Chemistry Form 4: Chapter 6
advertisement

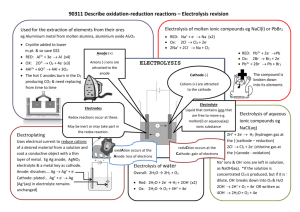
Chemistry Form 4: Chapter 6 - Electrolyte Electrolyte is a chemical substance which conducts electricity in the molten or aqueous state. Electrolytes are able to conduct electricity because there are freely move ions in the molten or aqueous state. Examples of electrolyte are salt solution, dilute acids and dilute alkalis Chemistry Form 4: Chapter 6 - Electrolysis of Lead (II) Bromide Electrolysis is a chemical process where a substance in its molten state or in an aqueous solution decomposed by electric current. This involves conversion of energy from electrical energy to chemical energy. Electrodes are the strips of metals inserted into electrolyte for conduction of electricity. he metal electrode connected to the positive terminal of the battery is called the anode (+). The metal electrode connected to the negative terminal of the battery is called the cathode (-). Electrolytic cell is the complete set-up for electrolysis. This consists of the vessel containing the electrolyte, anode, cathode, battery and wires. At the Cathode Observation When electricity is flowing, a silvery deposit of lead metal forms on the cathode. In fact, as it is molten, it is more likely to drip off in a molten blob. At the Anode Observation When electricity is flowing, brown fumes of bromine gas are seen at the anode. Half equation Pb2+ + 2e ---> Pb Half equation 2Br- ---> Br2 + e Explanation Explanation The lead(II) ions, as they are positive, move to the negative cathode, where each ion gains two electrons to form a lead atom. Any reaction at a cathode involved a gain in electrons. This is called reduction or more exactly, cathodic reduction . The bromide ions, as they are negative, move to the positive anode, where each loses an electron to form a bromine atom. Then two of these newly formed atoms combine to form bromine gas. Any reaction at an anode involves a loss of electrons. Chemistry Form 4: Chapter 6 - Electrolysis of Concentrated of Sodium Chloride Solution Electrolysis of dilute sodium chloride solution At anode : Anions : OH- and Cl- Electrolysis of concentrated sodium chloride solution At anode : Anions : OH- and Cl- OH- ions are selectively discharge according to the position of ions in the Electrochemical Series, ES. Cl- ions are discharged instead of OH- because of their higher concentration in the electrolyte. Gas bubbles are formed which lighted up a glowing wooden splinter. This gas is oxygen. A greenish-yellow gas with a pungent and choking smell is released. The gas turns the blue litmus paper red and then white. 4OH- ---> 2 H2O + O2 + 4e 2Cl- ---> Cl2 + 2e At cathode: Cations : H+ dan Na+ At cathode: Cations : H+ dan K+ H+ ions are selectively discharge according to the position of ions in the ES. H+ ions are selectively discharge according to the position of ions in the ES. Gas bubbles are formed. When a lighted wooden splinter is placed near the mouth of the test tube, a ‘pop’ sound is produced. Hydrogen gas is produced Gas bubbles are formed. When a lighted wooden splinter is placed near the mouth of the test tube, a ‘pop’ sound is produced. Hydrogen gas is produced 2H+ + 2e ---> H2 2H+ + 2e ---> H2 Chemistry Form 4: Chapter 6 - Types of Electrodes Affect Product of Electrolysis Carbon dan platinum electrodes are inert because both of these materials do not react with the electrolytes or the product of electrolysis. Other electrodes except carbon and platinum can react with the electrolytes. Chemistry Form Four : Chapter 6 - Electrolysis of copper (II) sulphate Electrolysis of copper (II) sulphate using carbon electrode. The bulb is lighted indicates that copper (II) sulphate is an electrolyte which can conduct electricity. Carbon electrode is inert which does not react with electrolyte or product of electrolysis. At anode : Hydroxide ions ions are selectively discharged. Gas bubbles are formed which lighted up a glowing wooden splinter. This gas is oxygen. At cathode : Copper ions are selectively discharged. Shiny brown metal deposited around cathode. This metal is copper. Electrolysis copper (II) sulphate solution with copper electrodes At cathode : Copper ions are selectively discharged. Shiny brown metal deposited, makes the cathode thicker. This metal is copper. At anode : Copper electrode eroded and became thinner. Copper anode dissolved to form copper ions. Chemistry Form Four: Chapter 6 - Electroplating of copper metal 1) Anode --> plating metal = copper 2) Cathode --> object to be electroplated = iron key 3) Electrolyte --> salt solution that contains electroplating metal ions = copper (II) sulphate cathode: A brown metal deposited on the surface of the iron key. anode: Copper anode becomes thinner. Chemistry Form 4: Chapter 6 - Purification of Metal The anode is a impure copper. The cathode is a pure copper. When electricity is passed through the cell, the copper is dissolved at anode where Cu2+ ions go into solution. Anode copper becomes thinner. At cathode, copper ions are selectively discharge and deposited. Cathode copper becomes thicker. The blue intensity of solution remains unchanged because concentration of copper (II) ions in the solution remains unchanged. The rate at which copper enter into electrolyte from anode is equal to the rate at which the copper ions discharged at cathode. Chemistry Form 4: Chapter 6 - Extraction of Aluminium From its Ore The ores of aluminium is called bauxite. Bauxite is a composition of aluminium oxide, (Al2O3). The position of aluminium in the reactivity series indicates that, it could not be extracted by reducing the oxide using carbon or carbon monoxide. Electrolysis of molten aluminium oxide ("alumina") will, however, give aluminium and oxygen. The function of cryolite is to lower the melting point of aluminium oxide. Aluminium metal forms at the cathode electrode and sinks to the bottom of the tank, where it is tapped off. Oxygen forms at the anode electrodes. This oxygen reacts with the carbon of the anode electrodes, forming carbon dioxide, and they gradually burn away. Consequently, the anode electrodes have to be replaced frequently. Chemistry Form 4: Chapter 6 - Daniell Cell Daniell cell is also known as Galvanic cell which using same working principle as voltaic cell. It converts chemical energy to electrical energy. Daniell cell is a part of a battery consisting of two metals joined by a salt bridge or a porous pot. More electropositive metal acts as negative terminal which undergoes oxidation process (release electron) Less electropositive metal acts as positive terminal which undergoes reduction (gain electron). Electron flow from negative terminal to positive terminal. The function of salt bridge or porous pot is to allow the movement of ions to pass through it to complete electrical circuit. Another function is to separate two electrolyte solutions. Examples of salt bridge are sodium chloride or potassium nitrate. At Negative Terminal: More electropositive metal ( Zinc ) Observation: Zinc plate dissolves or becomes thinner, its mass decreases Half chemical equation : Zn ------> Zn2+ + 2e At Positive Terminal: Less electropositive metal ( Copper ) Observation: Copper plate becomes thicker Half chemical equation : Cu2+ + 2e Cu Chemistry Form Four: Chapter 6 - Displacement of Metal from Salt Solution Electrochemical series can be constructed by using displacement reaction of metal from salt solution. A more electropositive (reactive) metal can displace a less electropositive metallic ion (less reactive) from its salt solution. For example: Reaction between magnesium and copper (II) sulphate produces copper and magnesium sulphate. Chemistry Form 4: Chapter 6 - Constructing Electrochemical Series based on the Potential Difference between Two Metals In a voltaic cell, two different metals are used to create a potential difference. A higher metal in the electrochemical series will be made negative terminal. A lower metal in the electrochemical series will be made positive terminal. Conversion of energy is chemical energy to electrical energy. Metals which are further apart in the electrochemical series produce a higher voltage than metals which are close to each other. By arranging the metals in order of their voltage readings, you can build up the electrochemical series.