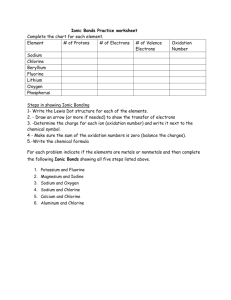
What is that symbol

What is that symbol?
Well, it is that time of the semester when you get to start memorizing the symbols and the names of the elements. Get ready, here we go:
H hydrogen (exists in nature as H
2
)
He helium
Li lithium
Be beryllium
B
C
N boron carbon nitrogen (exists in nature as N
2
)
O oxygen (exists in nature as O
2
)
F fluorine ( you’ll FLunk if you write Fl. And it is fluorine, not flourine. We’re not baking cookies.)
(exists in nature as F
2
)
Ne neon
Na sodium
Mg magnesium
Al aluminum
Si silicon
P phosphorus (P is for phosphorus, not potassium)
S sulfur (S is for sulfur, not sodium)
Cl chlorine (make sure you can recognize Cl as chlorine. Students will often confuse chlorine and call it carbon iodine. Cl is NOT carbon iodine) (exists in nature as Cl
2
)
Ar argon
K potassium
Ca calcium
Zn zinc
Pb lead
Fe iron
Cu copper
Au gold
Ag silver
Mn manganese
Ni nickel
Co cobalt
Br bromine (exists in nature as Br
2
)
I iodine (exists in nature as I
2
)
Fr francium
Hg mercury
Rn radon
Ra radium
Sn tin
Study hint: make flash cards. Put the symbol on one side of the card and the word on the other side of the card. Use sheets of paper if index cards are not available. You will be responsible for knowing these names and symbols on all tests. You might receive another list later, but for now this will do.