistry 342-2008 Lecture 31 Mukund P. Sibi efficient and tertiary halides do not undergo SN2 substitution. Tosylates, iodides, bromides, and chlorides can be used as a leaving group. Relative reactivity of alkylating agents X R X R Tosylate > I > Br > Cl allylic ~ benzylic > CH3 > RCH2- Malonic Ester Synthesis The hydrogens on alpha carbon of carbonyl compounds are only modestly acidic and require a strong base for complete deprotonation. On the other hand, the methylene groups of 1,3-dicarbonyl compounds have pKa from 9-13 and they can be deprotonated readily using simple inorganic bases. Note from our previous discussion, methylenes flanked by two carbonyl groups are quite acidic because the negative charge is highly delocalized. Malonic esters, acetoacetic esters, and malononitriles can be used as the nucleophile. Malonic esters (pKa = 13) can be alkylated readily using sodium alkoxide as a base. The monoalkylated malonic ester can be further alkylated with a second electrophile to provide dialkylated products. Monoalkylation of malonic esters H3CO O C O C C H H OCH3 NaOCH3 CH3OH Dimethylmalonate H3CO O C C O C Na+ OCH3 R X H O O R C C C OCH3 H3CO H Alkylated malonic ester Sodio malonic ester Dialkylation of malonic esters O O R C C H3CO C OCH3 H NaOCH3 CH3OH H3CO O C C O C Na+ OCH3 R Sodio malonic ester R1 X O O R C C C OCH3 H3CO R1 Dialkylated malonic ester The malonic esters can be hydrolyzed readily under aqueous acidic conditions. The esters are hydrolyzed to the corresponding acids. After this, the diacid undergoes decarboxylation to furnish a monoacid. O O R C C C OCH3 H3CO H O O R C C C OCH3 H3CO R1 H3O+ heat O O R C C HO C OH H H3O+ O O R C C HO C OH heat R1 O H C C OH R H + CO2 + 2 CH3OH O H C C OH R 1 R + CO2 + 2 CH3OH Lecture Notes Chemistry 342-2008 Lecture 31 Mukund P. Sibi The decarboxylation occurs only with malonic and acetoacetic acids. These compounds are β-keto acids. The decarboxylation occurs through a cyclic mechanism. For decarboxylation to occur it is essential that there be a carbonyl group beta to the carboxylic acid. H O O ! " C C O C OH H R H O O ! " C C 1 O C R H R malonic acid !-Keto acid Decarboxylation of malonic esters H O C H3CO C H O C R OCH3 O+ H3 heat O O C H Decarboxylation of !-Keto esters O C H3CO H C O C H3O+ heat R1 R C O O C OH -CO2 OH C R C OH H -CO2 OH R C 1 C R H R O C R C OH H H Carboxylic acid H O C C H O C R1 R R C H H O C R1 Ketone By applying the malonic ester reaction, one can convert an alkyl halide to a carboxylic acid. Overall the process adds two extra carbon atoms to the alkyl halides. This reaction provides a convenient method for the preparation of carboxylic acids from alkyl halides. H CO2CH3 C H CO2CH3 NaOCH3 + CH3Br H H 3C C CO2CH3 H H3O+ H3C CO2CH3 C H 3C CO2CH3 Br + H CO2H 3 carbon acid 1 carbon halide H C NaOCH3 CO2CH3 C H 3C CO2CH3 H3O+ C H 3C CO2CH3 Br H H C CO2CH3 NaOCH3 + CO2CH3 C CO2CH3 CO2CH3 Br H3O+ C H CO2H H CO2H Lecture Notes Chemistry 342-2008 Lecture 31 Mukund P. Sibi Acetoacetic Ester Synthesis Acetoacetic ester (a β-keto ester) undergoes alkylation similar to malonates. The alkylated product can be hydrolyzed to furnish a methyl ketone. Alkylation of Acetoacetic ester H3CO O C C H O C H CH3 NaOCH3 R-X H3CO O C C H O C R CH3 H3O+ heat !-Keto ester R C H H O C CH3 Methyl Ketone Similar to malonic esters, acetoacetic esters can also be dialkylated. The product β-keto esters can also be hydrolyzed under acidic conditions to produce methyl ketones. As noted before, alkylation of ketones requires a strong base and anhydrous conditions. In contrast, the acetoacetic ester method allows for a clever way of enhancing the acidity of alpha C-H bonds in ketones and the ability to alkylate under simple conditions. Once, the product is formed, simple hydrolysis gives the alkylated ketones. One important characteristic of β-ketoester synthesis is its generality. Any β-keto ester with an alpha CH bond can be used. Over all it is a three step process involving (1) enolate formation (2) alkylation and (3) hydrolysis and decarboxylation. Alkylation of Acetoacetic ester H3CO O C C O C H H O H O CH3 NaOCH3 CH3CH2Br H3CO O C H O NaOCH3 OCH3 C O C CH3 CH2CH3 O H 3O + heat H3O+ OCH3 O H3CH2C C C CH3 H H O heat Br Direct Alkylation of Ketones, Esters, and Nitriles As illustrated earlier, one can generate an enolate completely by the use of a strong nonnucleophilic base. For example, LDA will deprotonate cyclohexanone readily. LDA is a strong enough base that it can deprotonate the alpha C-H bonds of esters, lactones, and nitriles. The resulting enolates can be alkylated with alkylating agents. Aldehydes are too reactive and the enolate will react with the starting aldehyde very rapidly. The direct alkylation has some advantage over the acetoacetic ester reaction in that it takes only one Lecture Notes Chemistry 342-2008 Lecture 31 Mukund P. Sibi step. Controlling regiochemistry is an issue with ketones. In the cyclohexanone example the major product is the 2,6-disubstituted cyclohexanone. Ketone O O O H LDA CH3 CH3I THF O O H CH3 Br CH3 O CH3 LDA O THF O Br CH3 CH3 Ester H H O O O OCH3 LDA I OCH3 H H THF Lactone O H H O O LDA O O Br H O THF Nitrile H C N LDA H THF C N H OCH3 I C N