HW2_f2007
advertisement

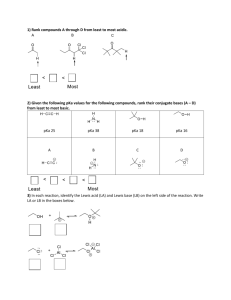
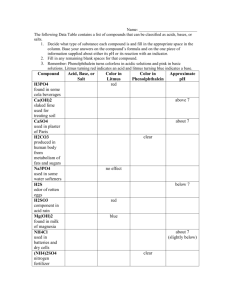
CHM 251 Organic Chemistry 1 NAME _________________________ Fall 2007 Homework #2 Due: Friday, October 19, 2007 by 5:00 pm Print out this assignment and complete all of your work on these pages. Question #1 Rank the following alkenes in order of MOST to LEAST exothermic upon catalytic hydrogenation of each to its corresponding alkane (this reaction is described in Section 10.2). H2 Alkene Alkane + HEAT Pd-C Provide SPARTAN molecular modeling data to help complete the reaction energy diagram template. Provide a brief explanation that describes why the reactions are ordered as observed. HINT: you’ll likely want to draw the structure of each before you proceed. (a) cis-2-hexene (b) 1-hexene (c) 2,3-dimethyl-2-butene (d) 2-methyl-2-pentene (e) trans-2-hexene energy relative enegy of product (a) (b) (c) (d) (e) Question #2 The following sets of question pertain to intermolecular forces: (a) Provide a valid structure for each of the following molecules and then, arrange the compounds in order of increasing boiling point (you may want to consult the Aldrich catalog, an MSDS, or go to chemfinder.com to find the bps). Briefly explain your ordering being sure to indicate the type and relative magnitude of intermolecular interactions (e.g. VDW interactions, dipole-dipole, and hydrogen bonding) that exist in each case. 1. 2. 3. 4. 5. 6. 2,3-dimethyl-2-butanol 2-amino-1,3-propanediol n-hexane ethyl 3-oxobutanoate (a.k.a. ethylacetoacetate) dichloroethanoic acid (or if you prefer, dichloroacetic acid) methyl butyl ether (b) Predict which of the following salts is likely to have unusually high solubility in a (nonaqueous) hydrocarbon solvent. Briefly explain your choice. Br CH 2CH 2CH2CH 3 N H3C(H 2C)15 vs. CH 2CH 2CH2CH 3 NH4 Cl CH 2CH 2CH2CH 3 (c) Normally, the drug shown below is much more soluble in lipid-like solvents (e.g. benzene) than it is in water. However, its water solubility INCREASES considerably when the aqueous solution contains moderate amounts of strong acid (e.g. in the stomach) as shown by the reaction below. Explain the solubility differences. H H O + soluble in benzene O O H H + H2O Question #3 Consider the reaction shown below. O H N HO + NaCN + Cl (a) Name the reactant on the left. (b) Circle the most acidic hydrogen in the reactant on the left. (c) Provide a brief explanation as to your choice of most acidic hydrogen in part (b) in terms of the major factors contributing to its acidity (e.g. electronegativity effect, resonance effect, etc) (d) The other compound is sodium cyanide. What type of compound is sodium cyanide? What is the role of CN? What is the role of Na? Type of compound = Role of CN = Role of Na = (e) Realizing that the reactant and sodium cyanide will react with one another in an acid-base reaction, use the curved-arrow formalism to show the flow of electrons as the reaction proceeds from reactants to products (forward direction). You may have to redraw NaCN. (f) Which species is acting as your Bronsted-Lowry acid? Which species is acting as your Bronsted-Lowry base? B.L. acid = B.L. base = (g) Which species is your nucleophile (Lewis base) and which is your electrophile (Lewis acid)? Nucleophile (Lewis base) = Electrophile (Lewis acid) = (h) Which species will utilize its HOMO and which will utilize its LUMO in the reaction? Utilizes HOMO = Utilizes LUMO = (i) Draw the structures for the expected products of this reaction based upon the mechanism you derived in part (d) – place in the boxes provided (above). (j) Report the appropriate pKa values for both acids in your equilibrium. (k) Designate each of the species in your reaction as a strong acid (SA), strong base (SB), weak acid (WA), or weak base (WB) – label directly on the reaction above. (l) Show the conjugate acid-base pairs – show directly on the reaction above. (m) For the major organic product (after hydrogen removal), draw all important resonance structures to show how resonance contributes significantly to stabilizing this anion. (n) Calculate the net pKa (from the individual pKa’s) (o) Convert your net pKa to an overall Ka. (p) Use the Ka from part (o) to find G°rxn at room temperature (26 °C). (q) Is this a product or reactant favored equilibrium? Explain. Question #4 Consider the following compounds: A H O H H B H O N H OH Cl SH Cl D C O E CH3 O H F HN H CH3 Br G CH3 H H H I HO OH H H O N O a. For compounds B, H, and I, some formal charges are missing. Fill in missing lone electron pairs on heteroatoms and find the missing formal charges (you need to calculate them!!) b. Circle the most acidic hydrogen atom (if it’s not shown, please draw it in before circling). c. Report an estimated pKa value for each circled hydrogen (SAM is a good place to look). d. For each compound, indicate the major effect(s) that contribute to the acidity of the circled hydrogen (that is, enhance the stability of the conjugate base anion) – formal charge effect, electronegativity effect, inductive effect, and resonance effect. Explain/show the occurrence of the effect(s) in each compound. e. Name compounds A, D, E, G, H, and I. A= D= E= G= H= I= f. Which compound will exhibit the greatest resonance effect after deprotonation of the acidic hydrogen atom? Draw all important resonance structures for this compound. g. Arrange the compounds above in order of INCREASING stability of the conjugate base anion by filling in the chart below. Rank according to pKa values that you determined. If there are roughly equivalent compounds draw in an equal sign to indicate this fact. Least stable Most stable