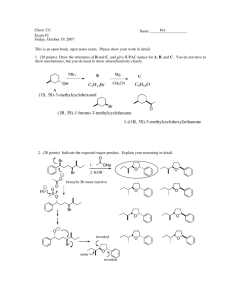
Chemistry 125: Lecture 56
February 25, 2011
Generalized Aromaticity
Cycloaddition – Diels-Alder
Electrocyclic Stereochemistry
Dewar Benzene
This
For copyright
notice see final
page of this file
Generalization of
Aromaticity:
4n+2 Stability
Transition State “Aromaticity”
Cycloadditions &
Electrocyclic Reactions
e.g. J&F Sec. 13.6 pp. 582-595
Generalized Aromaticity
H
H
H
H
H
OH-
H
H
pKa 15
H
H
cyclo-C7H8
R
H
H
6 electrons (4n+2)
cyclo-C7H7- pKa 39 (despite more resonance structures)
e.g. J&F Sec. 13.6 p. 591
8 electrons (4n, antiaromatic)
R
H
+
R
vs. 16 for H2O
R
Ph3C+
unusually
stable cation
(triply benzylic)
Same for cyclo-C7H8 + Ph3C+
(cycloheptatrienyl or “tropylium”)
+
+
R
Ph3CH
R
even more stable
2 electrons (4n+2)
cyclo-C7H7+ e.g. J&F Sec. 13.6pp. 587, 592
6 electrons (4n+2)
Pericyclic Reactions
(in which transition states are “aromatic”)
Cycloadditions: Diels-Alder
(e.g. J&F Sec. 12.12, 14.3)
Electrocyclic Reactions
Cycloadditions: Diels-Alder
diene
ene
Ring
4 + 2 electrons
4 + 2 electrons
How does become ?
H
H
HH
HH
E
H
trans
Z
HOMO
H
H
Z
LUMO
Approach parallel
to p-orbital axes.
H
folded
transition
state
flattened
product
H
cis
H
Cycloadditions: Diels-Alder
Regiochemistry
Note: DieneHis
over C=O
H as
well as C=C
Perhaps Steric
Hindrance?
CH3
H
H
H
H
CO2CHH33C
9% yield
H
Perhaps an
allylic+ / enolateintermediate
20°C
H
H
H
CO2CH3
CO2CH3
CO2CH3
?
H
CH3
H
H
H
H3C
45% yield
stabilized by terminal CH3
or unsymmetrical Transition State?
Cycloadditions: Diels-Alder
Stereochemistry (Ene)
Diene just “sits down” on Ene
forming two -bonds simultaneously from the same face.
No rotatable intermediate with only one new bond
H
CO2CH
3
150-160°C
CO2CH3
H
68% yield
H
H
CO2CH3
CO2CH3
cis alkene
CH3O2C
cis cyclohexene
H
H
150-160°C
CO2CH3
H
H
CO2CH3
trans alkene
84% yield
CO2CH3
trans cyclohexene
e.g. J&F Sec. 12.12, p. 549
Cycloadditions: Diels-Alder
Stereochemistry (Diene)
maleic anhydride
CH2OH
H
CH2OH
O
H
H
O
H
H
O
H
5 min
O
120°C
O
CH3
(2E,4E)-2,4-hexadien-1-ol
H
O
Prefers
H
s-trans
H3C
15 hr
conformation,
O
150°C
which is not
H
H
H
reactive.
CHO3
CH3
(2E,4Z)-2,4-hexadiene
H
81% yield
H
O
CH3
H
CH
H 3
H
all cis
O
O
H
CH3
one trans
H
O
Diels-Alder Variety
CH3
CH3
CO2CH3
C
CO2CH3
150°C
C
CO2CH3
CO2CH3
20°C
k ~1 M-1s-1
NC
CN
NC
CN
CN
CN
160°C
O
O
O
H
H
O
O
H
H
propenal (“acrolein”)
O
H
H
e.g. J&F Sec. 14.3, pp. 628-630
p. 1351
Transition
State
Motion
HOMO
LUMO
Transition
State
HOMO-1
HOMO
HOMO
LUMO
front view
Diels-Alder Reaction
cyclic electron
transition state
side view
Transition
State
Motion
front view
Diels-Alder Reaction
cyclic electron
transition state
side view
?
HOMO () orthogonal to LUMO (*)
h
Shift electron from HOMO to LUMO
e.g. J&F p. 1046
chain
chain
H
DNA Double Helix
N
O
H
N
N
O
N
H
H
O
CH3CH3
O
h (UVB)
T-T
T-T
Thymine photodimerization causes
a chain kink that inhibits DNA replication & transcription and is
A-T-T-G
believed to T-A-A-C
be the main source of mutation / melanomas.
Pericyclic Reactions
(in which transition states are “aromatic”)
Cycloadditions: Diels-Alder
Electrocyclic Reactions
David Benbennick
node
Möbius
Preserves
Axis
top
touches
bottom
(odd # of nodes)
requires twist
in 1 of 2 ways
Transition
Preserves
State
Motion
Mirror
top
touches
top
Hückel
conrotation disrotation
(even #
of nodes)
6
5
4
3
2
1
4
3
!
2
1
6
5
Track the MOs
of hexatriene
4
they transform
into those
3
of cyclohexadiene:
2
1
as
123
Möbius
Aromatic Analogue (Hückel Connectivity)
Preserves
Axis
Möbius
Hückel
conrotation disrotation
6
5
4
3
2
1
4
3
2
1
Preserves
Mirror
Hückel
How to study whether Conrotation
is preferred for 4n-electron shift?
The transition state favored in going from A to B, must also be
favored in going from B to A. (“Microscopic Reversibility”)
DH
CH3
CH3
+11 kcal/mole
CH3
••
CH3
(less stable isomer)
DH
-16 kcal/mole
CH3
CH3
CH3
CH3
(forms the less stable isomer)
Disrotation preferred for 6-electron shift
(4n+2)
4-electron
cycloaddition!
(less stable isomer)
CON
4e
DIS
6e
280°C
CH3 H3C
CH3
CH3
CH3
CH3
CH3
99.9%
~0.005%
Bias >11 kcal/mole
DIS for 4n+2
CH3
CH3
CH3
(forms the less stable isomer)
CON for 4n
CON
8e
-10°C
CH3
CH3
CH3
CH3
e.g. J&F Sec. 27.2 pp. 1343-1346
If you could run
it forwards!
2
1
4e Möbius
conrotation
Transition State HOMO -1
bottom touches top
(odd # of nodes)
6e Hückel
disrotation
top touches top
(even # of nodes)
Opening Dewar Benzene
Strained
Really wants
to open up
Stable
(1866)
Calculated
Isomers of
Benzene
(2004)
84 are calculated to be < 100 kcal above benzene.
6 > 100 kcal above benzene have been prepared.
(single bond breaking gives even less stable species)
Dewar Benzene (1963) is 74 kcal above benzene
but lasts 2 days at room temperature!
4-electron
disrotation!
van Tamelen & Pappas (1963)
CCC angles require
disrotatory
motion
66 kcal/mole more exothermic,
but only 8 kcal/mole “faster”?
t1/2 = 2 days (room temp)
25
33
more strain
*
LUMO
HOMO
-11 kcal
*
LUMO
HOMO
conrotatory
good for 4n electrons
-75 kcal
aromatic
But shouldn’t “aromatic” 6--electron
transition state be good for disrotation?
It is more fundamental that LUMO
doesn’t overlap HOMOs (& vice versa).
Spectroscopy for
Structure and Dynamics
Electronic (Visible/UV)
e.g. F&J sec. 12.7-12.8 pp. 533
Vibrational (Infrared)
e.g. F&J sec. 15.4, pp. 707-713
NMR (Radio)
e.g. F&J sec. 15.5-15.9, pp. 713-749
O.E.D.
“Specters or straunge Sights, Visions and Apparitions” (1605)
“Sunbeams..passing through a Glass Prism to the opposite
Wall, exhibited there a Spectrum of divers colours”
Newton (1674)
End of Lecture 56
February 25, 2011
Copyright © J. M. McBride 2011. Some rights reserved. Except for cited third-party materials, and those used by visiting
speakers, all content is licensed under a Creative Commons License (Attribution-NonCommercial-ShareAlike 3.0).
Use of this content constitutes your acceptance of the noted license and the terms and conditions of use.
Materials from Wikimedia Commons are denoted by the symbol
.
Third party materials may be subject to additional intellectual property notices, information, or restrictions.
The following attribution may be used when reusing material that is not identified as third-party content:
J. M. McBride, Chem 125. License: Creative Commons BY-NC-SA 3.0