The Professional Thermographers Association
Welcome
All of us at The Professional Thermographers
Association would like to welcome you to this
course. We greatly appreciate you taking the time
to be here. This course is designed to accomplish
two primary goals:
Welcome
Welcome
The
TheProfessional
ProfessionalThermographers
ThermographersAssociation
Association
1. Provide you with a world class education experience for you; where the focus is placed on your
continued development as a professional thermographer.
2. This course will help you to learn how to conduct infrared inspections and manage your program.
You will leave with tools that you can put right to work on the job, next week; these tools will help
you to get an even greater return on your investment that you are currently getting.
We will have a lot to do in the next few days. How should you approach this course? There is no single
right way. If we may make a few suggestions:
First make sure to enjoy yourself. We are all working hard to make sure that learning about
Infrared Thermography is an exciting process.
Participate. In this course, please share your knowledge with others. So we can all learn from
your experiences as well.
Please feel free to ask questions. This is the place to ask.
Make mistakes. This is the place to try new things, to break new ground, to ask stupid questions.
Better here than back on the job where you may get into real trouble! This a safe place to learn. If
you aren't making mistakes, then you are not trying hard enough.
All of us at The Professional Thermographers Association, and your fellow thermographers wish to thank
you for taking the time out of your schedules to be here, improve your knowledge, skill sets and help to
improve the industry that we all work so hard to uphold. We look forward to working with you during the
next few days.
Sincerely,
Fred Colbert Director
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 1
The Professional Thermographers Association
Student Bio
Please take a moment to compose your self a brief bio on yourself. You will be asked to introduce yourself
to the class and speak about:
Your Company:
________________________________________________________________________
Your position in your company:
_______________________________________________________________________
What does your job require you to do…
_______________________________________________________________________
What type of IR camera do you have?
_______________________________________________________________________
What type of computer software for report generation and image analysis do you have?
_______________________________________________________________________
The reason that your company purchased / or is in the process of purchasing an IR camera.
_______________________________________________________________________
What applications will you be performing?
________________________________________________________________________
What has been your past experience with infrared?
_________________________________________________________________________
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 2
The Professional Thermographers Association
What would you like to get out of this course?
Please take a moment to outline some of the goals that you want to achieve by taking this course. List
what it is that you would like for us to cover, and please rate them in order of importance to you.
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
Rate
____
Goal
______________________________________________________________________
____________________________________________________________________________
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 3
The Professional Thermographers Association
I know what Infrared is...Exam!
A lot of people have been exposed to “Infrared Thermal Imaging” from the media today. TV being the major
pipeline of their perspective as to what this technology is all about. From the Discovery Channel, to NEWS
to Hollywood movies, it is almost impossible to find someone that does not think that they know something
about infrared imaging. Infrared camera manufacturers help this along with “point and shoot” features that
promise to do everything and more, if there is a problem in the world then their must be a solution to it,
using a infrared camera. From UAV (unmanned aerial vehicles) for the military to small miniature IR
cameras in the grill of a Cadillac with a heads up display for night vision driving, infrared imaging is here to
stay. But managing perception is not as easy as it may seem.
Take the test below and see what your perceptions are, and at the same time ask your self, “What are the
perceptions of the general public”, as far as what this technology can do?
1. Yes / No - Police use IR cameras to look for Pot growers, they drive through neighborhoods with IR
cameras searching for houses where pot is grown.
2. Yes / No - Police IR cameras can see through walls of your house.
3. Yes / No - The US military has IR cameras in UAV that can see through the walls of a commercial
building, or military compound.
4. Yes / No - The US has spy satellites that use Infrared to see individual people, and can track you
where ever you go.
5. Yes / No - You can see through someone’s clothes with a IR camera.
6. Yes / No - Sony Corporation pulled a model of their video cameras off of the market because they
used infrared and you could use them to see right through woman’s blouses.
7. Yes / No - The US government has Black Helicopters that fly around in “whisper” mode that use IR
cameras that see right through brick walls and closed windows and curtains.
8. Yes / No - US Navy Seals use Night Vision/Thermal on their rifle scopes to see through walls of
buildings.
9. Yes / No - You can see ghosts using thermal imaging.
10. Yes / No - You can see space aliens using IR
11. Yes / No - You can see invisible people using IR.
12. Yes / No – You can see Vampires with IR.
13. Yes / No - In the US Thermography is commonly
used as a way to screen patients for cancer or
other medical problems.
14. Yes / No – You can effectively scan people coming
off of airplanes or ships to screen for SARS.
Next “circle” which group your answers fall in to:
___Group A = No Yeses, ___Group B = 1 Yes,
___Group C = 2 Yeses, ___Group D = 3 to 5 Yeses,
___Group F = 6 or greater.
What group do you thing the general public would fall into? Place a “check” next to this group please.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 4
The Professional Thermographers Association
How has Hollywood help to
educate people about Infrared?
What movies have you seen that used infrared in someway in
their story line? What was the first movie you saw that used
Infrared? What was your emotional response to how they
showed it being used? Today whether it is good or not, we are
influenced tremendously by what Hollywood has shown us, and
thermal imaging has been a large part of it. How did Hollywood
portray the use of IR in the following Movies?
Movie or TV Show
being able to….
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
Blue Thunder
Predator
Navy Seals
Behind Enemy Lines
Patriot Games
The Invisible Man
CSI
TAPS
Others
______________
______________
______________
Hollywood
Hollywood has
has educated
educated us…
us…
You
Youbelieve
believeeverything
everythingyou
yousee
seeininthe
themovies
movies…right?
…right?
IR was portrayed as
____________________________________________________________________________________________________
__________________________________________________
__________________________________________________
__________________________________________________
__________________________________________________
__________________________________________________
__________________________________________________
___________________________________________________
___________________________________________________
___________________________________________________
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 5
The Professional Thermographers Association
Know your camera!
What is the brand of your camera? _______________________________________________________
What is the model of your camera? _______________________________________________________
What is the SS# of your IR camera? ______________________________________________________
What is the SS# of your lens? ___________________________________________________________
How many lenses do you have?__________________________________________________________
What is the temperature measurement range of your camera? From what _________ to what _________?
How many temperature ranges does your camera have? ______________________________________
What are they? Range 1 =_______________Range 2 = ________________ Range 3 =______________
Is your dynamic range of your camera a 8, 12, or 14 bit camera? ________________________________
What is the minimum distance that you can be from a target to your camera lens? __________________
What is the wavelength of your camera? ___________________________________________________
What is the size of your infrared detector? __________________________________________________
What is the FOV of your camera? ________________________________________________________
What is the IFOV of your camera at 10 feet? ________________________________________________
What is the MFOV of your camera at 10 feet? _______________________________________________
What is the depth of field at the cameras minimum focal distance? _______________________________
What type of IR detector is in your camera?_________________________________________________
How long will it take to charge your battery? ________________________________________________
How long will your camera run on a single battery? ___________________________________________
How many batteries do you have? ________________________________________________________
How many hours of operation can you get if you use all of your batteries? _________________________
What does your IR camera store images onto? ______________________________________________
How big is a image file? ________________________________________________________________
What type of IR image file extension is on your IR image? _____________________________________
How many images will your camera store? _________________________________________________
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 6
The Professional Thermographers Association
Your Manual
Your manual has been compiled from a number of sources to provide you with a comprehensive resource
for you to be able to refer back to as you further your skills as a professional thermographer. It includes a
comprehensive collection, topically / subject orientated, that you can use for furthering your study for your
level I, II and III, as well as supporting information that helps to provide a strong foundation for knowledge
base.
The manual is written as to provide you with basic information for your level I skills, and then goes deeper
in the subjects for further study for your level II and III. There are also key points that are highlighted that
are very important for you to understand if you are going to be knowledgeable the specific subjects.
You will notice that there are icons used in this book that point you to important information that will help
you to differentiate what material you will want to know for each certification Level.
This icon points to information that is important to understand for your Level I
certification.
This icon points to information that is important to understand for your Level II
certification.
This icon points to information that is important to understand for your Level III
certification.
This icon warns you of impending danger of not understanding the information /
concepts completely, and that you will be making a big mistake if you don’t pay
attention to the material..
This icon points to helpful hints, key points, and helpful information that you will
want to know about at any certification level. The information that this icon
refers to will most likely not be on your written exam for certification, but will be
of real importance in the actual tasks of performing, and managing your IR
programs.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 7
The Professional Thermographers Association
Table of contents
Welcome ........................................................................................................................................... 1
Student Bio ........................................................................................................................................ 2
What would you like to get out of this course? ....................................................................................... 3
Know your camera! ............................................................................................................................. 6
Your Manual....................................................................................................................................... 7
Table of contents ................................................................................................................................ 8
ASNT Thermal/Infrared Testing Method .............................................................................................. 12
(Training Course Outline TC-10) ........................................................................................................ 12
Recommended Initial Training and Experience Levels ......................................................................... 20
Level I Recommended Training.......................................................................................................... 21
Level II Recommended Training ......................................................................................................... 23
Level III Outline ................................................................................................................................ 26
What you need to do to get your Level I Certificate .............................................................................. 32
Recommended Training References .................................................................................................. 34
Books and Publications ................................................................................................................. 34
The Discovery of Infrared Radiation by William Herschel .................................................................. 37
Introduction to Temperature Measurement .......................................................................................... 41
Selecting an Infrared Camera for your needs ...................................................................................... 50
Thermographic Imaging ................................................................................................................ 52
Thermographic camera ..................................................................................................................... 54
Theory of operation ....................................................................................................................... 54
Types .......................................................................................................................................... 54
Cooled infrared detectors .......................................................................................................... 55
Uncooled infrared detectors ....................................................................................................... 55
Basic Camera Operation ................................................................................................................... 59
Basic Camera Operation ................................................................................................................... 59
Thermodynamics ............................................................................................................................. 64
Overview ..................................................................................................................................... 64
History ......................................................................................................................................... 64
The laws of thermodynamics ......................................................................................................... 65
Conservation of energy ..................................................................................................................... 67
. .............................................................................................................................................. 67
Historical development .................................................................................................................. 67
Modern physics ............................................................................................................................ 69
Mathematical formulations ............................................................................................................. 69
Second law of thermodynamics.......................................................................................................... 71
. .............................................................................................................................................. 71
General description ....................................................................................................................... 71
History ......................................................................................................................................... 72
Heat ................................................................................................................................................ 73
Notation ....................................................................................................................................... 73
Changes of phase ........................................................................................................................ 74
Joule ............................................................................................................................................... 76
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 8
The Professional Thermographers Association
Definition ..................................................................................................................................... 76
Conversions ................................................................................................................................. 76
Multiples ...................................................................................................................................... 77
British thermal unit ............................................................................................................................ 78
Conversions ................................................................................................................................. 78
Associated units ........................................................................................................................... 78
Calorie ............................................................................................................................................. 80
Nutrition ....................................................................................................................................... 80
Versions ...................................................................................................................................... 80
Temperature .................................................................................................................................... 82
Overview ..................................................................................................................................... 82
Applications ............................................................................................................................. 82
Temperature measurement ........................................................................................................... 83
Units of temperature ................................................................................................................. 83
Negative temperatures .............................................................................................................. 84
Articles about temperature ranges: ............................................................................................. 84
Theoretical foundation of temperature ............................................................................................ 84
Zeroth-law definition of temperature ........................................................................................... 84
Temperature in gases ............................................................................................................... 85
Temperature of the vacuum ....................................................................................................... 86
Second-law definition of temperature .......................................................................................... 86
Temperature conversion formulas ...................................................................................................... 89
Summary ..................................................................................................................................... 89
Kelvin .......................................................................................................................................... 89
Celsius ........................................................................................................................................ 89
Fahrenheit ................................................................................................................................... 90
Rankine ....................................................................................................................................... 90
Delisle ......................................................................................................................................... 90
Newton ........................................................................................................................................ 90
Réaumur...................................................................................................................................... 91
Rømer ......................................................................................................................................... 91
Heat transfer mechanisms ............................................................................................................. 92
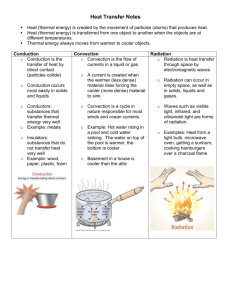
Conduction............................................................................................................................... 92
Convection ............................................................................................................................... 93
Radiation ................................................................................................................................. 93
Heat transfer features ................................................................................................................... 93
Heat dissipation ............................................................................................................................ 94
Preventing heat transfer ................................................................................................................ 94
Specific heat capacity ....................................................................................................................... 95
Factors that influence heat capacity measurements ......................................................................... 95
Table of specific heat capacities ..................................................................................................... 96
Heat capacity ................................................................................................................................... 97
Specific Heat Capacity .................................................................................................................. 97
Thermal capacitance ..................................................................................................................... 97
Gas Phase Heat Capacities ........................................................................................................... 98
Latent heat ..................................................................................................................................... 101
Latent heat ..................................................................................................................................... 101
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 9
The Professional Thermographers Association
. ............................................................................................................................................ 101
Heat of fusion ................................................................................................................................. 102
Reference Values ....................................................................................................................... 102
Application ................................................................................................................................. 103
Heat of vaporization ........................................................................................................................ 103
Heats of vaporization of the elements ........................................................................................... 104
Heat conduction ............................................................................................................................. 106
. ............................................................................................................................................ 106
Conductance .............................................................................................................................. 107
Newton's law of cooling ............................................................................................................... 108
Thermal conductivity ....................................................................................................................... 109
. ............................................................................................................................................ 109
Examples ................................................................................................................................... 109
Related terms ............................................................................................................................. 110
First definition (general)........................................................................................................... 110
Second definition (buildings) .................................................................................................... 110
Molecular origins ........................................................................................................................ 111
Convection ..................................................................................................................................... 112
Free and forced convection ......................................................................................................... 112
Atmospheric convection .............................................................................................................. 113
Oceanic convection .................................................................................................................... 113
Thermal radiation............................................................................................................................ 115
Radiant energy ............................................................................................................................... 115
Analysis ..................................................................................................................................... 115
Open systems ............................................................................................................................ 116
SI radiometry units ...................................................................................................................... 116
Electromagnetic radiation ............................................................................................................ 117
Physics ...................................................................................................................................... 117
Theory ................................................................................................................................... 117
Properties .............................................................................................................................. 117
Wave model ........................................................................................................................... 118
Particle model ........................................................................................................................ 118
Speed of propagation .............................................................................................................. 118
Electromagnetic spectrum ........................................................................................................... 120
Wavelength .................................................................................................................................... 120
Spectra of objects ....................................................................................................................... 123
Classification systems ................................................................................................................. 123
Gamma rays .......................................................................................................................... 124
X-rays .................................................................................................................................... 124
Ultraviolet light........................................................................................................................ 124
Visible radiation Light .............................................................................................................. 124
Infrared radiation .................................................................................................................... 125
Microwaves ............................................................................................................................ 126
Radio frequency ..................................................................................................................... 126
Radio waves .......................................................................................................................... 126
When EM radiation impinges upon a conductor, it couples to the conductor, travels along it, and induces an
electric current on the surface of that conductor by exciting the electrons of the conducting material. This
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 10
The Professional Thermographers Association
effect (the skin effect) is used in antennas. EM radiation may also cause certain molecules to absorb
energy and thus to heat up; this is exploited in microwave ovens. ....................................................... 126
Electric energy ....................................................................................................................... 127
Infrared .......................................................................................................................................... 128
Different regions in the infrared ................................................................................................ 128
The Earth as an infrared emitter ................................................................................................... 129
Applications for Infrared .............................................................................................................. 129
Night Vision............................................................................................................................ 129
Other Imaging ........................................................................................................................ 130
Thermography ........................................................................................................................ 130
Heating .................................................................................................................................. 130
Communications ..................................................................................................................... 130
Spectroscopy ......................................................................................................................... 130
Stefan-Boltzmann law ..................................................................................................................... 132
Derivation of the Stefan-Boltzmann law .................................................................................... 132
Temperature of the Sun .......................................................................................................... 133
Gustav Kirchhoff ............................................................................................................................. 134
Kirchhoff's law of thermal radiation ................................................................................................... 135
. ............................................................................................................................................ 135
Black body ..................................................................................................................................... 137
Details ....................................................................................................................................... 137
Equations governing black bodies ................................................................................................ 139
Planck's law of black-body radiation ......................................................................................... 139
Wien's displacement law ......................................................................................................... 139
Stefan-Boltzmann law ............................................................................................................. 139
Derivation of temperature relation between earth and sun .............................................................. 140
Assumptions .......................................................................................................................... 140
Derivation .............................................................................................................................. 140
The result............................................................................................................................... 141
Temperature of the sun ........................................................................................................... 141
Footnotes................................................................................................................................... 141
Planck's law of black body radiation ................................................................................................. 143
. ........................................................................................................................................... 143
Wien's displacement law ................................................................................................................. 145
Emissivity....................................................................................................................................... 146
Glossary of terms ........................................................................................................................... 150
Appendix: IR The History of Heat / Temperature and Infrared imaging ................................................. 166
Line Array Appendix: Building Heat Loss .......................................................................................... 176
Appendix: Building Heat Loss .......................................................................................................... 177
Heat Load Calculation Worksheet ................................................................................................ 180
Appendix: Home inspections and the Law ......................................................................................... 184
Appendix: 12 STEPS FOR SETTING UP A SUCCESSFUL CONDITION MONITORING PROGRAM .... 188
Appendix: Existing IR Related Standards .......................................................................................... 194
Appendix: Electrical Inspections ....................................................................................................... 203
Standard Guide for Examining Electrical Power Distribution Equipment with Infrared Thermography:
DRAFT as of 9/18/2006 ................................................................................................................... 203
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 11
The Professional Thermographers Association
ASNT Thermal/Infrared Testing Method
(Training Course Outline TC-10)
Recommended
Hours of
Instruction
A*
B*
Level I
Basic physics course
Basic operating course
Basic applications course
Total
12
10
10
32 (Four – 8 hour
days)
10
10
10
30
12
10
12
34 (Four – 8 hour
days + 2 hours)
10
10
12
32
Level II
Intermediate physics course
Intermediate operating course
Intermediate applications course
Total
*A High School graduate or equivalent.
*B Completion with passing grades of at least two years of engineering or science study at a university,
college or technical school.
This is a progressive training course. Consideration as Level I is based on satisfactory completion of the
Level I training course. Consideration as Level II is based on satisfactory completion of both Level I and
Level II training courses.
Topics in the training outline may be deleted or expanded to meet the employer’s specific
applications or for limited certification and may be accompanied by a corresponding change in
training hours.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 12
The Professional Thermographers Association
4.0 Levels of Qualification
4.1 There are three basic levels of qualification. The employer may subdivide these levels for situations where
additional levels are deemed necessary for specific skills and responsibilities.
4.2 While in the process of being initially trained, qualified and certified, an individual should be considered a trainee. A
trainee should work with a certified individual. The trainee shall not independently conduct, interpret, evaluate, or report
the results of any NDT test.
4.3 The three basic levels of qualification are as follow:
4.3.1 NDT Level I. An NDT Level I individual should be qualified to properly perform specific calibrations,
specific NDT, and specific evaluations for acceptance or rejection determinations according to written
instructions and to record results. The NDT Level I should receive the necessary instruction or supervision
from a certified NDT Level II or III individual.
4.3.2 NDT Level II. An NDT Level II individual should be qualified to set up and calibrate equipment and
to interpret and evaluate results with respect to applicable codes, standards and specifications. The
NDT Level II should be thoroughly familiar with the scope and limitations of the methods for which
qualified and should exercise assigned responsibility for on-the-job training and guidance of trainees and
NDT Level I personnel. The NDT Level II should be able to organize and report the results of NDT tests.
4.3.3 NDT Level III. An NDT Level III individual should be capable of developing, qualifying, and
approving procedures, establishing and approving techniques, interpreting codes, standards,
specifications and procedures; and designating the particular NDT methods, techniques and
procedures to be used. The NDT Level III should be responsible for the NDT operations for which qualified
and assigned and should be capable of interpreting and evaluating results in terms of existing codes,
standards and specifications. The NDT Level III should have sufficient practical background in
applicable materials, fabrication and product technology to establish techniques and to assist in
establishing acceptance criteria when none are otherwise available. The NDT Level III should have
“general familiarity” with other appropriate NDT methods, as demonstrated by an ASNT Level III
Basic examination or other means. The NDT Level III, in the methods in which certified, should be capable
of training and examining NDT Level I and II personnel for certification in those methods.
5.0 Written Practice
5.1 The employer shall establish a written practice for the control and administration of NDT personnel training,
examination and certification.
5.2 The employer’s written practice should describe the responsibility of each level of certification for determining
the acceptability of materials or components in accordance with the applicable codes, standards, specifications and
procedures.
5.3 The employer’s written practice shall describe the training, experience and examination requirements for
each level of certification.
5.4 The employer’s written practice shall be reviewed and approved by the employer’s NDT Level III.
5.5 The employer’s written practice shall be maintained on file.
7.0 Training Programs
7.1 Personnel being considered for initial certification should complete sufficient organized training to become
thoroughly familiar with the principles and practices of the specified NDT method related to the level of
certification desired and applicable to the processes to be used and the products to be tested.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 13
The Professional Thermographers Association
7.2 The training program should include sufficient examinations to ensure understanding of the necessary
information.
7.3 Recommended training course outlines and references for NDT Levels I, II and III which may be used as
technical source material are contained in the Recommended Training Courses section of this Recommended
Practice.
7.4 The employer who purchases outside training services is responsible for ensuring that such services meet
the requirements of the employer’s written practice.
8.0 Examinations
8.1 Administration and Grading
8.1.1 An NDT Level III shall be responsible for the administration and grading of examinations specified in
Section 8.3 through 8.8 for NDT Level I, II, or other Level III personnel. The administration and grading of
examinations may be delegated to a qualified representative of the NDT Level III and so recorded. A
qualified representative of the employer may perform the actual administration and grading of Level III
examinations specified in 8.8.
8.1.2 For Level I and II personnel, a composite grade should be determined by simple averaging of the
results of the general, specific and practical examinations described below. For Level III personnel, the
composite grade should be determined by simple averaging of the results of the basic, method and specific
examinations described below.
8.1.3 Examinations administered for qualification should result in a passing composite grade of at least 80
percent, with no individual examination having a passing grade less than 70 percent.
8.1.4 When an examination is administered and graded for the employer by an outside agency and the
outside agency issues grades of pass or fail only, on a certified report, then the employer may accept the
pass grade as 80 percent for that particular examination.
8.1.5 The employer who purchases outside services is responsible for ensuring that the examination services
meet the requirements of the employer’s written practice.
8.2 Vision Examinations
8.2.1 Near-Vision Acuity: The examination should
ensure natural or corrected near distance acuity in
at least one eye such that the applicant is capable of
reading a minimum of Jaeger Number 2 or equivalent
type and size letter at the distance designated on the
chart but not less than 12 inches (30.5 cm) on a
standard Jaeger test chart. The ability to perceive an
Ortho-Rater minimum of 8 or similar test pattern is
also acceptable. This should be administered
annually.
8.2.2 Color Contrast Differentiation: The
examination should demonstrate the capability of
distinguishing and differentiating contrast among
colors or shades of gray used in the method as determined by the employer. This should be conducted
upon initial certification and at three-year intervals thereafter.
8.3 General (written for NDT Levels I and II)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 14
The Professional Thermographers Association
8.3.1 The general examinations should address the basic principles of the applicable method.
8.3.2 In preparing the examinations, the NDT Level III should select or devise appropriate questions covering
the applicable method to the degree required by the employer’s written practice.
8.3.3 See the Appendix for example questions.
8.3.4 The minimum number of questions that should be given is as follows:
Test Method
Number of
Level I
Questions
Number of
Level II
Questions
Thermal/Infrared Testing
40
40
8.4 Specific (written for NDT Levels I and II)
8.4.1 The specific examination should address the equipment, operating procedures and NDT
techniques that the individual may encounter during specific assignments to the degree required by the
employer’s written practice.
8.4.2 The specific examination should also cover the specifications or codes and acceptance criteria
used in the employer’s NDT procedures.
8.4.3 The minimum number of questions that should be given is as follows:
Test Method
Thermal/Infrared Testing
Number of
Level I
Questions
20
Number of
Level II
Questions
20
8.5 Practical (for NDT Level I and II)
8.5.1 The candidate should demonstrate familiarity with and ability to operate the necessary NDT
equipment, record and analyze the resultant information to the degree required.
8.5.2 At least one flawed specimen or component should be tested and the results of the NDT test
analyzed by the candidate.
8.5.3 The description of the specimen, the NDT procedure, including check points, and the results of
the examination should be documented.
8.5.4 NDT Level I Practical Examination. Proficiency should be demonstrated in performing the applicable
NDT on one or more specimens or machine problems approved by the NDT Level III and in evaluating the
results to the degree of responsibility as described in the employer’s written practice. At least ten (10)
different checkpoints requiring an understanding of test variables and the employer’s procedural
requirements should be included in this practical examination.
8.5.5 NDT Level II Practical Examination. Proficiency should be demonstrated in selecting and performing
the applicable NDT technique within the method and interpreting and evaluating the results on one or
more specimens or machine problems approved by the NDT Level III. At least ten (10) different
checkpoints requiring an understanding of NDT variables and the employer’s procedural
requirements should be included in this practical examination.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 15
The Professional Thermographers Association
8.6 Sample questions for general examinations are presented in the separate question booklets that can be obtained
from ASNT Headquarters. These questions are intended as examples only and should not be used verbatim for
qualification examinations. The following is a list of the booklets:
Question
Test Method
Booklets
Thermal/Infrared Testing
J*
* In course of preparation
8.7 Additional Written, Specific and Practical Examination Criteria
8.7.1 Level I, II and III Written Examinations
8.7.1.1 All Level I, II and III written examinations should be closed book except
that necessary data, such as graphs, tables, specifications, procedures, utilizing such reference
materials should require an understanding of the information rather than merely locating the
appropriate answer. All questions used for Level I and Level II examinations should be approved
by the responsible Level III.
8.7.1.2 A valid endorsement on an ACCP Level I or Level II certificate fulfills the corresponding
examination criteria described in paragraphs 8.3 and 8.5 for each applicable NDT method.
8.7.1.3 The employer may delete the specific examination (paragraph 8.4) if the candidate has a
valid ACCP certificate, provided the examinations administered meet the technical needs of the
employer.
8.7.2 Level I and II Specific Examinations
The employer may delete the specific examination if the candidate has a valid ACCP Level I or Level II
certificate in the method and if documented experience exists to permit such.
8.7.3 Practical Examinations
Successful completion of ACCP Level I or Level II general hands-on practical examinations may be
considered as fulfilling the requirements of paragraphs 8.3 and 8.5 respectively.
8.8 NDT/PdM Level III Examinations
8.8.1 Basic Examinations
8.8.1.1 NDT Basic Examination (required only once when more than one method examination is
taken). The minimum number of questions that should be given is as follows:
8.8.1.1.1 Fifteen (15) questions relating to understanding the SNT-TC-1A document.
8.8.1.1.2 Twenty (20) questions relating to applicable materials, fabrication and product
technology.
8.8.1.1.3 Twenty (20) questions that are similar to published Level II questions for other
appropriate NDT methods.
8.8.1.2 PdM Basic Examination (required only once when more than one method examination is taken). The
minimum number of questions that should be given is as follows:
8.8.1.2.1 Fifteen (15) questions relating to understanding the SNT-TC-1A document.
8.8.1.2.2 Twenty (20) questions relating to applicable machinery technology.
8.8.1.2.3 Thirty (30) questions that are similar to published Level II questions for other appropriate
PdM methods.
8.8.2 Method Examination (for each method)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 16
The Professional Thermographers Association
8.8.2.1 Thirty (30) questions relating to fundamentals and principles that are similar to published
ASNT Level III questions for each method, and
8.8.2.2 Fifteen (15) questions relating to application and establishment of techniques and
procedures that are similar to the published ASNT Level III questions for each method, and
8.8.2.3 Twenty (20) questions relating to capability for interpreting codes, standards and
specifications relating to the method.
8.8.3 Specific Examination (for each method)
8.8.3.1 Twenty (20) questions relating to specifications, equipment, techniques and procedures
applicable to the employer’s product(s) and methods employed and to the administration of the
employer’s written practice.
8.8.3.2 The employer may delete the specific examination if the candidate has a valid ASNT Level
III certificate in the method and if documented evidence of experience exists, including the
preparation of procedures to codes, standards, or specifications and the evaluation of test results.
8.8.4 A valid endorsement on an ASNT Level III certificate fulfills the examination criteria described in 8.8.1
and 8.8.2 for each applicable method.
8.8.5 A valid endorsement of an ACCP Professional Level III certificate fulfills the examination criteria
described in 8.8.1 and 8.8.2 for each applicable method.
8.9 Reexamination
Those failing to attain the required grades should wait at least thirty (30) days or receive suitable additional training as
determined by the Level III before reexamination.
9.0 Certification
9.1 Certification of all levels of NDT personnel is the responsibility of the employer.
9.2 Certification of NDT personnel shall be based on demonstration of satisfactory qualification in accordance
with Sections 6, 7 and 8, as described in the employer’s written practice.
9.3 At the option of the employer, an outside agency may be engaged to provide NDT Level III services. In such
instances, the responsibility of certification of the employees shall be retained by the employer.
9.4 Personnel certification records shall be maintained on file by the employer for the duration specified in the
employers written practice and should include the following:
9.4.1 Name of certified individual.
9.4.2 Level of certification and NDT method.
9.4.3 Educational background and experience of certified individuals.
9.4.4 Statement indicating satisfactory completion of training in accordance with the employer’s written
practice.
9.4.5 Results of the vision examinations prescribed in 8.2 for the current certification period.
9.4.6 Current examination copy(ies) or evidence of successful completion of examinations.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 17
The Professional Thermographers Association
9.4.7 Other suitable evidence of satisfactory qualifications when such qualifications are used in lieu of the
specific examination prescribed in 8.8.3.2 or as prescribed in the employer’s written practice.
9.4.8 Composite grade(s) or suitable evidence of grades.
9.4.9 Signature of the Level III that verified qualifications of candidate for certification.
9.4.10 Date of certification and/or recertification and the date of assignments to NDT.
9.4.11 Certification expiration date.
9.4.12 Signature of employer’s certifying authority.
10.0 Technical Performance Evaluation
10.1 NDT personnel may be reexamined any time at the discretion of the employer and have their certificates extended
or revoked.
10.2 Periodically, as defined in the employers written practice, the technical performance of Level I and II personnel
should be evaluated and documented by an NDT Level III. The evaluation and documentation should follow the format
and guidelines described in section 8.5.
11.0 Interrupted Service
11.1 The employer’s written practice should include rules covering the types and duration of interrupted service.
11.2 The written practice should specify the requirements for reexamination and/or recertification for the interrupted
service.
12.0 Recertification
12.1 All levels of NDT personnel shall be recertified periodically in accordance with one of the following criteria:
12.1.1 Evidence of continuing satisfactory technical performance.
12.1.2 Reexamination in those portions of the examinations in Section 8 deemed necessary by the
employer’s Level III.
12.2 Recommended maximum recertification intervals are:
12.2.1 Levels I and II – 3 years
12.2.2 Level III – 5 years
13.0 Termination
13.1 The employer’s certification shall be deemed revoked when employment is terminated.
13.2 A Level I, Level II, or Level III whose certification has been terminated may be certified to the former NDT level by
a new employer based on examination, as described in Section 8, provided all of the following conditions are met to
the new employer’s satisfaction:
13.2.1 The employee has proof of prior certification.
13.2.2 The employee was working in the capacity to which certified within six (6) months of termination.
13.2.3 The employee is being recertified within six (6) months of termination.
13.2.4 Prior to being examined for certification, employees not meeting the above requirements should
receive additional training as deemed appropriate by the Level III.
14.0 Reinstatement
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 18
The Professional Thermographers Association
14.1 A Level I, Level II, or Level III whose certification has been terminated may be reinstated to the former NDT level, without a
new examination, provided all of the following conditions are met:
14.1.1 The employer has maintained the personnel certification records required in
section 9.4.
14.1.2 The employees certification did not expire during termination.
14.1.3 The employee is being reinstated within six (6) months of termination.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 19
The Professional Thermographers Association
Recommended Initial Training and Experience Levels
NDT
Method
Level
Infrared
High School
Graduate or
equivalent
Minimum
Required Work
Experience in
Method (Hours)
Permitted time
frame to obtain
required work
experience in
method ( Months)
32 hours
Completion with a
passing grade of
at least 2 years of
engineering or
science study at a
university college
or technical school
30 hours
210 hours (26.25
Days, or 5.25
Weeks, 1.3
Months)
1.5 to 9 Months
34 hours
32 hours
1,260 hours
(157.5 Days, or
31.5 Weeks, or
7.8 Months)
9 to 27 Month
Level II
Notes:
1.0 For Level II certification, the experience shall consist of time at Level I or equivalent. If a person is
being qualified directly to Level II with no time at Level I, the required experience shall consist of the sum of
the times required for Level I and Level II and the required training shall consist of the sum of the hours
required for Level I and Level II.
2.0 For Level III certification, the required experience shall consist of the sum of the time required for Level
I and Level II, and the additional requirements listed in 6.3.2. The required formal training shall consist of
the Level I and Level II training, plus any additional formal training as defined in the employer’s written
practice.
3.0 Listed training hours may be adjusted as described in the employer’s written practice depending on the
candidate’s actual education level, e.g. grammar school, college graduate in engineering, etc.
4.0 Training shall be outlined in the employer’s written practice.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 20
The Professional Thermographers Association
Level I Recommended Training
Basic Physics Course
1.0
2.0
3.0
4.0
The nature of heat – what is it and how is it measured/expressed?
1.1
Instrumentation
1.2
Scales and conversions
Temperature – what is it and how is it measured/expressed?
2.1 Instrumentation
2.2 Scales and conversions
Heat Transfer Modes Familiarization
3.1
Heat conduction fundamentals
3.1.1 Fourier’s law of heat conduction (concept)
3.1.2 Conductivity/resistance basics
3.2
Heat convection fundamentals
3.2.1 Newton’s law of cooling (concept)
3.2.2 Film coefficient/film resistance basics
3.3
Heat radiation fundamentals
3.3.1 Stefan-Boltzmann law (concept)
3.3.2 Emissivity/absorptivity/reflectivity/transmissivity basics (Kirchhoff’s Law)
Radiosity Concepts Familiarization
4.1
Reflectivity
4.2
Transmissivity
4.3
Absorptivity
4.4
Emissivity
4.5
Infrared radiometry and imaging
4.6
Spatial resolution concepts
4.6.1 Field of view (FOV)
4.6.2 Instantaneous field of view (IFOV) – ref. ASTM E-1149
4.6.3 Spatial resolution for temperature measurement – the Split Response Function
(SRF)
4.6.4 Measurement Instantaneous Field of View (MIFOV)
4.7
Error potential in radiant measurements (an overview)
Basic Operating Course
1.0
Introduction
1.1
Thermography defined
1.2
How infrared imagers work
1.3
Differences among imagers and alternative equipment
1.4
Operation of infrared thermal imager
1.4.1 Selecting the best perspective
1.4.2 Image area and lens selection for required details
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 21
The Professional Thermographers Association
1.5
2.0
3.0
4.0
1.4.3 Optimizing the image
1.4.4 Basic temperature measurement
1.4.5 Basic emissivity measurement
Operation of support equipment for infrared surveys
Checking Equipment Calibration with Blackbody References
Infrared Image and Documentation Quality
3.1
Elements of a good infrared image
3.1.1 Clarity (focus)
3.1.2 Dynamic range of the image
3.1.3 Recognizing and dealing with reflections
3.1.4 Recognizing and dealing with spurious convection
3.2
Recording
3.2.1 Video tape
3.2.2 Photographic images
3.2.3 Video photo cameras
3.2.4 Digital recording
3.2.5 Videoprinters
Support Data Collection
4.1
Environmental data
4.2
Emissivity
4.2.1 Measurement
4.2.2 Estimation
4.2.3 Surface modification
4.3
Surface reference temperatures
4.4
Identification and other
Basic Applications Course
1.0
2.0
3.0
4.0
5.0
6.0
7.0
7.1
7.2
7.3
Detecting Thermal Anomalies Resulting from Differences in Thermal Resistance
(Quasi-steady-state Heat Flow)
1.1
Large surface-to-ambient temperature difference
1.2
Small surface-to-ambient temperature difference
Detecting Thermal
Using System or Environmental Heat Cycles
Detecting Thermal Anomalies Resulting from Differences in Physical State
Detecting Thermal Anomalies Resulting from Fluid Flow Problems
Detecting Thermal Anomalies Resulting from Friction
Detecting Thermal Anomalies Resulting from Nonhomogeneous Exothermic or
Endothermic Conditions (Non-uniform, Heat Releasing or Heat Absorption Conditions).
Field Quantification of Point Temperatures
Simple techniques for emissivity
Typical (high emissivity) applications
Special problem of low emissivity applications
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 22
The Professional Thermographers Association
Level II Recommended Training
Intermediate Physics Course
1.0
2.0
3.0
Basic Calculations in the Three Modes of Heat Transfer
1.1
Conduction – principles and elementary calculation
1.1.1 Thermal resistance – principles and elementary calculations
1.1.2 Heat capacitance – principles and elementary calculations
1.2
Convection – principles and elementary calculations
1.3
Radiation – principles and elementary calculations
The Infrared Spectrum
2.1
Planck’s law/curves
2.1.1 Typical detected bands
2.1.2 Spectral emissivities of real surfaces
2.2
Effects due to semitransparent windows and/or gasses
2.3
Filters
Radiosity Problems
3.1
Blackbodies – theory and concepts
3.2
Emissivity problems
3.2.1 Blackbody emissivity
3.2.2 The graybody and the nongraybody
3.2.3 Broadband and narrowband emitter targets
3.2.4 Specular and diffuse emitters
3.2.5 Lambertian and non-Lambertian emitters (the angular sensitivity of emissivity)
(Definition: Lambertian, A perfectly diffusing surface; the intensity of the light
emanating in a given direction from any small surface component is
proportional to the cosine of the angle of the normal to the surface. The
brightness (luminance, radiance) of a lambertian surface is constant
regardless of the angle from which it is viewed.)
3.2.6 Effects of emissivity errors
Calculation of emissivity, reflectivity and transmissivity (practical use of Kirchhoff’s law)
Reflectivity problem
3.4.1 Quantifying effects of unavoidable reflections
3.4.2 Theoretical corrections
3.5
Transmissivity problem
3.5.1 Quantified effects of partial transmittance
3.5.2 Theoretical corrections
Resolution Tests and Calculations
4.1
IFOV, FOV and MIFOV measurements and calculations
4.2
MRTD measurements and calculations
4.3
Slit response function – measurement, calculations, interpretations and comparisons
4.4
Resolution versus lens and distance
4.5
Dynamic range
3.3
3.4
4.0
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 23
The Professional Thermographers Association
4.6
4.7
4.8
Data acquisition rate/data density
Frame rate and field rate
Image data density
4.8.1 Lines of resolution
4.8.2 IFOVs/line
4.8.3 Computer pixels/line
Intermediate Operating Course
1.0
2.0
3.0
4.0
Operating for Infrared Measurements (Quantification)
1.1
Simple infrared energy measurement
1.2
Quantifying the emissivity of the target surface
1.3
Quantifying temperature profiles
1.3.1 Use of blackbody temperature references in the image
1.3.2 Use of temperature measurement devices for reference surface temperatures
1.3.3 Common sources of temperature measurement errors
1.4
Computer processing to enhance imager data
Operating for High Speed Data Collection
2.1
Producing accurate images of transient processes
2.2
Recording accurate images of transient processes
2.3
Equipment selection and operation for imaging from moving vehicles
Operating Special Equipment for “Active” Techniques
3.1
Hot or cold fluid energy sources
3.2
Heat lamp energy sources
3.3
Flash-lamp energy sources
3.4
Electromagnetic induction
3.5
Laser energy sources
Reports and Documentation
4.1
Calibration requirements and records
4.2
Report data requirements
4.3
Preparing reports
Intermediate Applications Course
1.0
2.0
3.0
Temperature Measurement Applications
1.1
Isotherms/alarm levels – personnel safety audits, etc.
1.2
Profiles
Energy Loss Analysis Applications
2.1
Conduction losses through envelopes
2.1.1 Basic envelope heat-flow quantification
2.1.2 Recognizing and dealing with wind effects
2.2
Mass transfer heat exchange (air or other flows into or out of the system)
2.2.1 Location
2.2.2 Quantification
“Active” Applications
3.1
Insulation flaws
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 24
The Professional Thermographers Association
4.0
5.0
3.2
De-Lamination of composites
3.3
Bond quality of coatings
3.4
Location of high heat-capacity components
Filtered Applications
4.1
Sunlight
4.2
Furnace interiors
4.3
Semitransparent targets
Transient Applications
5.1
Imaging a rapidly moving process
5.2
Imaging from a vehicle
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 25
The Professional Thermographers Association
Level III Outline
1.0 Principles/Theory
1.1 Conduction
1.2 Convection
1.3 Radiation
1.4 The nature of heat and flow
1.4.1 Exothermic or endothermic conditions
1.4.2 Friction
1.4.3 Variations in fluid flow
1.4.4 Variations in thermal resistance
1.4.5 Thermal capacitance
1.5 Temperature measurement principles
1.6 Proper selection of thermal/infrared testing as technique of choice
1.6.1 Differences between thermal/infrared testing and other techniques
1.6.2 Complementary roles of thermal/infrared testing and other methods
1.6.3 Potential for conflicting results between methods
1.6.4 Factors that qualify/disqualify the use of thermal/infrared testing
2.0 Equipment/Materials
2.1 Temperature measurement equipment
2.1.1 Liquid-in-glass thermometers
2.1.2 Vapor – pressure thermometers
2.1.3 Bourdon – tube thermometers
2.1.4 Bimetallic thermometers
2.1.5 Melting point indicators
2.1.6 Thermochromic liquid crystal materials
2.1.7 (Irreversible) Thermochromic change materials
2.1.8 Thermocouples
2.1.9 Resistance thermometers
2.1.9.1 RTDs
2.1.9.2 Thermistors
2.1.10 Optical pyrometers
2.1.11 Infrared pyrometers
2.1.12 Two color infrared pyrometers
2.1.13 Laser/infrared pyrometers
2.1.14 Integrating hemisphere radiation pyrometers
2.1.15 Fiberoptic thermometers
2.1.16 Infrared photographic films and cameras
2.1.17 Infrared line – scanners
2.1.18 Thermal/infrared imagers
2.1.18.1 Pyroelectric vidicons
2.1.18.2 Single detector scanners
2.1.18.3 Multi-detector scanners
2.1.18.4 Staring arrays
2.2 Heat flux indicators
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 26
The Professional Thermographers Association
2.3 Performance parameters of non-contact devices
2.3.1 Absolute precision and accuracy
2.3.2 Repeatability
2.3.3 Sensitivity
2.3.4 Spectral response limits
2.3.5 Response time
2.3.6 Drift
2.3.7 Spot size ratio
2.3.8 Instantaneous field of view
2.3.9 Minimum resolvable temperature difference
2.3.10 Slit response function
3.0
Techniques
3.1
Contact temperature indicators
3.1.1 Calibration
3.2
Noncontact pyrometers
3.2.1 Calibration of equipment
3.2.2 Quantifying emissivity
3.2.3 Evaluating background radiation
3.2.4 Measuring (or mapping) radiant energy
3.2.5 Measuring (or mapping) surface temperatures
3.2.6 Measuring (or mapping) surface heat flows
3.2.7 Use in high temperature environments
3.2.8 Use in high magnetic field environments
3.2.9 Measurements on small targets
3.2.10 Measurements through semitransparent materials
3.3
Infrared line scanners
3.3.1 Calibration of equipment
3.3.2 Quantifying emissivity
3.3.3 Evaluating background radiation
3.3.4 Measuring (or mapping) surface radiant energy
3.3.5 Measuring (or mapping) surface temperatures
3.3.6 Measuring (or mapping) surface heat flows
3.3.7 Use in high temperature environments
3.3.8 Use in high magnetic field environments
3.3.9 Measurements on small targets
3.3.10 Measurements through semitransparent materials
3.4
Thermal/Infrared imaging
3.4.1 Calibration of equipment
3.4.2 Quantifying emissivity
3.4.3 Evaluating background radiation
3.4.4 Measuring (or mapping) surface radiant energy
3.4.5 Measuring (or mapping) surface temperatures
3.4.6 Measuring (or mapping) surface heat flows
3.4.7 Use in high temperature environments
3.4.8 Use in high magnetic field environments
3.4.9 Measurements on small targets
3.4.10 Measurements through semitransparent materials
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 27
The Professional Thermographers Association
3.5
3.6
Heat flux indicators
3.5.1 Calibration of equipment
3.5.2 Measurement of heat flow
Exothermic or endothermic investigations: typical examples may include, but are not
limited to, the following:
3.6.1 Power distribution systems
3.6.1.1 Exposed electrical switchgear
3.6.1.2 Enclosed electrical switchgear
3.6.1.3 Exposed electrical busses
3.6.1.4 Enclosed electrical busses
3.6.1.5 Transformers
3.6.1.6 Electric rotating equipment
3.6.1.7 Overhead power lines
3.6.1.8 Coils
3.6.1.9 Capacitors
3.6.1.10
Circuit breakers
3.6.1.11
Indoor wiring
3.6.1.12
Motor control center starters
3.6.1.13
Lighter arrestors
3.6.2 Chemical processes
3.6.3 Foam-in-place insulation
3.6.4 Fire fighting
3.6.4.1 Building investigations
3.6.4.2 Outside ground base investigations
3.6.4.3 Outside airborne investigations
3.6.5 Moisture in airframes
3.6.6 Underground investigations
3.6.6.1 Airborne coal mine fires
3.6.6.2 Utility locating
3.6.6.3 Utility pipe leak detection
3.6.6.4 Void detection
3.6.7 Locating and mapping utilities concealed in structures
3.6.8 Mammal location and monitoring
3.6.8.1 Ground investigations
3.6.8.2 Airborne investigations
3.6.8.3 Sorting mammals according to stress levels
3.6.9 Fracture dynamics
3.6.10 Process heating or cooling
3.6.10.1
Rate
3.6.10.2
Uniformity
3.6.11 Heat tracing or channelized cooling
3.6.12 Radiant heating
3.6.13 Electronic components
3.6.13.1
Assembled circuit boards
3.6.13.2
Bare printed circuit boards
3.6.13.3
Semiconductor microcircuits
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 28
The Professional Thermographers Association
3.6.14 Welding
3.6.14.1
Welding technique parameters
3.6.14.2
Material parameters
3.6.15 Mapping of energy fields
3.6.15.1
Electromagnetic fields
3.6.15.2
Electromagnetic heating processes
3.6.15.3
Radiant heat flux distribution
3.6.15.4
Acoustic fields
3.6.16 Gaseous plumes
3.6.16.1
Monitoring
3.6.16.2
Mapping
3.6.17 Ground frostline mapping
3.7
Friction investigations: typical examples may include, but are not limited to, the
following:
3.7.1 Bearings
3.7.2 Seals
3.7.3 Drive belts
3.7.4 Drive couplings
3.7.5 Exposed gears
3.7.6 Gearboxes
3.7.7 Machining processes
3.7.8 Aerodynamic heating
3.8
Fluid flow investigations: typical examples may include, but are not limited to, the
following:
3.8.1 Fluid piping
3.8.2 Valves
3.8.3 Heat exchangers
3.8.4 Fin fans
3.8.5 Cooling ponds
3.8.6 Cooling towers
3.8.7 Distillation towers
3.8.7.1 Packed
3.8.7.2 Trays
3.8.8 HVAC systems
3.8.9 Lake and ocean current mapping
3.8.10 Mapping civil and industrial outflows into waterways
3.8.11 Locating leaks in pressure systems
3.8.12 Filters
3.9
Thermal resistance (steady state heat flow) investigations: typical examples may include,
but are not limited to, the following:
3.9.1 Thermal safety audits
3.9.2 Low temperature insulating systems
3.9.4 Industrial insulation systems
3.9.5 Refractory systems
3.9.6 Semitransparent walls
3.9.7 Furnace interiors
3.9.8 Disbonds in lined process equipment
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 29
The Professional Thermographers Association
3.10
4.0
5.0
6.0
Thermal capacitance investigations: typical examples may include, but are not limited to,
the following:
3.10.1 Tank levels
3.10.2 Rigid injection molding
3.10.3 Thermal laminating processes
3.10.4 Building envelopes
3.10.5 Roof moisture
3.10.5.1
Roof level investigations
3.10.5.2
Airborne investigations
3.10.6 Underground voids
3.10.7 Bridge deck laminations
3.10.8 Steam traps
3.10.9 Paper manufacturing moisture profiles
3.10.10 Subsurface discontinuity detection in materials
3.10.11 Coating disbond
3.10.12 Structural materials
3.10.12.1 Subsurface discontinuity detection
3.10.12.2 Thickness variations
3.10.12.3 Disbonding
Interpretation/Evaluation
4.1
Exothermic or endothermic investigation: typical examples may include, but are not limited
to, the examples shown in Section 3.6.
4.2
Friction investigations: typical examples may include, but are not limited to, the examples
shown in Section 3.7.
4.3
Fluid flow investigations: typical examples may include, but are not limited to, the
examples shown in Section 3.8.
4.4
Differences in thermal resistance (stead state heat flow) investigations: typical examples
may include, but are not limited to, the examples shown in Section 3.9.
4.5 Thermal capacitance investigations: typical examples may include, but are not limited to, the
examples shown in Section 3.10.
Procedures
5.1
Existing codes and standards
5.2
Elements of thermal/infrared testing job procedure development
Safety and Health
6.1
Safety responsibility and authority
6.2
Safety for personnel
6.2.1 Liquefied nitrogen handling
6.2.2 Compressed gas handling
6.2.3 Battery handling
6.2.4 Safety clothing
6.2.5 Safety ropes and harnesses
6.2.6 Ladders
6.2.7 Safety backup personnel
6.3
Safety for client and facilities
6.4
Safety for testing equipment
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 30
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 31
The Professional Thermographers Association
What you need to do to get your Level I Certificate
Pass a Vision test as outlined in SNT TC1A.
Study requirements of 32 hours (or 4-8 hr days) of course material as recommended in the SNT
TC -1A outline.
A minimum of 210 hours (26.25 - 8 hour days, or 5.25 weeks) of actual field work experience.
Receive a passing composite grade of 80% with no indivual exam score lower then 70%.on the:
General (minimum 40 questions),
Specific(minimum 20 questions)
Practical exams(minimum of 10 documented thermal anomalies summited in a report), or higher
The Report
An Introduction
Who the inspection was for and when
A summary of the results
Number of problems found and the severity
Specific Inspection information as to:
Who did the inspection,
What camera was used
How the inspection was preformed.
Complete Inventory of:
What was to be tested
What was tested
What was not tested and why.
Prioritized list.
Temperature Rise
Crucially to Operation
Before vs. After Failure
10 to 12 “problems” documented with Thermograms
10 to 12 “Individual Problems”
Documented with:
Thermogram
Matching Photo
Temperature of the problem Component
Temperature of a reference Component
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 32
The Professional Thermographers Association
Temperature Rise (deference) between the problem and reference component.
Equipment Location Information
Name of the equipment
Detailed description of the problem condition.
Where the “Problem” temperature measurement was made from
Where the “Reference” temperature measurement was made from.
If Electrical, Rated Load and Tested % of Load
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 33
The Professional Thermographers Association
Recommended Training References
Books and Publications
Applied Infrared Photography. Eastman Kodak Company, 1987.
Asimov, Isaac. Understanding Physics. Three volumes in one, Dorset Press, 1988.
ASNT’s Infrared Thermography Topical Conference 1997 Paper Summaries Book. Columbus, OH:
The American Society for Nondestructive Testing, Inc., 1997.*
Bernard, Bertram. The ABC’s of Infrared. Howard W. Sams & Co., Inc., 1970.
Buckely, Robert E. and Gene D. Nutter. Handbook of Applied Termal Design. New York: McGraw Hill,
1988.
Burnay, S. G., et. al., eds. Application of Thermal Imaging. Bristol, UK: Adam Hilger, 1988.
Cohen, Julius. (1983, May). Elements of Thermography for Nondestructive Testing, NBS Technical
Note, V1177 GPO S/N 83020123.
Colwell, Robert N., ed. Manual of Remote Sensing, second edition, Volumes I and II. American Society of
Photogrammetry, 1983.
DeWit, D. P. Theory and Practice of Radiation Thermometry. New York: John Wiley & Sons, Inc., 1989.*
Electro-Optics Handbook. Burle Industries, 1974.
Falk, David S., Dieter R. Brill, and David G. Stork. Seeing the Light. New York: John Wiley and Sons,
1977.
Gaussorgues, G. Infrared Thermography (Original French Title: La Thermographie Infrarouge).
Translated
by S. Chomet. Microwave Technology Series 5. English Language Edition. Chapman & Hall, 1994.**
Geankoplis, Christie J. Transport Processes and Unit Operations. Boston, MA: Allyn & Bacon, 1978.**
Guyer, Eric C. Handbook of Applied Thermal Design. New York: McGraw-Hill, 1989.
Hackforth, Henry L. Infrared Radiation. McGraw-Hill, 1960.**
Holman, J. P. Experimental Methods for Engineers, seventh edition. New York: McGraw-Hill, 2000.
Holman, J. P. Heat Transfer. New York: McGraw-Hill, 1997.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 34
The Professional Thermographers Association
Holst, Gerald C. Testing and Evaluation of Infrared Imaging Systems, second edition. JCD Publishing
Co.,
1998.
Holst, Gerald C. Common Sense Approach to Thermal Imaging. JCD Publishing Co., SPIE – The
International Society for Optical Engineering, 2000.
Hougan, O. A., K. M. Watson, and R.A. Ragatz. Chemical Process Principles, third edition. New York:
John
Wiley & Sons, Inc., 1985.
Incopera, Frank P. and David P. DeWitt. Fundamentals of Heat and Mass Transfer, fourth edition. New
York: John Wiley & Sons, Inc., 1996.*
Kaplan, Herbert. Practical Applications of Infrared Thermal Sensing and Imaging Equipment, Vol.
TT13. Bellingham, WA: SPIE Press, 1993.
Kerlin, Thomas W. and Robert L. Shepard. Industrial Temperature Measurement. Research Triangle
Park, NC: Instrument Society of America, 1982.
Kreith, Frank and Mark S. Bohn. Principles of Heat Transfer. New York: Harper & Row, 1986.
Lloyd, J. M. Thermal Imaging Systems. New York: Harper and Row, 1986.
Lucier, Ronald D., ed. Selected Papers on Temperature Sensing: Optical Methods, volume MS 116,
Milestone
Series. SPIE Optical Engineering Press, 1995.
Madding, Robert P. Thermographic Instruments and Systems. University of Wisconsin-Extension,
Department of Engineering and Applied Science, 423 North Lake Street, Madison, WI 53706.**
Magnum, B. W. and G.T. Furukawa. Guidelines for Realizing the International Temperature Scale of 1990.
Washington D.C.: U.S. Government Printing Office, 1990.
Maldague, Xavier P. V. Nondestructive Evaluation of Materials by Infrared Thermography. London:
Springer-Verlag, 1993.*
Maldague, Xavier P.V., editor. Infrared Methodology and Technology. Gordon and Breach Science
Publishers, 1994.
Manual for Thermographic Analysis of Building Enclosures. 149-GP-2MP, Committee on Thermography.
Canadian General Standards Board, 1986. MIL-HDBK-728 Vol. 1 “Nondestructive Testing.”
Pettersson, Bertil. Thermography, Testing of the Thermal Insulation and Airtightness of Building. Swedish
Council for Building Research, 1980.
Rohsenow, Warren M. and James P. Hartnett, eds. Handbook of Heat Transfer. McGraw-Hill, 1973.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 35
The Professional Thermographers Association
Schlessinger, Monroe. Infrared Technology Fundamentals, Optical Engineering Series/46. second edition,
revised and expanded. Dekker, Marcel Inc., 1994.
Simon, Ivan. Infrared Radiation. D. Van Nostrand Company, Inc., Van Nostrand Momentum Book #12,
1966.**
Standards, Tentative Standards, and Special Technical Publications (STP) Relating to Infrared Imaging
Applications. Philadelphia, PA: American Society for Testing and Materials. Latest issue.
Stanley, Roderick K., technical ed.; Paul McIntire and Patrick O. Moore, eds. Nondestructive Testing
Handbook, volume 9: second edition, Special Nondestructive Testing Methods. Columbus, OH: The
American Society for Nondestructive Testing, Inc., 1995.*
Thomas, Roderick A. The Thermography Monitoring Handbook, first edition. Coxmoor Publishing Company,
1999.
Tipler, Paul A. Modern Physics. second edition. Worth Publishers, Inc., 1978.
Turner, William C. and John F. Malloy. Thermal Insulation Handbook. New York: McGraw-Hill, 1981.**
Va_ko, Antonín, D. Sc., Infra-red Radiation. English translation edited by P.S. Allen, B.Sc., PhD., Lecturer,
Department of Physics, University of Nottingham. Translated by Sylvia E. Myhre-Finková and Karel
Fink 1967. English edition © Iliffe Books Ltd., 1968.**
von Baeyer, Hans Christian. Warmth Disperses and Time Passes – The History of Heat. Random House,
1999.
Walder, Jearl. The Flying Circus of Physics. John Wiley & Sons, 1977.
Wolfe, William L. and George J. Ziessis, eds. The Infrared Handbook. The Environmental Research
Institute of Michigan (prepared for The Department of Navy), 1985.
Zemansky, Mark W. Temperatures: Very Low and Very High. Dover Publications, 1981.
Ziessis, George J., ed. The Infrared & Electro-Optical Systems Handbook, Volumes 1-8. Bellington, WA:
SPIE Press, 1993.
* Available from the American Society for Nondestructive Testing, Columbus, Ohio.
** This book is a Recommended Reference because of the valuable data it contains. This title is currently out
of print, however, and is not available from ASNT.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 36
The Professional Thermographers Association
The Discovery of Infrared Radiation
by William Herschel
The discovery of infrared radiation is commonly ascribed to
William Herschel, the astronomer in the early 19th century
(1800’s). Herschel used a prism to refract light from the sun
and detected the infrared, beyond the red part of the
spectrum, through an increase in the temperature recorded
on a thermometer.
Sir Frederick William Herschel (1738-1822) was born in
Hanover, Germany and became well known as both a
musician and as an astronomer. He moved to England in
1757 and, with his sister Caroline, constructed telescopes to
survey the night sky. Their work resulted in several catalogs
of double stars and nebulae. Herschel is famous for his
discovery of the planet Uranus in 1781.
Herschel made another dramatic discovery in 1800. He wanted to know how much heat was passed
through the different colored filters he used to observe sunlight. He noted that filters of different colors
seemed to pass different amounts of heat. Herschel thought that the colors themselves might be of varying
temperatures, so he devised a clever experiment to investigate his hypothesis.
He directed sunlight through a glass prism to create a
spectrum (the rainbow created when light is divided into its
colors) and then measured the temperature of each color.
Herschel used three thermometers with blackened bulbs (to
better absorb heat) and, for each color of the spectrum,
placed one bulb in a visible color while the other two were
placed beyond the spectrum as control samples. As he
measured the individual temperatures of the violet, blue,
green, yellow, orange, and red light, he noticed that all of
the colors had temperatures higher than the controls.
Moreover, he found that the temperatures of the colors
increased from the violet to the red part of the spectrum.
After noticing this pattern Herschel decided to measure the
temperature just beyond the red portion of the spectrum in
a region where no sunlight was visible. To his surprise, he
found that this region had the highest temperature of all.
Herschel performed additional experiments on what he called "calorific rays" (derived from the Latin word
for heat) beyond the red portion of the spectrum. He found that they were reflected, refracted, absorbed
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 37
The Professional Thermographers Association
and transmitted in a manner similar to visible light. What Herschel had discovered was a form of light
(or radiation) beyond red light, now known as infrared radiation. [The prefix infra means below.]
Herschel's experiment was important because it marked the first time that someone demonstrated that
there were types of light that we cannot see with our eyes.
Recent developments in detector technology have led to many useful applications using infrared radiation.
Medical infrared technology is used for the non-invasive analysis of body tissues and fluids. Infrared
cameras are used in police and security work, as well as in military surveillance. In fire fighting, infrared
cameras are used to locate people and animals caught in heavy smoke and for detecting hot spots in forest
fires. Infrared imaging is used to detect heat loss in buildings, to test for stress and faults in mechanical and
electrical systems, and to monitor pollution.
Infrared satellites are routinely used to measure ocean temperatures, providing an early warning for El Nino
events that usually impact climates worldwide. These satellites also monitor convection within clouds,
helping to identify potentially destructive storms. Airborne and space-based cameras also use infrared light
to study vegetation patterns and to study the distribution of rocks, minerals and soil. In archaeology,
thermal infrared imaging has been used to discover hundreds of miles of ancient roads and footpaths,
providing valuable information about vanished civilizations.
Fascinating, new discoveries are being made about our Universe in the field of infrared astronomy. The
universe contains vast amounts of dust, and one way to peer into the obscured cocoons of star formation
and into the hearts of dusty galaxies is with the penetrating eyes of infrared telescopes. Our universe is
also expanding as a result of the Big Bang, and the visible light emitted by very distant objects has been
red-shifted into the infrared portion of the electromagnetic spectrum.
An Example of Herschel Infrared Experiment
In the year 1800, Sir William Herschel discovered
the existence of infrared by performing an
experiment very similar to the one we show here.
Herschel passed sunlight through a prism. As
sunlight passes through the prism, the prism
divides it into a rainbow of colors called a spectrum.
A spectrum contains all of the colors which make
up sunlight. Herschel was interested in measuring
the amount of heat in each color. To do this he
used thermometers with blackened bulbs and
measured the temperature of the different colors of
the spectrum. He noticed that the temperature
increased from the blue to the red part of the
spectrum. Then he placed a thermometer just past the red part of the spectrum in a region where there was
no visible light and found that the temperature there was even higher. Herschel realized that there must be
another type of light which we cannot see in this region. This light is now called infrared.
It is very easy to recreate this experiment. All you need is an equilateral glass prism, 3 alcohol
thermometers, a white piece of paper and a box. The cost of the prism we used was about $7.50 and the
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 38
The Professional Thermographers Association
thermometers were 75 cents a piece. You will need to blacken the bulbs of the thermometers to make this
experiment work. To do this we painted the bulbs with flat black paint. The bulbs of the thermometers were
blackened to better absorb heat.
In the above image you can see how to set up this experiment for use outdoors. We placed a white piece of
paper at the bottom of a cardboard copy paper box. Next we rotated the prism until a good wide spectrum
appeared on the white paper at the bottom of the box. To get a good spectrum we had to tilt the box up on
the prism end by placing a flat rock under it.
First check the temperature of the thermometers away from the spectrum in the
shaded area of the box. The above image shows the temperature before the
thermometers are placed in the spectrum. All 3 read 76 degrees, which is the outdoor
shade temperature.
Now let's place the thermometers in the spectrum. We will place the left bulb in the
blue part of the spectrum, the middle bulb in the yellow part of the spectrum, and the
right bulb just past the red part of the spectrum in a region where there is no visible
light.
The above image shows the temperature readings after about 1 minute. It takes a few minutes for the
temperatures to reach their final value. Within 1 minute you can already see a difference in temperature.
The thermometer in the blue part of the spectrum shows the lowest reading which is not much higher than
the shade temperature. The yellow part of the spectrum is showing a much higher temperature than the
blue. The thermometer on the right, which is in the dark region just past the red, is showing the highest
temperature of all 3 regions. (The sun had moved slightly by the time this picture was taken, and hence the
right-most bulb just started to have a small portion of the red spectrum shining on it).
The differences between the 3 temperature readings continue to grow larger until the final temperatures are
reached (shown above). You can now see that the dark area shows a much higher temperature than the
areas which are in regions of visible light.
Final readings are: blue: 80 degrees yellow: 83 degrees infrared: 86 degrees
Note: The differences between the temperatures of the colors of the spectrum vary with the width of the
spectrum, which depends on time of day, and the distance from the prism. In all cases the trend of
temperature increasing from blue to infrared should still appear.
Herschel's experiment was important not only because it led to the discovery of infrared light, but also
because it was the first time that someone showed that there were forms of light that we cannot see with
our eyes. As we now know, there are many other types of light that we cannot see and visible colors are
only a very small part of the entire range of light which we call the electromagnetic spectrum.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 39
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 40
The Professional Thermographers Association
Introduction to Temperature Measurement
With the advent of organizations such as the International Organization for Standardization (ISO), there has
been increased pressure to assure that all measurement including temperature measurement is both
accurate and repeatable to set standards. The goal of ISO is to develop and promote common standards
for manufacturing and sciences worldwide. In the US the American National Standards Institute (ANSI) is
the motivating organization behind this effort.
Most of the documents issued by these organizations require that the accuracy of measurement
instruments be traceable to an accepted standard. With temperature measurement the accepted standards
are those set by the National Institute of Standards and Technology (NIST) in Washington. In collaboration
with the International Bureau of Weights and Measures they provide the basis for making and
communicating measurements.
To adhere to proper measurement standards all test equipment must be calibrated at known intervals to
national standards. The calibration procedures must be documented and calibration records maintained.
Most manufacturers of temperature measurement equipment supply a calibration document with delivery of
each system. This document should indicate the frequency for recalibration, as well as the precision and
accuracy of your specific piece of equipment.
There are a number of specialized tests to determine the measurement precision and accuracy of infrared
imaging radiometers. They are described in standards documented by the American Society for Testing
and Materials (ASTM).
Infrared/Thermal programs do not rely entirely on Infrared noncontact devices to record accurate
temperature. There are a number of times when contact measurement must be employed as a
secondary tool or to verify specific results. There are cases where contact measurement is both
easier and less time consuming. To this end it is necessary to consider what devices are available for
performing this task. The Thermographer should be aware of the many different types of contact and noncontact devices and the basic theory behind their operation and use.
Contact Temperature Measurement Instruments
One way to define temperature is the energy level of matter evidenced by some physical measure of that
matter. Even though temperature sensors vary greatly they all measure temperature by sensing some
physical characteristic.
Seven basic types of contact temperature sensors are: thermocouples, resistive temperature
devices/thermistors, bimetallic devices, liquid-expansion devices/thermometers, molecular change-of-state
devices, filled system thermometers (bourdon) and silicon diodes. Because these devices vary greatly in
their advantages, limitations and costs, you need to familiarize yourself with their differences to compare
them objectively. Being able to do so also will allow you to choose the appropriate measurement instrument
for a specific application.
Thermocouples
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 41
The Professional Thermographers Association
Thermocouples can provide fast, accurate and inexpensive temperature measurement over a wide
temperature range. Thermocouples are the most commonly used and most rugged temperature measuring
devices in industrial settings. Compared to other methods of measuring temperature, thermocouples are
economical, especially when base metals are used instead of platinum or other expensive metals.
How a Thermocouple Works
Thermocouples operate on the principle of Thermal Electromotive Force (EMF), first observed by Thomas
Seebeck in 1821. Seebeck discovered that two dissimilar metal wires, joined at both ends, will produce a
current in the thermoelectric circuit when the junctions are at different temperatures. The current produced
is a function of the junction temperature and the type of metals used. The relationship between thermal
EMF and temperature is now known as the Seebeck effect.
The thermal EMF can be measured by breaking the thermocouple loop at any point and measuring the
open circuit voltage. For example, a typical K-type thermocouple, made of chrome/alumel generates about
40 microvolts per degree Celsius.
All dissimilar metals exhibit the Seebeck effect, but the voltage/temperature relationship varies depending
on the metals. Various combinations of metals are used depending on the output voltage required and the
temperature range.
Resistive Temperature Devices and Thermistors
Like thermocouples these devices measure an electrical parameter. Rather than sensing an EMF level,
these units employ another characteristic of matter that changes with temperature: electrical resistance.
Resistive temperature devices (RTDs) and thermistors are second only to thermocouples in popularity.
They use electrical characteristics that lend themselves to easy measurement, transmission and recording.
Of the three (thermocouples, RTD’s, & thermistors) RTDs are the most stable, over a temperature range up
to 850ºC. In general, RTDs also are more linear than thermocouples. As temperatures increase, the RTD’s
resistance increases in a positive direction.
The thermistor is constructed differently from the RTD. It is a non-linear semiconductive device in which
resistance decreases as temperature increases. While thermocouples may be the most popular of these
three devices, thermistors are the most accurate. They too however, have a restricted temperature range.
They can not be used in applications where the temperature may reach several hundred degrees.
Being resistive devices, thermistors and RTDs have another disadvantage compared to thermocouples.
Measuring resistance requires that a small current pass through these devices. Unfortunately this current
generates heat as it passes through these sensors. This ultimately affects the temperature of the media
being measured and thus, affects the accuracy of the measurement. This problem is minimized by keeping
the current small and hence keeping the generated heat small. Nevertheless, the self-heating in resistive
sensors important if you are taking measurements in still air or in a partial vacuum. In such cases, less of
the generated heat is carried away than in the case of a dense or moving medium. Depending on your
application, this limitation could be an important factor in making a purchase choice.
Bimetallic Devices
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 42
The Professional Thermographers Association
Bimetallic devices, sometimes called bimetal thermometers, indicate temperature by measuring the
differential expansion of two heated metals. The two metals are bonded together and mechanically linked
to a pointer. When heated, one side of the bimetallic strip expands more than the other. This expansion
moves the pointer, which then indicates the temperature.
Bimetallic devices are convenient because they are portable and do not need a power supply. They
typically are not as accurate as electrical devices, such as thermocouples, RTDs and thermistors.
Furthermore, you cannot record their temperature values easily. In many applications, their portability
make up for these disadvantages.
Liquid-Expansion Devices/Thermometers
Generally speaking, these devices measure the expansion of a liquid in a capillary tube and are available
with two liquids: mercury and red organic fluid. The distinction between the two is notable.
Unlike the organic models, mercury devices have limitations on where you can use them and on how you
can transport them. Mercury is considered an environmental contaminant.
Be aware of using them in food related applications. As well, be sure to check the latest restrictions for air
transportation of mercury products before shipping.
Molecular Change-of-State Devices
These units indicate temperature according to a material's state of being. For example, a state-change
takes place when ice becomes water or water becomes steam. These changes occur at characteristic
temperatures (for a given atmosphere pressure); so when a material changes state, the temperature of the
system is automatically known. Change-of-state devices are available in a variety of forms: labels, pellets,
crayons and paints — each is suited for various situations.
In one application, you might find temperature labels on steam traps. When a trap needs adjustment, it
becomes hot. The heat activates a white dot on the label, which turns black, indicating a rise in
temperature beyond the rated level. The dot then remains black, even if the temperature of the steam trap
returns to normal. This is an example of a non-reversible label.
A reversible label operates on a different principle using a liquid crystals. This material changes color, from
black to brown, blue, or green, depending on the temperature reached. A typical label is all black when
temperature of the equipment it monitors is below the label's setpoint. But, as the temperature rises, the
color changes. For example, if the label's set point is 5oC, the label may turn blue, then green, and finally,
brown as it passes 5oC.
In any particular liquid crystal device, you usually will see two color spots adjacent to each other - a blue
one slightly below the temperature indicator, and a brown slightly above. These spots allow you to estimate
the temperature as being between 30oC and 39oC, for example. Though not precise, this type of device
does offer the advantage of being small, rugged and non-electrical.
Other types of change-of-state devices do not change colour such as lacquer and crayons. Marks made
with them simply disappear when the temperature exceeds the rated level. If you check for a mark and it is
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 43
The Professional Thermographers Association
no longer there, then you know the temperature of the marked item exceeded the temperature that the
sensor was designed to detect. Pellet-version sensors work similarly. You simply place the pellets on the
equipment of interest. The pellets become visually deformed or melt away at predetermined temperatures.
A limitation of change-of-state devices is that they have relatively slow response times. So they may not
detect short term temperature spikes during a particular process. As well, their overall accuracy is not as
good as other devices. However, because change-of-state devices are non-electrical, they have an
advantage in certain applications. For example, non-reversible temperature labels are useful, either for
engineering or legal reasons, for confirming that the temperature of an item did not exceed a certain level
during shipment.
Filled System Devices
This type of measurement device is designed to provide an indication or record of temperature at some
distance from the point of measurement. There are usually four components to this type of system. They
have an element sensitive to temperature change (bulb), an element sensitive to pressure or volume
change (bourdon spring, bellows or diaphragm), an indicator or recording device convert the information
into a usable temperature value and a means of connecting these together (capillary tubing).
The temperature sensing element contains a fluid which changes its volume or pressure with temperature.
The pressure-sensitive element (bourdon) responds to the changes by delivering a motion or a force to the
indicator or recording device. This is usually a mechanical device that moves a pointer on a gauge or pen
on a paper recording device. It can also be a pneumatic or electronic device which transmits the
temperature information over long distances. This measurement device is frequently used for process
control purposes.
There are two types of filled system devices. They are those in which the bourdon responds to volume
changes and those that respond to pressure changes. The volume change devices are completely filled
with a liquid while the pressure filled devices are either filled with a gas or partially filled with a volatile
liquid.
Silicon Diode Sensors
Silicon diode sensors measure the cryogenic (very low) temperature range. They are linear devices in
which the conductivity of a silicon diode varies almost linearly with changes in temperature. Their accuracy
is greatest at temperatures below 100K (-173oC). A typical upper limit for the best silicon diode sensor is
75K (-199oC).
By contrast, some silicon diode sensors measure temperatures as low as 1.5K with great precision
(+0.25K).
As with RTDs and thermistors, self heating may be a problem in a specific application; but again, the lower
the current, the less power dissipated in the sensor.
Clearly, silicon diode sensors are devices for special applications. There are various models available to
accommodate the degree of precision required for a specific cryogenic application.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 44
The Professional Thermographers Association
Non-contact Temperature Measurement devices
Non-contact temperature calculations began as far back in history as mans first experiences with firing
pottery, making glass and working with metals. The human eye was the device used to determine specific
allowable temperature ranges for many applications. Even today the human eye is still the device used by
the modern blacksmith to determine by color when the temperature of a metal is just right to apply the
hammer to shape the object. Glass blowers likewise use the human eye to determine by color the
temperature of his work piece and the action he/she will pursue.
Today we have a number of sophisticated, non-contact measurement devices with which to accurately
calculate temperatures to very close tolerances. Radiation thermometry has in the last few years,
experienced incredible growth due to both public awareness and the advancement of equipment in the
areas of accuracy and convenience. As manufacturers push the limits of some of the new technology, there
may be instruments introduced in the market place that do not relate well to all applications but rather to
specific application areas.
Measurement Devices
There are a number of noncontact measurement devices which can be grouped into a set of instruments
known as radiometers. A radiometer is an electro-optical system designed to measure radiant power. A
radiation thermometer is an instrument that has been calibrated to a standard and has the capability of
calculating temperature by a specific means. These have been given various names such as pyrometers or
spot radiometers. We will first discuss some of these instruments
Optical Pyrometers
From the turn of the century until about 1956, the disappearing filament optical pyrometer was the most
accurate instrument for measuring high temperatures. This device is comprised of a low powered telescope
containing a small tungsten-filament vacuum lamp, a red glass filter and two aperture stops. The lamp
filament lies in the plane of the target image formed by the objective lens with the optical axis intersecting
the center of the filament. Flat windows in the lamp envelope permit an undistorted view of the target, and
the magnified image of the lamp filament as viewed through the telescope is seen to be superimposed on
the image of the target.
Direct current supplied to the filament is manually adjusted to cause the luminance of the center of the
filament to match that of the target . Since the color of the glowing tungsten filament is somewhat different
than that of the target even when at the same luminance, a red glass filter is placed between the lamp and
the eye. When the luminance is equal the filament “disappears” . The instrument is calibrated by matching
the current to known temperatures. This gives a scale of temperatures based on the current readings
necessary to match the color of the filament to the radiant color of the target . These instruments have for
the most part been replaced now by spot radiometers.
Radiation Ratio Thermometers or Two Color Pyrometers
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 45
The Professional Thermographers Association
Radiation ratio thermometry involves measuring thermal radiance at two different wavelengths and inferring
the temperature from the ratio of these spectral radiances. Although measuring two signals rather than one
provides increased difficulty, radiation ratio thermometers can be significantly more accurate that single
band devices.
These devices can be both emissivity independent, spot size independent and atmospheric attenuation
independent due to the fact that we are considering the ratio of signals received rather than the total
radiation. There are some difficulties with these instruments due to the fact that you are gathering
information in two different wavelengths.
Spot Radiometers
The radiation thermometer commonly known as a spot radiometer is a single wavelength band instrument
that has been calibrated to a blackbody at known temperatures and has the ability to calculate
temperatures from the measured radiated power of a target. It is by far the most popular noncontact
measurement device in the industrial commercial market.
The radiometer is made up of some type of focusing system that directs the energy from the target, a
detector, electronics and a digital read out . As most of the objects encountered in real life are not
blackbody sources, consideration must be given to emissivity, reflection, atmospheric attenuation and
relative humidity if accurate temperature calculations are required.
The detector is usually a longwave thermal detector that requires no cooling and can be inexpensively
produced. Industrial models are priced as low as $199 for models that are very basic with no user
controlled functions. For most applications the accuracy and repeatability of these units are acceptable.
The largest restriction on the use of these devices is the target spot size. The ratio of spot size to distance
is often the limiting factor in the use of spot radiometers. Models vary from a 1:12 ratio all the way up to a
1:360 ratio.
When considering whether or not a spot radiometer is suitable for your measurement application attention
must be given to:
1. target emissivity
2. target size verses distance
3. estimated temperature range
4. accuracy required
5. atmospheric conditions
6. measurement points required
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 46
The Professional Thermographers Association
When purchasing a spot radiometer attention should be given to the following product characteristics:
1. User available controls
a. emissivity
b. background reflection
c. distance
2. Spot size ratio
3. Temperature range
4. Sighting capabilities
5. Wavelength
6. Data storage and output signal
7. Calibration information and traceability
Optical Fiber Thermometry
Optical Fiber Thermometry (OTF) is being used worldwide in demanding and hostile applications in
industrial processes, semiconductor application, and in research. Systems can be contact or non-contact.
The sensor is coupled to flexible optical fiber, which conducts light emitted from the target to a photo
detector in the electronics unit. The detector converts the light energy into an electrical signal, which is
then digitized. The signal is converted into a temperature value using Planck’s radiation equation. These
products use short wavelength (infrared) pyrometry and blackbody sensing technology to provide highprecision temperature sensing solution in hostile environments. They are highly precise and accurate, and
immune to electromagnetic interference. Their range of operation is 100 C to 4 000 C.
One such system uses patented Ripple Technology to provide non-contact, in situ, real-time wafer
temperature measurement in lamp heated environments. Ripple Technology takes advantage of small
changes in radiant output created by the alternating current in heating lamps. The AC component of the
lamp is detected in the light reflected from the wafer surface and is analyzed to determine the emissivity
and thermal radiance of the wafer. This allows an accurate measurement of the wafer surface
temperature, independent of emissivity, heat flow, reflected radiation, or chamber history and configuration.
Wafer temperatures can be measured to within 2 oC in a range from 600 oC to 1200 oC from outside the
process chamber. Changes in wafer emissivity can be resolved within 0.001.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 47
The Professional Thermographers Association
Thermal Imaging Systems
In our consideration of thermal imagers, we could categorize them in a number of different ways. They can
be divided by wavelength, by detector type, by cooling system or by image generation method. They can
also be subdivided into imagers and imaging radiometers.
Thermal Imagers
Thermal imagers are imaging systems which do not have measurement capabilities. They are used
mainly for applications such as forest fire protection, search and rescue, surveillance, flat roof inspections
and electronic news gathering. The main consideration in evaluating an imager is of course the image
quality. This involves factors such as spatial resolution, noise equivalent temperature difference and
possibly minimum resolvable temperature difference. Many other factors such as ease of operation,
portability, durability, service, support and warranty must be taken into account. For most industrial
predictive maintenance applications, these instruments are not suitable due to their inability to measure
temperature.
Imaging Radiometers
Imaging radiometers are infrared systems that have the ability to measure the radiated energy incident
upon the detector unit from the scene in the field of view. With the assistance of onboard computers,
temperatures can be calculated across the image.
In general, older imaging radiometers were scanning systems or pyroelectric starring systems while the
newer generation of equipment is Focal Plane Array (FPA) staring systems. Scanning systems are both
single element and multi element. They use the motion of prisms or mirrors to scan the field of view and
produce an image. In general the spatial resolution of these systems is not as high as those of the new
FPA staring systems. The scanning systems also require a number of moving parts. They use cooled
photon detectors. In photon detectors incident infrared photons are absorbed producing free charge
carriers that change an electrical characteristic of the responsive element. This process is carried out
without any significant change in the temperature of the responsive element. Photon detectors are cooled
detectors. They are cooled by liquid nitrogen, a stirling cycle cooler, or thermoelectrically by the Peltier
effect.
Pyroelectric Vidicons are staring systems which also have lower spatial resolution than the new FPA
systems. They make use of an uncooled thermal detector. When the responsive element absorbs an
incrementally increased or decreased amount of infrared radiation, its temperature rises or falls, changing
the surface charge. With the appropriate external circuit, this change in charge leads to a signal voltage.
With no change in temperature there is no charge and no signal. This means that the vidicon must either be
constantly moved or the field of view changed by a “chopper” continuously changing the temperature to
produce an image.
The new generation of infrared instruments on the market today are focal plane arrays. These systems
stare constantly at the scene. Some systems have a variable dwell time which allows for fast imaging in the
order of 1/400 of a second on some R&D models.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 48
The Professional Thermographers Association
FPAs are high resolution systems which have either 65,536 (256 X 256) elements or 76,800 (320 X 240)
elements. At the time of the writing of this manual these are the two common commercial systems
available. Some research type systems have 262,144 (512 X 512) elements producing an incredibly high
resolution image.
These arrays consist of either cooled photon detectors or uncooled thermal detectors. The short wave
cooled systems use a stirling cycle cooler which cools the detector to -196oC. Most of the cooled systems
on the market today use helium as the medium. The gas is compressed and then allowed to expand thus
cooling the element. The typical life span of these coolers is approximately 3 - 5 years. At that time they
will need to be serviced or replaced. These cooled systems operate in the short-wave region between
approximately 3.5 - 5 microns.
The uncooled thermal detector FPAs use either the bolometric effect or the ferroelectric effect. The
bolometric effect is a change in the electrical resistance of the responsive element due to temperature
changes produced by absorbed incident infrared radiation. Although these systems are labeled uncooled
they must have some type of thermal stabilization in order to maintain accuracy and repeatability. Some
use a thermoelectric stabilizer which allows the system to have no moving parts.
These uncooled systems operate in the longwave region between approximately 7 - 13 microns.
Despite the wavelength differences both types of systems claim a temperature range of - 20 oC to + 2000
oC with the use of special filters. When considering which system is best for your application, one must
consider the operating wavelength band.
All of the new systems available are 14 bit systems. This feature allows the operator to store the full
increments in the dynamic range of the system. The older systems are 8 bit systems and only allow storage
of 256 increments of the dynamic range. This is a very important when considering post processing issues.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 49
The Professional Thermographers Association
Selecting an Infrared Camera for your needs
Selecting which instrument to purchase can be a stressful endeavor if one does not objectively approach
the task at hand. Many times pressure from management or from salespeople or from peers can bias your
choice and produce a faulty decision. The best way to evaluate instruments for purchase is to use an
instrument evaluation matrix which allows you to objectively grade each instrument based on a prescribed
set of parameters vs. your needs.
The first step in this process is to assess your application needs and decide if there are any areas that
should be given added weight. For instance if the application entailed the operator to be constantly climbing
up and down a ladder or stairs, system portability and weight might be given priority in the grading process.
Contrary to some sales arguments both longwave and shortwave systems do work in most general
applications.
In the decision matrix, minor considerations could have a possible grade of 0 - 3, intermediate concerns a
possible grade of 0 - 6, and critical issues a possible grade of 0 - 10. This type of a grading system allows
important issues to have more weight in your choice of equipment.
The decision matrix may have any number of items depending on the applications that must be
accommodated. Choosing the proper instrument may affect the success of your program in many ways.
Issues such as service, longevity and support may only be derived by some calls to existing customers for
third party input.
Manufacturer: ______________________________________________
System:___________________________________________________
ITEM
Accuracy
Batteries
Controls
Cooler
Ergonomics
Filters
Focus
IFOV
Image Qual.
Image Storage
IMFOV
Maintenance
Measurement
Menus
MRTD
NETD
Operation
Specification
Weight
Grade
Ass.
Conf.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 50
The Professional Thermographers Association
Portability
Price
Service
Software
Startu p
Support
Temp Range
Warranty
Wavelength
Weight
Zoom
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 51
The Professional Thermographers Association
Thermography
Thermography can refer to a printing process and a imaging process.
Thermographic Imaging
Thermography, or thermal imaging, is
a type of infrared imaging.
Thermographic cameras detect radiation
in the infrared range of the
electromagnetic spectrum (roughly 3 to
14 microns) and produce images of that
radiation. Since infrared radiation is
emitted by all objects at ambient
temperature, according to the black body
radiation law, thermography makes it
possible to "see" one's environment with
or without visible illumination. The
amount of radiation emitted by an object increases with temperature, therefore thermography allows you to
see variations in temperature, hence the name. With a thermographic camera warm objects stand out well
against cooler backgrounds. Humans and other warm-blooded animals become easily visible against the
environment day or night, hence historically its extensive use can be ascribed to military and security
services.
Thermal imaging finds many
Temperature
other uses. For example,
Temperature Zones
Zones Weather
Weather Maps
Maps
firefighters use it to see through
and
and Thermograms
Thermograms
smoke, find persons, and
In
localize hotspots of fires. With
In some
some ways
ways the
the map
map of
of the
the
US
showing
the
temperatures
US
showing
the
temperatures
thermal imaging, power lines
is
is similar
similar to
to aa thermogram
thermogram of
of
maintenance technicians locate
electrical
electrical equipment:
equipment:
overheating joints and parts, a
telltale sign of their failure, to
Example:
Example:
eliminate potential hazards.
–– They
Theyboth
both show
show temperatures
temperatures
across
Where thermal insulation
across the
the surface
surface of
of the
the
images
images
becomes faulty, building
–– But
But there
there isis aa big
big difference
difference
construction technicians can see
between
between them
them based
based on
onthe
the
data
data that
that creates
creates the
the two
two
heat leaks to improve the
images.
images.
efficiencies of cooling or heating
air-conditioning. Thermal
Both
Bothhave
haveTemperature
TemperatureScales
Scaleswith
withassociated
associatedcolor
colorpallets
pallets
imaging cameras are also
installed in some luxury cars to
aid the driver, the first being the 2000 Cadillac DeVille. Some physiological activities, particularly
responses, in human beings and other warm-blooded animals can also be monitored with thermographic
imaging.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 52
The Professional Thermographers Association
The appearance and operation of a modern thermographic camera is often similar to a camcorder.
Enabling the user to see in the infrared spectrum is a function so useful that ability to record their output is
often optional. A recording module is therefore not always inbuilt.
Instead of CCD sensors, thermal imaging cameras use microbolometer arrays. Their resolution is
considerably lower than of optical cameras, mostly only 160x120 or 320x240 pixels. Thermographic
cameras are much more expensive than their visible-spectrum counterparts, and higher-end models are
often export-restricted. Older bolometers or more sensitive models require cooling; only the modern
uncooled arrays allow construction of small, rugged handheld devices.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 53
The Professional Thermographers Association
Thermographic camera
A thermographic camera, sometimes called a FLIR (Forward Looking Infra-Red), or an infrared camera
less specifically, is a device that forms an image using infrared radiation, similar to a common camera that
forms an image using visible light. Instead of the .45–.75 micron range of the visible light camera, infrared
cameras operate in wavelengths as long as 40 microns.
Theory of operation
All objects emit a certain amount of black-body radiation as a function of their temperatures. Generally
speaking, the higher an object's temperature is, the more infrared radiation as black-body radiation it emits.
A special camera can detect this radiation in a way similar to an ordinary camera does visible light. It works
even in total darkness because ambient light level does not matter. This makes it useful for rescue
operations in smoke-filled buildings and underground.
Images from infrared cameras tend to be monochromatic because the cameras are generally designed with
only a single type of sensor responding to single wavelength range of infrared radiation. Color cameras
require a more complex construction to differentiate wavelength and color has less meaning outside of the
normal visible spectrum because the differing wavelengths do not map uniformally into the system of color
vision used by humans. Sometimes these monochromatic images are displayed in false color, where
changes in color are used rather than changes in intensity do display changes in the signal. This is useful
because although the humans have much greater dynamic range in intensity detection than color overall,
the ability to see fine intensity differences in bright areas is fairly limited.
For use in temperature measurement the brightest (warmest) parts of the image are customarily colored
white, intermediate temperatures reds and yellows, and the dimmest (coolest) parts blue. A scale should be
shown next to a false color image to relate colors to temperatures. Their resolution is considerably lower
than of optical cameras, mostly only 160x120 or 320x240 pixels. Thermographic cameras are much more
expensive than their visible-spectrum counterparts, and higher-end models are often deemed as dual-use
and export-restricted.
Thermal imaging photography finds many other uses. For example, firefighters use it to see through smoke,
find persons, and localize hotspots of fires. With thermal imaging, power line maintenance technicians
locate overheating joints and parts, a telltale sign of their failure, to eliminate potential hazards. Where
thermal insulation becomes faulty, building construction technicians can see heat leaks to improve the
efficiencies of cooling or heating air-conditioning. Thermal imaging cameras are also installed in some
luxury cars to aid the driver, the first being the 2000 Cadillac DeVille. Some physiological activities,
particularly responses, in human beings and other warm-blooded animals can also be monitored with
thermographic imaging.
Types
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 54
The Professional Thermographers Association
Thermographic cameras can be broadly divided into two types: those with cooled infrared image detectors
and those with uncooled detectors.
Cooled infrared detectors
Cooled detectors are typically contained in a vacuum-sealed case and cryogenically cooled. This greatly
increases their sensitivity since their own temperatures are much lower than that of the objects from which
they are meant to detect radiation. Typical cooling temperatures range from 70 K to 110 K, 80 K being the
most common. Without cooling, these sensors (which detect and convert light in much the same way as
common digital cameras, but are made of different materials) would be 'blinded' or flooded by their own
radiation. The drawbacks of cooled infrared cameras are that they are expensive both to produce and to
run. Cooling and evacuating are power- and time-consuming. The camera may need several minutes to
cool down before it can begin working. Although the components that lower temperature and pressure
make generally bulky and expensive, cooled infrared cameras provide superior image quality compared to
uncooled ones.
Materials used for infrared detection include a wide range of narrow gap semiconductors. Examples:
indium antimonide
indium arsenide
HgCdTe
lead sulfide
lead selenide
Uncooled infrared detectors
Uncooled thermal cameras use sensors that operate at room temperature. Modern uncooled detectors all
use sensors that work by the change of resistance, voltage or current when heated by infrared radiation.
These changes are then measured and compared to the values at the operating temperature of the sensor.
Uncooled infrared sensors can be stabilized to an operating temperature to reduce image noise, but they
are not cooled to low temperatures and do not require bulky, expensive cryogenic coolers. This makes
infrared cameras smaller and less costly. However, their resolution and image quality tend to be lower than
cooled detectors. This is due to difference in their fabricational processes, still limited by available
technology.
Uncooled detectors are mostly based on pyroelectric materials or microbolometer technology.
Ferroelectric focal plane arrays
Texas Instruments, in 1976, began developing a radically new thermal imaging sensor approach. Based on
using the pyroelectric properties of certain ceramic materials, techniques where developed to fabricate twodimensional arrays with enough pixels to provide adequate resolution for all but the most demanding
applications. Available today from Raytheon Commercial Infrared, BST array sensor technology is widely
used in commercial infrared imaging products such as Cadillac’s night vision system, the majority of fire
service cameras and the PalmIR series of hand held cameras.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 55
The Professional Thermographers Association
Basic Sensing Principle
Pyroelectric sensors exhibit internal spontaneous electrical polarization, which can be measured as a
voltage at electrodes placed on opposite faces of a sample of the ferroelectric material. At a constant
temperature of the sample, this internal polarization is neutralized by mobile charges on the surface of the
sample; thus there is no voltage difference measured by the electrodes. If the temperature is suddenly
changes to a new value, the internal polarization will change, resulting in a measurable voltage difference.
Thus pyroelectric detectors based on ferroelectric materials do not have a DC signal. The surface
temperature must be modulated in time. This is usually accomplished by means of a radiation chopper. The
detector’s output becomes an alternating current in the external circuit connected to opposite faces of the
detector
BST Sensor and Chopper Assembly
Advantages of AC Coupled Systems
Greatly reduced fixed pattern noise.
Microbolometer based system typically
have significant fixed pattern noise that
appears as rows or columns. Due to sensor signal drift an offset calibration must be performed
periodically based on a number of factors. A chopper-based system effectively continuously
performs this same function providing an image with very uniform flat output.
Ambient temperature drift immunity. All uncooled thermal detector based systems are unshielded,
meaning that up to 80% of the infrared radiation that reaches the detector elements comes from
the lens housing and other non scene contributions. Any temperature change in the camera
creates something similar to a DC level drift. Since the ferroelectric systems are AC coupled there
is no need to perform any ambient temperature offset correction. This attribute has been a critical
discriminator in the selection by automotive companies to use the ferroelectric uncooled thermal
imaging technology in their cars.
BST Focal Plane Array Fabrication
A pixel made from a crystal of ferroelectric material
such as the barium strontium titanate (BST) found in
Raytheon PalmIR500 is hybridized to a CMOS
multiplexer circuit mounted which is in contact with a
thermal electric cooler/stabilizer. The devices
temperature set point is set close to the BST crystal’s
Curie temperature, or the narrow temperature band at
which the crystal material exhibits the highest
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 56
The Professional Thermographers Association
polarization relative to the detector’s temperature change resulting from scene illumination.
The pyroelectric effect is an electrical polarization that can be measured as a transient electrical charge on
opposite faces of the crystal element. As the device reaches temperature equilibration, neutralization of the
surface charge occurs. This requires the infrared radiation illuminating the infrared pixel to be modulated
through the use of a chopper mechanism. The detector is hybridized to a silicon multiplexer circuit that
samples the differential signals and the camera’s electronics generate the video/digital signal seen in the
viewfinder or on the color LCD display
Microbolometer focal plane arrays
Starting in the mid-1980s scientists at Honeywell Research
Center under a classified development contract began work
on a new class of infrared sensors. Until this time infrared
sensors were photon-sensing devices that converted infrared
energy or photons into electrons. Photon detectors typically
operate at very cold temperatures and rely on costly cryogenic
refrigeration units to achieve –200 C operating temperatures.
While new photon type infrared sensors were being
developed that operated at elevated temperatures that solidstate thermal electric coolers could achieve, those devices
only achieved spectral response in the short and mid
wavelength regions. The US military determined that for most night vision applications the preferred
wavelength region was 7-14 microns
Scanning Electron Microscope Image of microbolometer array elements
Sensing temperature using a Bolometer is a well-understood technique and is the basis of a radical new
design that relies on a new silicon processing called MEMS or micro-electro-mechanical-systems. Starting
with a silicon wafer, CMOS multiplexer circuitry is fabricated by a commercial silicon foundry. The infrared
detector foundry then deposits successive layers of silicon nitride and silicon dioxide to fabricate a bridge
structure only 50 microns across. A layer of amorphous silicon or vanadium oxide is used to form a thin film
resistor over the surface of the bridge deck. Integral thin legs are released from the successive layers of
SiO2, which provides structural support of the detector and electrical contact between the CMOS
multiplexer and a thin film resistor deposited on the bridge deck. Once all the layers necessary to build the
complete pixel structure have been deposited a gas is used to dissolve away the sacrificial materials and a
freestanding suspended bridge is left standing. The wafer with individual detector die is tested and good
parts are diced and packaged into a vacuum package.
It is difficult to image just how little mass each of these detector elements really have. As the detector
absorbs photon energy, its surface heats up proportional to the incident radiation falling on it. A voltage or
current is passed across the detector and the signal sampled. The device is so sensitive that a temperature
change of only 50mk on an object provides enough signal change to create an image. The signals are
processes by the integrated circuit readout and an image is created through signal processing.
The performance of a microbolometer is determined by 5 design considerations:
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 57
The Professional Thermographers Association
1. Readout noise. CMOS devices exhibit some level of noise and maybe a significant contributing
factor to device performance.
2. The thermal coefficient of resistance (TCR), which is a measure of the change in resistance as a
function of temperature change.
3. Thermal isolation of the active device from the substrate.
4. The vacuum level in the sealed detector package. This eliminates the convective transfer of energy
between pixels.
5. Detector time constant. Even though the detector must be thermally isolated it must be able to
transfer some energy to the substrate so that the device doesn’t heat up due to constant radiation
impinging on it. Most designs try to optimize the time constant to provide high sensitivity and
integration time of approximately 10 milliseconds.
Key Features :
1. Monolithic device – Detectors are built directly on a CMOS wafer.
2. MEMS construction techniques utilized.
3. Good instantaneous dynamic range.
4. Wafer level packaging (Amorphous Silicon Technology) reduces costs.
5. Rugged (after packaging).
6. High vacuum packaging required (10-6 Torr).
7. Scalable (multiple formats available).
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 58
The Professional Thermographers Association
Basic Camera Operation
Infrared cameras provide you with an image based on the about of infrared radiation that you are viewing
through the camera. There are some basic concepts that need to be understood to be able to get a good
image with the camera. Many of them are based on standard fundamentals of photography, and others are
very specific to thermography. It is very important to understand that it takes time to develop good camera
skills, and that the focus of this section is based on the understanding just like in the photographic industry,
there are amateurs and professionals, we will focus on understand the professional camera skill sets and
terminology that you need to know.
Field of View (FOV)
The Field of View is the horizontal and vertical area that is viewed through your camera. It is defined by the
lens that you have on your camera, usually in degrees horizontally and vertically. It will establish how much
over all area you can actually see at a specific distance.
It is important to know what the field of view is of your cameras lens so that you can match the correct lens
to the application. Having the wrong lens can mean that the images and temperature measurements that
you are getting are completely wrong.
Instantaneous Field of View (IFOV)
The Instantaneous Field of View is the Field of View of one detector pixel. Is considerably smaller then the
FOV of the lens. IFOV is usually measured in “milliradians” or (one thousandth of a radian. A radian is the
measure of a central angle subtending an arc equal in length to the radius: equal to 57.2958° ). The size of
the IFOV is very important because it defines exactly how much detail in the target that you are viewing that
you can perceive, and to what degree of accuracy you can make a temperature measurement.
Regions of Interest (ROI)
The term ROI or Regions of Interest refers to areas or subjects in the image that you are paying specific
attention to. They represent a specific region in the image, disregarding what ever is in the rest of the
image. ROI can be used very broadly and genetically or can be very specific depending on the usage. If
you are looking at a hot and cold cup of water with your IR camera. The RIO may be just the hot cup, and
you are completely disregarding the image of the cold cup, or vise versa.
Some Software programs refer to analysis tools like spot measurement, line profile, or areas as ROI’s
because you would use one of these tools to measure the temperature of some…region of interest to
you…in the image.
Lenses
Many IR cameras come with options for additional lens. Some require you to take off the basic lens that
came with the IR camera, and others piggy-back on to the existing lens that is part of the camera. Unlike
typical visual cameras that use glass as a optical lens, IR Lens material is typically made up of germanium
or silicon, that has been specifically coated to increase their transparency in the IR wave lengths. One of
the biggest expenses in making IR cameras today is in the cost of lenses, and therefore you must take
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 59
The Professional Thermographers Association
extremely good care of them. A typical lens for a IR camera can easily cost from $3,000.00 to $15,000.00
US each.
Knowing the limitations of your camera lens is very important. Knowing what the minimum and maximum
focal distance from or to a object is, what the FOV is, and what effect it will have on your IFOV will all come
in to play when it comes to selecting the correct lens / lenses for the application. Remember that there is a
big difference between using “Zoom” electronic magnification on your camera, vs. using the correct
telephoto lens.
Lenses range from wide angle (approximately 40 to 30 degrees FOV) to standard (approximately 30 to 20
degrees) to telephoto (approximately 20 to 10) to extreme telephoto (10 to 2 degrees FOV).
Tip: When using a extreme telephoto it is very useful to use some type of camera / lens stabilization to
avoid un-necessary shaking and waving of the FOV through the camera.
Tip: When cleaning your lens it is very important to be very careful of the IR antireflective coating on the
surface of the lens. It is very easy to scratch the coating when you are trying to clean it. It is best to use
long grain cotton (the type of cotton that you buy in a box that is roll up in paper), and a mild solution of
“Dawn” dishwashing detergent and water. Never scrub the lens, instead you should dab the lens with the
cotton and Dawn/water solution to soften and lift the dirt on the lens. Then rinse with cotton and water.
When you are in a very dirty location it may be advisable to protect your lens with a piece of polyethylene
film, or a plastic shopping bag or garbage bag, since they are semi-transparent to IR. This way you will
avoid getting your lens dirty in the first place. (Note: you may have to re-calibrate your measurements to
accommodate for the attenuation of the plastic bag that you are trying to image / measure through.)
Size of the IR detector
Today’s cameras are based on a two dimensional array of infrared detectors that are typically called Focal
Plane Arrays or FPA’s. They come in many different sizes, 160 x120, 320 x 240, 640 x 480, etc. Each
array is a matrix of individual IR detector pixels. It is a over simplification to say that the more pixels, the
better the resolution of the camera, as a generalization this rule of thumb holds true. But what you must
remember is, what is the FOV of the lens and the IFOV of the individual detector when placed on the target
that you are trying to image/measure, will define what your requirements are for ultimate resolution for the
application. A camera with a 640 by 480 detector and a wide FOV lens will have a larger IFOV then a 320
by 240 with a telephoto lens ( the smaller detector / lens combination will out perform the detector with
more pixels). Its all about matching the detector, lens and application requirements to get the desired
results.
Telephoto lens vs. Zoom (Electronic Magnification)
When you are using, for example a 320 x 240 detector, with a 20 Degree FOV lens, then you are dividing
the horizontal FOV up in to 320 individual pixels. If you are at a distance of 75 feet, then each pixel is
seeing a average of an area of approximately 1 inch across. If you use the “Zoom” function on your
camera then it only electronically magnifies the pixel, making it appear bigger on your display, it does not
actually increase your resolution, it just blows up the image.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 60
The Professional Thermographers Association
In contrast if you were to change the lens to a 10 degree FOV lens then you would be dividing up the 10
degree horizontal FOV in to 320 pixels, which would equal a IFOV of ½ inch, or resulting in better resolution
on the target.
When ever possible it is important if you want to get the best resolution to start with the larges detector size
that you can reasonably use, matched with the correct lens for the distance that you will be working at for
the specific application so that your FOV and IFOV is optimized as much as possible for the target that you
are trying to image/measure.
Focus (optical focus)
Many of the cameras today come with the ability to focus the camera either by rotating the lens that is in
front of the camera, or is motor driven internally by using the cameras controls, and some cameras can
even adjust the focus of the camera automatically. Focusing is one of the most important skill sets that you
must master if you are going to get a good thermal image. Once a image is stored, you can not change the
optical focus if you got it wrong in the filed. It is best to “Freeze, preprocess, and review” the image before
you actually store the image so that you know that it is the one that you want when you get back to your
office. If it is not in focus, then you can un-freeze the image in the camera and try again to get it right. One
of the important aspects of your camera with regards to focusing your camera on the subject is to know
what your cameras depth of field is (the amount of area that is in focus in the image) so that you can make
sure to know the limits of your camera and lens.
Using the Zoom on the IR camera to help you focus the camera on the subject is very helpful in being able
to actually see the degree of focus, or out of focus you actually are. It is best to try to focus the camera as
best as possible, then zoom in on the subject and fine tune your focus, next zoom back out and then store
your image.
Depth of field (depth of focus)
It will surprise you to know that the depth of filed is very shallow / thin with most IR cameras. This is even
further complicated by the fact that there are not a lot of pixels / low resolution in the first place to be able to
see just how narrow the depth of filed is, since the image is by most visual camera standards very low
resolution in the fist place. The depth of field may only be a few inches deep, requiring you to take several
images of a subject if there are a number of measurements you need to make if the subject runs away from
you.
Remember: if it is not in focus, then you can not measure it correctly, just because it is in the FOV, does not
mean that you can measure it correctly, Try to place your camera as close to a 90 degree angle as possible
(perpendicular to the surface of the subject) if you need to get measurements across the surface of the
object, or you will need to take several images, each with a different focus.
Freeze / Run - Live
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 61
The Professional Thermographers Association
The Freeze / Run – Live feature of a IR camera can be a very useful tool in your IR camera. It allows you
to Freeze a IR image and review it, show it to someone else, and decide if it is the one that you want to
store.
Tip: Sometime it is not easy to get a good shot of the subject because of the actual physical position that
you need to be in to get a good angle of the subject. It is best to frame the FOV the way you want the
image, make sure that you are in optical focus on the subject in the image, and then Freeze the image.
Once that the image is Frozen, you can adjust the level and span/sensitivity, color pallet, and analysis tools
in a more comfortable position once you have frozen the image..
Dialing in the “IR Window”
The IR camera presents to the thermographer a image based on the total radiance from the objects that
are being viewed. For the sake of keeping things simple at this point, let’s say that this image window has
a minimum and maximum temperature limits that the camera allows you to view, (you view everything in
between the minimum and maximum temperature limits. Getting a good image is a lot like trying to dial in a
old black and white TV when you were working with the balance and gain. You had to just dial in the best
image you could get given the picture in the screen. When working with a IR camera, you will need to do
the same thing manually to get the best IR image.
AUTO
Many cameras have a AUTO feature that will try to set the min and max for you automatically, but it is most
of the time not the best settings to get you the best image of the subject that you are looking at. What
happens is there is a auto histogram algorithm that runs when you press the “Auto” button, that looks at the
total radiance in the FOV and sets the span between the minimum and maximum for you. This may be
advantageous if you are looking a furnace wall, the side of a building, electrical motor and pump bearings
because it will get you in the ball park, but you will need to still manually fine tune the IR window to be able
to get a good image of your subject.
On electrical equipment, only use the auto to set the camera up to what you are looking at if the IR window
is “way out of whack”. Otherwise don’t even thinking about touching it! Especially for any kind of out door
utility work. The reason for this is because in electrical panels you have a lot of equipment that runs very
hot, as well as highly reflective surfaces that will look very cool, or very hot depending on what is in the
background. Using the Auto feature will set the span between your minimum and maximum to wide for you
to find a problem, and when you are performing outdoor inspections of substations and transmission,
distribution circuits you will have the very cold sky in the background (-40 F/C), that will set your minimum
to low. The best bet is to learn to intuitive use your Level, Span and Sensitivity controls.
Level (center point)
Level represents the center point between your minimum and maximum on your span. If you have a IR
window that is from 50 to 100 degrees F, then you have a span of 50 degrees and your Level is at 75
degrees. Adjusting your level will keep the 50 deg span, but will move it up or down depending on what
direction you want it to go.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 62
The Professional Thermographers Association
When working with a gray scale pallet anything that appears white will be greater then your IR window or
maximum, and anything that is black is less then your IR window or minimum. It is best to think of this as
an “IR compass” to our IR window, it will always tell you which way to move your Level. If the image is all
black, then everything that you are trying to view is colder then what your setting are and you need to move
the level….DOWN. If everything is white, then you are trying to view things that are warmer then your IR
window, and you need to move your level…UP.
Span / Sensitivity / Gain (Minimum – Maximum of your window)
The Span represents the difference between your minimum and maximum settings on your camera. If you
are looking at a hot cup of water that is 150 deg. F and the table that it is sitting on is 70 deg. F. And you
want to image the cup of hot water and the table, then you would want a span of 80 deg. so that you can
see both. But if all you wanted to see was the cup of hot water (your ROI), and the cup itself is a little bit
cooler then the hot water ( lets say that the cup is 140, and the water is 150, then you would want a span of
only 10 degrees.
Your span is directly associated with your sensitivity. If you have too wide of a span between the min and
max, then you will not have as much sensitivity to see the detail that you are looking for in your ROI. If your
span is to small, then you will have good sensitively, but you will need to raise or lower the level, to be able
to see all of the detail on your ROI,
Key point: when scanning it is best to have the span a little bit narrow, which will increase your sensitivity
and keep adjusting your level accordingly, to get the most information out of what you are scanning.
When working with a gray scale pallet, if everything is too gray then you must decrease the width of your
span, and increase your sensitivity. If everything in the image has to much contrast and you are having to
radically adjust your level, then you need to increase the width of your span/decrease your sensitivity to get
a better image.
Learning to work your level and span is one of the most important camera skills for you to learn!
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 63
The Professional Thermographers Association
Thermodynamics
Thermodynamics (Greek: thermos = heat and dynamis =
power) is the physics of heat, work, enthalpy, and entropy
changes in relation to the spontaneity of processes. In origins,
thermodynamics is the study of engines. Prior to 1698, with
the invention of the Savery Engine, horses were used to
"power" pulleys, attached to buckets, which lifted water out of
flooded salt mines in England. In the years to follow, more
variations of steam engines were built; as the Newcomen
Engine, and later the Watt Engine. In time, these early
engines would eventually be utilized in place of horses. Thus, each engine began to be associated with a
certain amount of "horse power" depending upon how many horses it had replaced! The main problem with
these first engines was that they were slow and clumsy, converting less than 2% of the input fuel into useful
work. In other words, large quantities of coal (or wood) had to be burned to yield only a small fraction of
work output. Hence the need for a new science of engine dynamics was born. Thermodynamics are the
laws which form the basis for cosmology, and several other fields of Science.
Overview
Thermodynamics, at present, designates the science of all transformations of matter and energy.
Definitively, thermodynamics can be divided into two main branches:
o
o
Equilibrium Thermodynamics: the study of systems as they approach equilibrium.
Classical Thermodynamics – macroscopic analysis of systems
Statistical Thermodynamics – microscopic analysis of systems
o
o
Non-equilibrium Thermodynamics: the study of systems away from equilibrium.
Near-equilibrium Thermodynamics – linear analysis of irreversible processes.
Far-from-equilibrium Thermodynamics – nonlinear analysis of irreversible processes.
From this base, over the years, other variations of thermodynamics have come into their own as: chemical
thermodynamics, thermal physics, biological thermodynamics, atmospheric thermodynamics, economic
thermodynamics, environmental thermodynamics, black hole thermodynamics, and others.
While dealing with processes in which systems exchange matter or energy, classical thermodynamics is
not concerned with the rate at which such processes take place, termed kinetics. For this reason, the use of
the term "thermodynamics" usually refers to equilibrium thermodynamics. In this connection, a central
concept in thermodynamics is that of quasistatic processes, which are idealized, "infinitely slow" processes.
Time-dependent thermodynamic processes are studied by non-equilibrium thermodynamics.
History
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 64
The Professional Thermographers Association
Most cite Sadi Carnot’s 1824 paper Reflections on the Motive Power of Fire as the starting point for
thermodynamics as a modern science. Carnot defined "motive power" to be the expression of the useful
effect that a motor is capable of producing. Herein, Carnot introduced us to the first modern day definition
of "work": weight lifted through a height. The desire to understand, via formulation, this useful effect in
relation to "work" is at the core of all modern day thermodynamics.
The name "thermodynamics", however, did not arrive until some twenty-five years later when in 1849, the
British mathematician and physicist William Thomson (Lord Kelvin) coined the term ‘thermodynamics' in a
paper on the efficiency of steam engines. In 1850, the famed mathematical physicist Rudolf Clausius
originated and defined the term enthalpy H to be the total heat content of the system, stemming from the
Greek word ‘enthalpein’ meaning to warm, and defined the term entropy S to be the heat lost or turned into
waste, stemming from the Greek word ‘entrepein’ meaning to turn.
In association with Clausius, in 1871, a Scottish mathematician and physicist James Maxwell formulated a
new branch of thermodynamics called Statistical Thermodynamics, which functions to analyze large
numbers of particles at equilibrium, i.e. systems where no changes are occurring, such that only their
average properties as temperature T, pressure P, and volume V become important.
Soon thereafter, in 1875, the Austrian physicist Ludwig Boltzmann formulated a precise connection
between entropy S and molecular motion:
being defined in terms of the number of possible states [W] such motion could occupy, where k is the
Boltzmann's constant. The following year, 1876, was a seminal point in the development of human thought.
During this essential period, chemical engineer Willard Gibbs, the first person in America to be awarded a
PhD in engineering (Yale), published an obscure 300-pg paper titled: On the Equilibrium of Heterogeneous
Substances, wherein he formulated one grand equality, the Gibbs free energy equation, which gives a
measure the amount of "useful work" attainable in reacting systems:
Building on these foundations, those as Lars Onsager, Erwin Schrodinger, and Ilya Prigogine, and others,
functioned to bring these engine “concepts” into the thoroughfare of almost every modern-day branch of
science.
The laws of thermodynamics
In Thermodynamics, there are four laws of very general validity, and as such they do not depend on the
details of the interactions or the systems being studied. This means they can be applied to systems about
which one knows nothing other than the balance of energy and matter transfer with the environment.
Examples of this include Einstein's prediction of spontaneous emission around the turn of the 20th century
and the current research into the thermodynamics of black holes. Alternative statements that are
mathematically equivalent can be given for each law, as follows:
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 65
The Professional Thermographers Association
Zeroth law (Thermodynamic equilibrium): If systems A and B are in thermodynamic equilibrium, and
systems B and C are in thermodynamic equilibrium, then systems A and C are also in thermodynamic
equilibrium.
When two systems are put in contact with each other, there will be a net exchange of energy and/or matter
between them unless they are in thermodynamic equilibrium. Two systems are in thermodynamic
equilibrium with each other if they stay the same after being put in contact.
While this is a fundamental concept of thermodynamics, the need to state it explicitly as a law was not
perceived until the first third of the 20th century, long after the first three laws were already widely in use,
hence the zero numbering. There is still some discussion about its status.
Thermodynamic equilibrium includes thermal equilibrium (associated to heat exchange and parameterized
by temperature), mechanical equilibrium (associated to work exchange and parameterized generalized
forces such as pressure), and chemical equilibrium (associated to matter exchange and parameterized by
chemical potential).
1st Law (Conservation of energy): The work exchanged in an adiabatic process depends only on the initial
and the final state and not on the details of the process.
This is equivalent to a statement of the conservation of energy, because no heat flows during an adiabatic
process. This means that the only energy flowing into or out of a system during an adiabatic process is
work done on or by the system.
This law is equivalent to
where U is the internal energy of a system, Q is the heat
flowing into the system, and W is the work done on the system. The energy received by the system is
positive.
2nd Law (Entropy): It is impossible to obtain a process that, operating in cycle, produces no other effect
than the subtraction of a positive amount of heat from a reservoir and the production of an equal amount of
work. (Kelvin-Planck Statement)
The entropy of a thermally isolated macroscopic system never decreases (see Maxwell's demon), however
a microscopic system may exhibit fluctuations of entropy opposite to that dictated by the second law (see
Fluctuation Theorem). In fact the mathematical proof of the Fluctuation Theorem from time-reversible
dynamics and the Axiom of Causality, constitutes a proof of the Second Law. In a logical sense the Second
Law thus ceases to be a "Law" of Physics and instead becomes a theorem which is valid for large systems
or long times.
3rd Law (Absolute Zero): As temperature goes to 0, the entropy of a system approaches a constant.
It is important to remember that the laws of thermodynamics are only statistical generalizations. That is,
they simply describe the tendencies of macroscopic systems. On the quantum level, the laws of
thermodynamics often break down. Furthermore, as evidenced by Maxwell's demon, it is theoretically
possible to specifically engineer a quantum system to break the laws of thermodynamics. The first law of
thermodynamics, however, i.e. the law of conservation, has become the most sound of all laws in science.
Its validity has never been disproved.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 66
The Professional Thermographers Association
Conservation of energy
.
Conservation of energy, also known as the first law of
thermodynamics, is possibly the most important, and certainly the most
practically useful of several conservation laws in physics.
Laws of thermodynamics
Zeroth law of thermodynamics
The law states that the total inflow of energy into a system must equal
the total outflow of energy from the system, plus the change in the
energy contained within the system. In other words, energy can be
converted from one form to another, but it cannot be created or
destroyed.
First law of thermodynamics
The law of conservation of energy excludes the possibility of perpetuum
mobile of the first kind.
Philosophy of thermal and
statistical physics
Second law of thermodynamics
Third law of thermodynamics
.
Conservation Law
Conservation Law and 1st Law Of Thermodynamics
• Conservation, i.e. accounting for all of the
constituents of a system.
• The law explains that everything in a system
must be accounted for because nothing can just
“pop” into existence from nothing, and that it can
not just disappear out of existence, but must in
its basic elements still be there, or be changed
into something else.
• This law applies to both mater and energy!
When Infrared Energy hits a object it is…
– Remember E=mc2
Absorbed (A)
into the object
Reflected (R)
off of the object
Transmitted (A)
through the object
Infrared Energy
Infrared Energy = A + R + T
“Conservation”, i.e. accounting for all of the constituents of a system.
1st Law Of Thermodynamics :
Energy cannot be created or destroyed, but must be conserved or exactly accounted for. It
can be changed from one form to another. Heat that flows into an object must go somewhere.
Energy in = Energy out + Energy stored
The law explains that everything in a system must be accounted for because nothing can just “pop” into
existence from nothing, and that it can not just disappear out of existence, it must be in its basic elements still
there, or be changed into something else. This law applies to both mater and energy since they are both the
same thing….E=mc2
Copyright 2005 Professional Thermographers Association All rights Reserved www.ProThermographer.com
Copyright 2005 Professional Thermographers Association All rights Reserved www.ProThermographer.com
Historical development
Although ancient philosophers as far back as Thales of Miletus had inklings of the first law, it was the
German Gottfried Wilhelm Leibniz during 1676-1689 who first attempted a mathematical formulation.
Leibniz noticed that in many mechanical systems (of several masses, mi each with velocity vi) the quantity:
was conserved. He called this quantity the vis viva or living force of the system. The principle represents an
accurate statement of the approximate conservation of kinetic energy in many situations. However, many
physicists were influenced by the prestige of Sir Isaac Newton in England and of René Descartes in
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 67
The Professional Thermographers Association
France, both of whom had set great store by the conservation of momentum as a guiding principle. Thus
the momentum:
∑ mivi
i
was held by the rival camp to be the conserved vis viva. It was largely engineers such as John Smeaton,
Peter Ewart, Karl Hotzmann, Gustave-Adolphe Hirn and Marc Séguin who objected that conservation of
momentum alone was not adequate for practical calculation and who made use of Leibniz's principle. The
principle was also championed by some chemists such as William Hyde Wollaston.
Members of the academic establishment such as John Playfair were quick to point out that kinetic energy is
clearly not conserved. This is obvious to a modern analysis based on the second law of thermodynamics
but in the 18th and 19th centuries, the fate of the lost energy was still unknown. Gradually it came to be
suspected that the heat inevitably generated by motion was another form of vis viva. In 1783, Antoine
Lavoisier and Pierre-Simon Laplace reviewed the two competing theories of vis viva and caloric[1]. Count
Rumford's 1798 observations of heat generation during the boring of cannons added more weight to the
view that mechanical motion could be converted into heat. Vis viva now started to be known as energy,
after the term was first used in that sense by Thomas Young in 1807.
The recalibration of vis visa to
was largely the result of the work of Gaspard-Gustave Coriolis and Jean-Victor Poncelet over the period
1819-1839. The former called the quantity quantité de travail and the latter, travail mécanique and both
championed its use in engineering calculation.
In a paper Uber die Natur der Warme, published in the Zeitschrift für Physik in 1837, Karl Friedrich Mohr
gave one of the earliest general statements of the doctrine
of the conservation of energy in the words: "besides the 54
known chemical elements there is in the physical world one
agent only, and this is called Kraft [energy]. It may appear,
according to circumstances, as motion, chemical affinity,
cohesion, electricity, light and magnetism; and from any
one of these forms it can be transformed into any of the
others."
A key stage in the development of the modern conservation
principle was the demonstration of the mechanical
equivalent of heat. The caloric theory maintained that heat
could neither be created nor destroyed but conservation of
energy entails the contrary principle that heat and
mechanical work are interchangeable.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 68
The Professional Thermographers Association
The mechanical equivalence principle was first stated in its modern form by the German surgeon Julius
Robert von Mayer.[2] Mayer reached his conclusion on a voyage to the Dutch East Indies, where he found
that his patients' blood was a deeper red because they were consuming less oxygen, and therefore less
energy, to maintain their body temperature in the hotter climate. He had discovered that heat and
mechanical work were both forms of energy, and later, after improving his knowledge of physics, he
calculated a quantitative relationship between them.
Joule's apparatus for measuring the mechanical equivalent of heat
Meanwhile, in 1843 James Prescott Joule independently discovered the mechanical equivalent in a series
of experiments. In the most famous, now called the "Joule apparatus", a descending weight attached to a
string caused a paddle immersed in water to rotate. He showed that the gravitational potential energy lost
by the weight in descending was equal to the thermal energy (heat) gained by the water by friction with the
paddle.
Over the period 1840-1843, similar work was carried out by engineer Ludwig A. Colding though it was littleknown outside his native Denmark.
Both Joule's and Mayer's work suffered from resistance and neglect but it was Joule's that, perhaps
unjustly, eventually drew the wider recognition.
For the dispute between Joule and Mayer over priority, see
Mechanical equivalent of heat: Priority
Drawing on the earlier work of Joule, Sadi Carnot and Émile Clapeyron, in 1847, Hermann von Helmholtz
postulated a relationship between mechanics, heat, light, electricity and magnetism by treating them all as
manifestations of a single force (energy in modern terms). He published his theories in his book Über die
Erhaltung der Kraft (On the Conservation of Force, 1847). The general modern acceptance of the principle
stems from this publication.
In 1877, Peter Guthrie Tait claimed that the principle originated with Sir Isaac Newton, based on a creative
reading of propositions 40 and 41 of the Philosophiae Naturalis Principia Mathematica. This is now
generally regarded as nothing more than an example of Whig history.
Modern physics
With the discovery of special relativity by Albert Einstein, mass and energy were shown to be
interchangeable by the famous equation E = mc2. As a result, the rule of conservation of energy was shown
to be a special case of a more general rule, the conservation of mass and energy, which is now usually just
referred to as conservation of energy.
Mathematical formulations
One formulation for the first law of thermodynamics is
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 69
The Professional Thermographers Association
Q = ΔU + W
(1)
where Q is heat transferred into the system from the surroundings, W is work done by the system, and
U is the internal energy of the system. This energy is mostly kinetic energy: the potential energy can be
assumed to be negligible. Pressure-volume work (e.g. done by a gas on a piston) is defined to be
W = PΔV.
(2)
Equation (1) can be interpreted as follows: Q is heat energy being input into the system. The system
then can use this incoming energy to do two things: (1) do work, or (2) increase its own internal energy.
Here is an analogy: Q is income, which can then be spent to buy things (W), or it can be saved in a bank
account (ΔU).
If all the heat is used to do work (Q = W and ΔU = 0) then the system is undergoing an isothermal process,
which means that its temperature remains constant. This is because the system's internal energy is
proportional to its temperature.
If all the heat is used to increase internal energy, (Q = ΔU and W = 0) then the system is undergoing an
isochoric process, also called isometric process. This is a process in which the system's volume is
constant: ΔV = 0 so that, according to equation (2), W = 0.
It is also possible for the heat energy to be used up partially by doing work and partially by increasing
internal energy. An example of such processes is the isobaric process.
Equation (1) is the one preferred by engineers. Another form preferred by chemists is
ΔU = Q + W
(3)
where W is work done on the system by the surroundings. In this case pressure-volume work is defined
to be
W = − PΔV.
(4)
Equation (3) can be interpreted to mean that heat Q and work W are energies being transferred into or
out of the system. The system then responds by increasing its internal energy accordingly. In Equation (3)
neither Q nor W are state functions. A state function does not depend on which particular thermodynamic
process is chosen to connect the initial and final thermostatic states.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 70
The Professional Thermographers Association
Second law of thermodynamics
.
The second law of thermodynamics is a law of thermodynamics that
states that all work tends towards the production of greater entropy over
time. Another way of saying this (known as the Clausius formulation) is
that it is impossible to construct a perfect refrigerator. (This is why
refrigerators always require an external power source.) An equivalent
statement, known as the Kelvin-Planck formulation, is that "It is
impossible for any cyclic process to occur whose sole effect is the
extraction of heat from a reservoir and the performance of an equivalent
amount of work." (If the reader is aware of the concept of heat engines,
please understand that heat engines work by allowing two heat
reservoirs of different temperatures to come in equilibrium with each
other, and as such they do not violate the 2nd law.)
(Note: the second law of thermodynamics is not a result of the
expansion of the universe - in fact it applies to any closed system.)
Laws of thermodynamics
Zeroth law of thermodynamics
First law of thermodynamics
Second law of
thermodynamics
Third law of thermodynamics
Philosophy of thermal and
statistical physics
.
In physics, the second law of thermodynamics, in its many forms, is a statement about the quality and
direction of energy flow, and it is closely related to the concept of entropy. This law and its derivatives, such
as the law of friction, define the arrow of time: most other physical laws are time-reversal invariant.
General description
The first law of thermodynamics states that one form of energy (such as kinetic, potential, electrical,
thermal, etc,) can be converted into another without loss. The second law states that thermal energy (heat)
is special: all forms of energy can be converted into heat, but it is not possible to convert the heat back fully
in its original form. In other words, heat is a form of energy of lower quality.
What makes heat so special? According to the kinetic theory, heat is due to the random movement of
atoms and molecules, so it looks much like kinetic energy. The difference is that those movements of
atoms and molecules that produce heat cannot be observed or predicted, while all the other forms of
energy are the result of some orderly movement of particles. The second law says that the amount of
random movement (entropy) can only increase in a closed system; in other words, the randomness cannot
be ordered without some external influence. (Some systems, such as living cells, spontaneously become
structured when they receive energy from the outside. See dissipative structures.)
The following example illustrates the law:
When a stone falls on earth, its kinetic energy is converted into heat (it becomes random
movements of particles). The second law says that this random movement will never become
ordered again. For example, the random movement will never become synchronized to throw the
stone back in the air: the heat energy cannot revert to the original kinetic energy.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 71
The Professional Thermographers Association
There is one thing predictable about heat: it flows from hot to cold bodies. This can be used to convert
some heat into mechanical energy, using a Carnot heat engine. The cycle stops when both bodies reach
the same temperature (equilibrium); it can be shown that the amount of random movements has not
decreased in the process.
The second law of
thermodynamics is
important in engineering
because it provides a way
to determine the quality
and amount of
degradation of energy
during a process. It is also
used to determine the
theoretical upper limits for
the performance of many
commonly-used
engineering systems,
such as refrigerators,
internal combustion
engines, and reactors.
Many claimed perpetual motion machines would have to violate the second law of thermodynamics to
function. In general, such machines seem to generate energy from "nowhere." In all cases, when
examined, the machine has some hidden mechanism for drawing energy in from the outside. One example
of this would be a device that can do work such as pumping water, simply by taking energy from the air.
Such claims are labeled "perpetual motion machines of the second kind."
History
The first theory on the conversion of heat into mechanical work is due to Nicolas Léonard Sadi Carnot in
1824. He was the first to realize correctly that the efficiency of the process depends on the difference of
temperature between the hot and cold bodies.
Recognizing the significance of James Prescott Joule's work on the conservation of energy, Rudolf
Clausius was the first to formulate the second law in 1850, in this form: heat does not spontaneously flow
from cold to hot bodies. While common knowledge now, this was contrary to the caloric theory of heat
popular at the time, which considered heat as a liquid. From there he was able to infer the law of Sadi
Carnot and the definition of entropy (1865).
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 72
The Professional Thermographers Association
Heat
A red-hot iron rod cooling after being worked by a blacksmith.
Heat (abbreviated Q, also called heat change) is the transfer of thermal energy between two bodies which
are at different temperatures. The SI unit for heat is the joule.
Heat is to thermal energy as work is to mechanical energy. Heat flows between regions that are not in
thermal equilibrium; in particular, it flows from areas of high temperature to areas of low temperature. All
objects (matter) have a certain amount of internal energy that is related to the random motion of their atoms
or molecules. When two bodies of different temperature come into thermal contact, they will exchange
internal energy until the temperature is equalized (This is known as reaching Thermal Equilibruim). The
amount of energy transferred is the amount of heat exchanged. It is a common misconception to confuse
heat with internal energy, but there is a difference: heat is related to the change in internal energy and the
work performed by the system. The term heat is used to describe the flow of energy, while the term internal
energy is used to describe the energy itself. Understanding this difference is a necessary part of
understanding the first law of thermodynamics.
Infrared radiation is often linked to heat, since objects at room temperature or above will emit radiation
mostly concentrated in the mid-infrared band (see black body).
Notation
Sign Convention: When a body releases heat into its surroundings, Q < 0 (-). When a body absorbs heat
from its surroundings, Q > 0 (+).
Total heat, heat transfer rate, and heat flux are all notated with different permutations of the letter Q. They
are often confusingly switched in different contexts.
Total heat is notated as Q, and is measured in joules in SI units.
Heat transfer rate, or heat flow per unit time, is labeled
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 73
The Professional Thermographers Association
to indicate a change per unit time. In Unicode, this is Q̇, though it may not display correctly in all browsers.
It is often shown as ˙Q, .Q, Q·, or as a Q with no dot, where it is not easy to produce a dotted Q. Some form
of dotted Q, such as .Q, is preferable, since undotted Q is used for total heat. It is measured in watts.
Heat flux is defined as amount of heat per unit time per unit cross-sectional area, is abbreviated q, and is
measured in watts per meter squared. It is also sometimes notated as Q″ or q″ or
.
Changes of temperature== The amount of heat energy, ΔQ, required to change the temperature of a
material from an initial temperature, T0, to a final temperature, Tf depends on the heat capacity of that
material according to the relationship:
The heat capacity is dependent on both the amount of material that is exchanging heat and its properties.
The heat capacity can be broken up in several different ways. First of all, it can be represented as a product
of mass and specific heat capacity (more commonly called specific heat):
or the number of moles and the molar heat capacity:
Both the molar and specific heat capacities only depend upon the physical properties of the substance
being heated, not on any specific properties of the sample. The above definitions of heat capacity only work
approximately for solids and liquids, but for gases they don't work at all most of the time. The molar heat
capacity can be "patched up" if the changes of temperature occur at either a constant volume or constant
pressure. Otherwise, it's generally easiest to use the first law of thermodynamics in combination with an
equation relating the internal energy of the gas to its temperature.
Changes of phase
A boiling pot of water, at sea level and normal atmospheric pressure, will always be at 100 °C no matter
how much heat is added. The extra heat changes the phase of the water from liquid into water vapor. The
heat added to change the phase of a substance in this way is said to be "hidden," and thus it is called
latent heat (from a Latin word for hidden). Latent heat is heat per unit mass necessary to change the state
of a given substance. Thus:
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 74
The Professional Thermographers Association
and
where Mo is the amount of mass initially in the new phase, and M is the amount of mass that ends up in the
new phase.
L generally doesn't depend on the amount of mass that changes phase, so the equation can normally be
written:
Sometimes L can be time-dependent if pressure and volume are time-varying, so that the integral can be
handled:
someone check the above, please, to see if the latent heat really depends on where on the (P, V, T) curve
the transition is taking place.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 75
The Professional Thermographers Association
Joule
The joule (symbol: J) is the SI unit of energy, or work. It is named in honor of the physicist James Prescott
Joule (1818–1889).
Definition
The joule is a derived unit defined as the work done,or energy required, to exert a force of one newton for a
distance of one metre, so the same quantity may be referred to as a newton metre or newton-metre (also
with meter spelling), with the symbol N·m or N m. It can also be written as kg·m2·s−2. However, the newton
metre is usually used as a measure of torque, not energy.
One joule is also:
the work required to move an electric charge of one coulomb through an electrical potential
difference of one volt; or one coulomb volt, with the symbol C·V.
the work done to produce power of one watt continuously for one second; or one watt second
(compare kilowatt-hour), with the symbol W·s
Conversions
1 joule is equal to:
10 000 000 erg (ergs)
c. 0.239 cal (calorie) (small calories)
0.0002390 calorie or kilocalorie (food)
4.184 J – 1 thermochemical calorie (exact)
4.1868 J – 1 International Table calorie (exact)
c. 0.000 948 BTU (British thermal unit)
c. 0.738 ft·lbf (foot pound force)
c. 23.7 ft·pdl (foot poundals)
0.0000002778 kilowatt-hour
0.0002778 watt-hour
0.0098692 litre-atmosphere
1 J – The energy required to lift a small apple (102 g) one metre against Earth's gravity
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 76
The Professional Thermographers Association
Multiples
Multiple
Name
Symbol Multiple
Name
Symbol
100
joule
J
101
decajoule daJ
10–1
decijoule dJ
102
hectojoule hJ
10–2
centijoule cJ
103
kilojoule
kJ
10–3
millijoule
106
megajoule MJ
10–6
microjoule µJ
109
gigajoule GJ
10–9
nanojoule nJ
1012
terajoule
TJ
10-12
picojoule pJ
1015
petajoule PJ
10-15
femtojoule fJ
1018
exajoule
EJ
10–18
attojoule
1021
zettajoule ZJ
10–21
zeptojoule zJ
1024
yottajoule YJ
10–24
yoctojoule yJ
mJ
aJ
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 77
The Professional Thermographers Association
British thermal unit
The British thermal unit (BTU or Btu) is a non-metric unit of energy, used in the United States and, to a
lesser extent, the UK (where it is generally only used for heating systems). The SI unit is the joule (J),
which is used by most other countries. A BTU is defined as the amount of heat required to raise the
temperature of one pound avoirdupois of water by one degree Fahrenheit; specifying the temperature
range over which this happens leads to a number of slightly different BTU values, varying over a range of
about 0.5%:
Name
Value (J)
IT
≡ 1055.05585262
39 °F
≈ 1059.67
Mean
≈ 1055.87
59 °F
≡ 1054.804
60 °F
63 °F
ISO
≈ 1054.68
≈ 1054.6
≡ 1054.5
Thermochemical ≡ 1054.35026444
Notes
The most widespread BTU, uses the International [Steam] Table
(IT) calorie, itself defined for water at 14.5 °C (58.1 °F) by the
Fifth International Conference on the Properties of Steam, held in
London in July 1956.
Uses the calorie value of water at its maximum density (39.1–
39.2 °F)
Uses a calorie averaged over the 32 °F to 212 °F range
Chiefly American. Uses the 15 °C calorie, itself defined as
exactly 4185.5 J (Comité international 1950; PV, 1950, 22, 7980)
Chiefly Canadian
Possibly apocryphal
ISO 31 Quantities and units (?)
(Calculated from 9489.152 380 4 ÷ 9) Uses the thermochemical
calorie of exactly 4.184 J
The BTU is often used to describe the heat value of fuels, or the heating and cooling power of a system
(such as a barbecue grill).
Conversions
One BTU is approximately:
252–253 cal (calories, small)
0.252–0.253 food calorie
778–782 ft·lbf (foot-pounds-force)
1054-1060 joules
Associated units
The BTU per hour (BTU/h) is the unit of power most commonly associated with the BTU.
1 horsepower is approximately 2540 BTU/h
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 78
The Professional Thermographers Association
1 watt is approximately 3.4 BTU/h
1000 BTU/h is approximately 293 W
A unit called the quad (short for quadrillion) is defined as 1015 BTU, which is about 1.055×1018 joules, and
the therm is defined in the United States and European Union as 100,000 BTU – but the U.S. uses the
BTU59°F whilst the E.C. uses the BTUIT.
The BTU should not be confused with the Board of Trade Unit (B.O.T.U.), which is a much larger quantity
of energy.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 79
The Professional Thermographers Association
Calorie
A calorie (cal) is a unit of thermal energy, equivalent to about 4.185 J.
By definition, one calorie is the energy needed to increase the temperature of 1 g of water by 1 °C at 15 °C
under normal atmospheric pressure (76 cmHg). In nutrition, it is used to quantify the energy of food and the
defining quantity of water is 1 kilogram, making the "food calorie" 1000 times as large as the other calorie.
Hence, 1 calorie (nutrition) = 1 kcal = 1000 cal, and it is also called the large calorie.
The calorie is not an SI unit where the joule is the only unit of energy. The kilocalorie is frequently used in
chemistry because it is a convenient measure of molar free energy: for example, 1.4 kcal/mol engenders a
change in equilibrium by a factor of 10.
Nutrition
Nutritionists measure the energy content of food today in both "calories" (large calories) or "kilojoules" (kJ),
a usage widely known in the general population and (confusingly) often referred to as a "kilocalorie" as
though small calories were being used. Sporadic attempts have been made, particularly in the United
States, to write the kilogram calorie capitalized as Calorie and abbreviated as Cal, to alleviate the
confusion with the chemical definition of the term "calorie" as a small calorie. This alternate notation has no
legal standing, and is not endorsed by any standards body.
The amount of food energy in a particular food is measured by completely burning the food in a bomb
calorimeter, a method known as direct calorimetry [1]. For example, dietary fat has about 9 kcal/g, while
proteins and carbohydrates have about 4 kcal/g. Recommended daily energy intake values for average
adults are 2500 kcal/d (10 MJ/d) for men and 2000 kcal/d (8 MJ/d) for women.
The "calorie" has become a common household term, because dietitians recommend in cases of obesity to
reduce body weight by increasing exercise (energy expenditure) and reducing energy intake.
Versions
Three definitions of the calorie are today recognized internationally:
Name
15 °C calorie
Symbol
cal15
Conversion factor
1 cal15 = 4.1855 J
Remarks
1 cal15 is the amount of heat
(energy) required to warm 1 g of
air-free water from 14.5 °C to
15.5 °C at a constant pressure
of 101.325 kPa (1 atm). The
conversion factor was published
by the CIPM in 1950 as the
most accurate value that could
be determined by experiment at
the time. It has an uncertainly of
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 80
The Professional Thermographers Association
I.T. calorie
calIT
1 calIT = 4.1868 J
thermochemical calorie
calth
1 calth = 4.184 J
0.0005 J.
The International Table calorie
was adopted by the Fifth
International Conference on
Properties of Steam (London,
July 1956), with 1 McalIT =
1.163 kWh (exactly).
Other historic definitions include
the 4 °C calorie, and
the mean 0 °C to 100 °C calorie.
Of all these, what is most commonly meant by calorie in contemporary English text is the 15 °C calorie.
The nutritional kilocalorie or Calorie equals 1000 cal15.
Since these many definitions are a source of confusion and error, all calories are now deprecated. The
International System of Units (SI) unit for heat (and for all other forms of energy) is the joule (J), while the
(obsolete) CGS system used the erg.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 81
The Professional Thermographers Association
Temperature
Temperature is the physical property of a system which underlies the common notions of "hot" and "cold";
the material with the higher temperature is said to be hotter. Temperature is a measure of the average
kinetic energy of the particles in a sample of matter. In other words, temperature is a measure of activity
and the frequency of collisions of molecules.
Overview
The formal properties of temperature are studied in thermodynamics. Formally, temperature is that property
which governs the transfer of thermal energy, or heat, between one system and another. When two
systems are at the same temperature, they are in thermal equilibrium and no heat transfer will occur.
When a temperature difference does exist, heat will tend to move from the higher-temperature system
to the lower-temperature system, until thermal equilibrium is established. This heat transfer may
occur via conduction, convection or radiation (see heat for additional discussion of the various
mechanisms of heat transfer).
Temperature is related to the amount of thermal energy or heat in a system. As more heat is added the
temperature rises, similarly a decrease in temperature corresponds to a loss of heat from the system. On
the microscopic scale this heat corresponds to the random motion of atoms and molecules in the
system. Thus, an increase in temperature corresponds in an increase in the rate of movement of the atoms
in the system.
Temperature is an intensive property of a system, meaning that it does not depend on the system size or
the amount of material in the system. Other intensive properties include pressure and density. By contrast,
mass and volume are extensive properties, and depend on the amount of material in the system.
Applications
Temperature plays an important role in almost all fields of science, including physics, chemistry, and
biology.
Many physical properties of materials including the phase (solid, liquid, gaseous or plasma), density,
solubility, vapor pressure, and electrical conductivity depend on the temperature. Temperature also plays
an important role in determining the rate and extent to which chemical reactions occur. This is one reason
why the human body has several elaborate mechanisms for maintaining the temperature at 37 °C, since
temperatures only a few degrees higher can result in harmful reactions with serious consequences.
Temperature also controls the type and quantity of thermal radiation emitted from a surface. One
application of this effect is the incandescent light bulb, in which a tungsten filament is electrically heated to
a temperature at which significant quantities of visible light are emitted.
Temperature-dependence of the speed of sound in air c, density of air ρ and acoustic impedance Z vs.
temperature °C
Impact of temperature
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 82
The Professional Thermographers Association
Temperature measurement
Temperature measurement using modern scientific
thermometers and temperature scales goes back at least as far
as the early 18th century, when Gabriel Fahrenheit adapted a
thermometer (switching to mercury) and a scale both
developed by Ole Christensen Rømer. Fahrenheit's scale is still
in use, alongside the Celsius scale and the Kelvin scale.
Units of temperature
T in °C c in m/s ρ in kg/m³ Z in N·s/m³
-10
325.4
1.341
436.5
-5
328.5
1.316
432.4
0
331.5
1.293
428.3
5
334.5
1.269
424.5
10
337.5
1.247
420.7
15
340.5
1.225
417.0
20
343.4 1.204
413.5
The basic unit of temperature (symbol: T) in the
25
346.3 1.184
410.0
International System of Units (SI) is the kelvin (K). One
kelvin is formally defined as 1/273.16 of the temperature of the
30
349.2 1.164
406.6
triple point of water (the point at which water, ice and water
vapor exist in equilibrium). The temperature 0 K is called absolute zero and corresponds to the point at
which the molecules and atoms have the least possible thermal energy. An important unit of temperature in
theoretical physics is the Planck temperature (1.4 × 1032 K).
In the field of plasma physics, because of the high temperatures encountered and the electromagnetic
nature of the phenomena involved, it is customary to express temperature in electron volts (eV) or kilo
electron volts (keV), where 1 eV = 11,605 K. In the study of QCD matter one routinely meets temperatures
of the order of a few hundred MeV, equivalent to about 1012 K.
For everyday applications, it is often convenient to use the Celsius scale, in which 0 °C corresponds to the
temperature at which water freezes and 100 °C corresponds to the boiling point of water at sea level. In
this scale a temperature difference of 1 degree is the same as a 1 K temperature difference, so the scale is
essentially the same as the kelvin scale, but offset by the temperature at which water freezes (273.15 K).
Thus the following equation can be used to convert from Celsius to kelvin.
In the United States, the Fahrenheit scale is widely used. On this scale the freezing point of water
corresponds to 32 °F and the boiling point to 212 °F. The following formula can be used to convert from
Fahrenheit to Celsius:
Comment
Absolute zero
Fahrenheit's ice/salt mixture
Comparison of temperature scales
kelvin¹ Celsius Fahrenheit Rankine Delisle Newton Réaumur Rømer
0
-273.15 -459.67
0
559.725 -90.14² -218.52
135.90
255.37 -17.78 0
459.67 176.67 -5.87 -14.22 -1.83
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 83
The Professional Thermographers Association
Water freezes (at standard
pressure)
Average human body
temperature³
Water boils
Titanium melts
The surface of the Sun
273.15 0
32
491.67 150
0
0
7.5
310.0 36.8
98.2
557.9
12.21
29.6
26.925
373.15 100
1941 1668
5800 5526
212
3034
9980
671.67 0
3494
-2352
10440 -8140
33
550
1823
80
1334
4421
60
883
2909
94.5
¹ Only the kelvin, Celsius, Fahrenheit, and Rankine scales are in use today.
² Some numbers in this table have been rounded off.
³ Normal human body temperature is 36.8 °C ±0.7 °C, or 98.2 °F ±1.3 °F.
Negative temperatures
For some systems and specific definitions of temperature, it is possible to obtain a negative temperature. A
system with a negative temperature is not colder than absolute zero, but rather it is, in a sense, hotter
than infinite temperature (sic).
Articles about temperature ranges:
10−12 K = 1 picokelvin (pK)
10−9 K = 1 nanokelvin (nK)
10−6 K = 1 microkelvin (µK)
10−3 K = 1 millikelvin (mK)
100 K = 1 kelvin
101 K = 10 kelvins
102 K = 100 kelvins
103 K = 1,000 kelvin = 1 kilokelvin (kK)
104 K = 10,000 kelvins = 10 kK
105 K = 100,000 kelvins = 100 kK
106 K = 1 megakelvin (MK)
109 K = 1 gigakelvin (GK)
1012 K = 1 terakelvin (TK)
Theoretical foundation of temperature
Zeroth-law definition of temperature
While most people have a basic understanding of the concept of temperature, its formal definition is rather
complicated. Before jumping to a formal definition, let us consider the concept of thermal equilibrium. If two
closed systems with fixed volumes are brought together, so that they are in thermal contact, changes may
take place in the properties of both systems. These changes are due to the transfer of heat between the
systems. When a state is reached in which no further changes occur, the systems are in thermal
equilibrium.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 84
The Professional Thermographers Association
Now a basis for the definition of temperature can be obtained from the so-called zeroth law of
thermodynamics which states that if two systems, A and B, are in thermal equilibrium and a third system C
is in thermal equilibrium with system A then systems B and C will also be in thermal equilibrium (being in
thermal equilibrium is a transitive relation; moreover, it is an equivalence relation). This is an empirical fact,
based on observation rather than theory. Since A, B, and C are all in thermal equilibrium, it is reasonable to
say each of these systems shares a common value of some property. We call this property temperature.
Generally, it is not convenient to place any two arbitrary systems in thermal contact to see if they are in
thermal equilibrium and thus have the same temperature. Also, it would only provide an ordinal scale.
Therefore, it is useful to establish a temperature scale based on the properties of some reference system.
Then, a measuring device can be calibrated based on the properties of the reference system and used to
measure the temperature of other systems. One such reference system is a fixed quantity of gas. The ideal
gas law indicates that the product of the pressure and volume (P · V) of a gas is directly proportional to the
temperature:
(1)
where 'T is temperature, n is the number of moles of gas and R is the gas constant. Thus, one can define a
scale for temperature based on the corresponding pressure and volume of the gas: the temperature in
kelvins is the pressure in pascals of one mole of gas in a container of one cubic metre, divided by 8.31... In
practice, such a gas thermometer is not very convenient, but other measuring instruments can be
calibrated to this scale.
Equation 1 indicates that for a fixed volume of gas, the pressure increases with increasing temperature.
Pressure is just a measure of the force applied by the gas on the walls of the container and is related to the
energy of the system. Thus, we can see that an increase in temperature corresponds to an increase in the
thermal energy of the system. When two systems of differing temperature are placed in thermal contact, the
temperature of the hotter system decreases, indicating that heat is leaving that system, while the cooler
system is gaining heat and increasing in temperature. Thus heat always moves from a region of high
temperature to a region of lower temperature and it is the temperature difference that drives the heat
transfer between the two systems.
Temperature in gases
As mentioned previously for a monatomic ideal gas the temperature is related to the translational motion or
average speed of the atoms. The kinetic theory of gases uses statistical mechanics to relate this motion to
the average kinetic energy of atoms and molecules in the system. For this case 7736 K = 7463 degrees
Celsius corresponds to an average kinetic energy of one electronvolt; to take room temperature (300 K) as
an example, the average energy of air molecules is 300/7736 eV, or 0.0388 electronvolt. This average
energy is independent of particle mass, which seems counterintuitive to many people. Although the
temperature is related to the average kinetic energy of the particles in a gas, each particle has its own
energy which may or may not correspond to the average. However, after an examination of some basic
physics equations it makes perfect sense. The second law of thermodynamics states that any two given
systems when interacting with each other will later reach the same average energy. Temperature is a
measure of the average kinetic energy of a system. The formula for the kinetic energy of an atom is:
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 85
The Professional Thermographers Association
(Note that a calculation of the kinetic energy of a more complicated object, such as a molecule, is slightly
more involved. Additional degrees of freedom are available, so molecular rotation or vibration must be
included.)
Thus, particles of greater mass (say a neon atom relative to a hydrogen molecule) will move slower than
lighter counterparts, but will have the same average energy. This average energy is independent of the
mass because of the nature of a gas, all particles are in random motion with collisions with other gas
molecules, solid objects that may be in the area and the container itself (if there is one). A visual illustration
of this from Oklahoma State University makes the point more clear. Not all the particles in the container
have different velocities, regardless of whether there are particles of more than one mass in the container,
but the average kinetic energy is the same because of the ideal gas law. In a gas the distribution of energy
(and thus speeds) of the particles corresponds to the Boltzmann distribution.
An electronvolt is a very small unit of energy, approximately 1.602×10-19 joule.
Temperature of the vacuum
When a satellite in empty space is heated by sunshine and cooled by radiating energy away it is not in
thermodynamic equilibrium and has no well-defined temperature.
A system in a vacuum will radiate its thermal energy, i.e. convert heat into electromagnetic waves. If
vacuum is filled with electromagnetic waves (say, radiation from walls of vacuum chamber, or relic
microwave radiation in space) then the system will exchange by energy with these waves and thermally
equilibrates at some finite (non zero) temperature.
Cosmic microwave background radiation being remnant of radiation of hot early universe when radiation
was in thermal equilibrium with matter has Planck spectrum (black body spectrum) with the temperature (at
present) of about 2.7 K.
Second-law definition of temperature
In the previous section temperature was defined in terms of the Zeroth Law of thermodynamics. It is also
possible to define temperature in terms of the second law of thermodynamics, which deals with entropy.
Entropy is a measure of the disorder in a system. The second law states that any process will result in
either no change or a net increase in the entropy of the universe. This can be understood in terms of
probability. Consider a series of coin tosses. A perfectly ordered system would be one in which every coin
toss would come up either heads or tails. For any number of coin tosses, there is only one combination of
outcomes corresponding to this situation. On the other hand, there are multiple combinations that can result
in disordered or mixed systems, where some fraction are heads and the rest tails. As the number of coin
tosses increases, the number of combinations corresponding to imperfectly ordered systems increases. For
a very large number of coin tosses, the number of combinations corresponding to ~50% heads and ~50%
tails dominates and obtaining an outcome significantly different from 50/50 becomes extremely unlikely.
Thus the system naturally progresses to a state of maximum disorder or entropy.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 86
The Professional Thermographers Association
Now, we have stated previously that temperature controls the flow of heat between two systems and we
have just shown that the universe, and we would expect any natural system, tends to progress so as to
maximize entropy. Thus, we would expect there to be some relationship between temperature and entropy.
In order to find this relationship let's first consider the relationship between heat, work and temperature. A
heat engine is a device for converting heat into mechanical work and analysis of the Carnot heat engine
provides the necessary relationships we seek. The work from a heat engine corresponds to the difference
between the heat put into the system at the high temperature, qH and the heat ejected at the low
temperature, qC. The efficiency is the work divided by the heat put into the system or:
(2)
where wcy is the work done per cycle. We see that the efficiency depends only on qC/qH. Because qC and qH
correspond to heat transfer at the temperatures TC and TH, respectively, qC/qH should be some function of
these temperatures:
(3)
Carnot's theorem states that all reversible engines operating between the same heat reservoirs are equally
efficient. Thus, a heat engine operating between T1 and T3 must have the same efficiency as one consisting
of two cycles, one between T1 and T2, and the second between T2 and T3. This can only be the case if:
which implies:
q13 = f(T1,T3) = f(T1,T2)f(T2,T3)
Since the first function is independent of T2, this temperature must cancel on the right side, meaning
f(T1,T3) is of the form g(T1)/g(T3) (i.e. f(T1,T3) = f(T1,T2)f(T2,T3) = g(T1)/g(T2)· g(T2)/g(T3) = g(T1)/g(T3)),
where g is a function of a single temperature. We can now choose a temperature scale with the property
that:
(4)
Substituting Equation 4 back into Equation 2 gives a relationship for the efficiency in terms of temperature:
(5)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 87
The Professional Thermographers Association
Notice that for TC = 0 K the efficiency is 100% and that efficiency becomes greater than 100% below 0 K.
Since an efficiency greater than 100% violates the first law of thermodynamics, this implies that 0 K is the
minimum possible temperature. In fact the lowest temperature ever obtained in a macroscopic system was
20 nK, which was achieved in 1995 at NIST. Subtracting the right hand side of Equation 5 from the middle
portion and rearranging gives:
where the negative sign indicates heat ejected from the system. This relationship suggests the existence of
a state function, S, defined by:
(6)
where the subscript indicates a reversible process. The change of this state function around any cycle is
zero, as is necessary for any state function. This function corresponds to the entropy of the system, which
we described previously. We can rearranging Equation 6 to get a new definition for temperature in terms of
entropy and heat:
(7)
For a system, where entropy S may be a function S(E) of its energy E, the temperature T is given by:
(8)
The reciprocal of the temperature is the rate of increase of entropy with energy.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 88
The Professional Thermographers Association
Temperature conversion formulas
Summary
Comparison of temperature scales
absolute zero water freezes human body temp. water boils
kelvin
0K
273.15 K
310.15 K 373.15 K
Celsius
−273.15 °C
0 °C
37 °C
100 °C
Fahrenheit
−459.67 °F
32 °F
98.6 °F
212 °F
Rankine
0 °Ra 491.67 °Ra
558.27 °Ra 671.67 °Ra
Delisle
559.73 °De
150 °De
94.5 °De
0 °De
Newton
−90.14 °N
0 °N
12.21 °N
33 °N
Réaumur
−218.52 °Ré
0 °Ré
29.6 °Ré
80 °Ré
Rømer
−135.9 °Rø
7.5 °Rø
26.93 °Rø
60 °Rø
Gas Mark 0.00000000142
0.00119
0.00752
0.174
Kelvin
Kelvin
Celsius [°C] = [K] − 273.15
[K] = [°C] + 273.15
Fahrenheit [°F] = [K] · 9/5 − 459.67
[K] = ([°F] + 459.67) · 5/9
Rankine [°Ra] = [K] · 9/5
[K] = [°Ra] · 5/9
Réaumur [°Ré] = ([K] − 273.15) · 4/5
[K] = [°Ré] · 5/4 + 273.15
Newton [°N] = ([K] − 273.15) · 33/100
[K] = [°N] · 100/33 + 273.15
Rømer [°Rø] = ([K] − 273.15) · 21/40 + 7.5 [K] = ([°Rø] − 7.5) · 40/21 + 273.15
Delisle [°De] = (373.15 − [K]) · 3/2
[K] = 373.15 − [°De] · 2/3
Celsius
Degree Celsius
Kelvin [K] = [°C] + 273.15
[°C] = [K] − 273.15
Fahrenheit [°F] = [°C] · 9/5 + 32
[°C] = ([°F] − 32) · 5/9
Rankine [°Ra] = [°C] · 9/5 + 491.67 [°C] = ([°Ra] − 491.67) · 5/9
Réaumur [°Ré] = [°C] · 4/5
[°C] = [°Ré] · 5/4
Newton [°N] = [°C] · 33/100
[°C] = [°N] · 100/33
Rømer [°Rø] = [°C] · 21/40 + 7.5 [°C] = ([°Rø] − 7.5) · 40/21
Delisle [°De] = (100 − [°C]) · 3/2 [°C] = 100 − [°De] · 2/3
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 89
The Professional Thermographers Association
Fahrenheit
Degree Fahrenheit
Kelvin [K] = ([°F] + 459.67) · 5/9
[°F] = [K] · 9/5 − 459.67
Celsius [°C] = ([°F] − 32) · 5/9
[°F] = [°C] · 9/5 + 32
Rankine [°Ra] = [°F] + 459.67
[°F] = [°Ra] − 459.67
Réaumur [°Ré] = ([°F] − 32) · 4/9
[°F] = [°Ré] · 9/4 + 32
Newton [°N] = ([°F] − 32) · 11/60
[°F] = [°N] · 60/11 + 32
Rømer [°Rø] = ([°F] − 32) · 7/24 + 7.5 [°F] = ([°Rø] − 7.5) · 24/7 + 32
Delisle [°De] = (212 − [°F]) · 5/6
[°F] = 212 − [°De] · 6/5
Rankine
Degree Rankine
Kelvin [K] = [°R] · 5/9
[°R] = [K] · 9/5
Celsius [°C] = [°R] · 5/9 − 273.15
[°R] = ([°C] + 273.15) · 9/5
Fahrenheit [°F] = [°R] − 459.67
[°R] = [°F] + 459.67
Réaumur [°Ré] = ([°R] · 5/9 + 273.15) · 4/5 [°R] = ([°Ré] · 5/4 − 273.15) · 9/5
Newton [°N] = ([°R] − 491.67) · 11/60
[°R] = [°N] · 60/11 + 491.67
Rømer [°Rø] = ([°R] − 491.67) · 7/24 + 7.5 [°R] = ([°Rø] − 7.5) · 24/7 + 491.67
Delisle [°De] = (671.67 − [°R]) · 5/6
[°R] = 671.67 − [°De] · 6/5
Delisle
Degree Delisle
Kelvin [K] = 373.15 − [°De] · 2/3 [°De] = (373.15 − [K]) · 3/2
Celsius [°C] = 100 − [°De] · 2/3
[°De] = (100 − [°C]) · 3/2
Fahrenheit [°F] = 212 − [°De] · 6/5
[°De] = (212 − [°F]) · 5/6
Rankine [°Ra] = 671.67 − [°De] · 6/5 [°De] = (671.67 − [°Ra]) · 5/6
Réaumur [°Ré] = 80 − [°De] · 8/15 [°De] = (80 − [°Ré]) · 15/8
Newton [°N] = 33 − [°De] · 11/50 [°De] = (33 − [°N]) · 50/11
Rømer [°Rø] = 60 − [°De] · 7/20 [°De] = (60 − [°Rø]) · 20/7
Newton
Degree Newton
Kelvin [K] = [°N] · 100/33 + 273.15 [°N] = ([K] − 273.15) · 33/100
Celsius [°C] = [°N] · 100/33
[°N] = [°C] · 33/100
Fahrenheit [°F] = [°N] · 60/11 + 32
[°N] = ([°F] − 32) · 11/60
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 90
The Professional Thermographers Association
Rankine
Réaumur
Rømer
Delisle
[°Ra] = [°N] · 60/11 + 491.67 [°N] = ([°Ra] − 491.67) · 11/60
[°Ré] = [°N] · 80/33
[°N] = [°Ré] · 33/80
[°Rø] = [°N] · 35/22 + 7.5
[°N] = ([°Rø] − 7.5) · 22/35
[°De] = (33 − [°N]) · 50/11 [°N] = 33 − [°De] · 11/50
Réaumur
Degree Réaumur
Kelvin [K] = [°Ré] · 5/4 + 273.15 [°Ré] = ([K] − 273.15) · 4/5
Celsius [°C] = [°Ré] · 5/4
[°Ré] = [°C] · 4/5
Fahrenheit [°F] = [°Ré] · 9/4 + 32
[°Ré] = ([°F] − 32) · 4/9
Rankine [°Ra] = [°Ré] · 9/4 + 491.67 [°Ré] = ([°Ra] − 491.67) · 4/9
Newton [°N] = [°Ré] · 33/80
[°Ré] = [°N] · 80/33
Rømer [°Rø] = [°Ré] · 21/32 + 7.5 [°Ré] = ([°Rø] − 7.5) · 32/21
Delisle [°De] = (80 − [°Ré]) · 15/8 [°Ré] = 80 − [°De] · 8/15
Rømer
Degree Rømer
Kelvin [K] = ([°Rø] − 7.5) · 40/21 + 273.15 [°Rø] = ([K] − 273.15) · 21/40 + 7.5
Celsius [°C] = ([°Rø] − 7.5) · 40/21
[°Rø] = [°C] · 21/40 + 7.5
Fahrenheit [°F] = ([°Rø] − 7.5) · 24/7 + 32
[°Rø] = ([°F] − 32) · 7/24 + 7.5
Rankine [°Ra] = ([°Rø] − 7.5) · 24/7 + 491.67 [°Rø] = ([°Ra] − 491.67) · 7/24 + 7.5
Réaumur [°Ré] = ([°Rø] − 7.5) · 32/21
[°Rø] = [°Ré] · 21/32 + 7.5
Newton [°N] = ([°Rø] − 7.5) · 22/35
[°Rø] = [°N] · 35/22 + 7.5
Delisle [°De] = (60 − [°Rø]) · 20/7
[°Rø] = 60 − [°De] · 7/20
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 91
The Professional Thermographers Association
Heat transfer mechanisms
As mentioned previously, heat tends to move from a high temperature region to a low temperature region.
This heat transfer may occur by the mechanisms conduction, and radiation. The term convection is used
to describe the combined effects of conduction and fluid flow. In the past, this has been regarded as a third
mechanism of heat transfer, but, logically, it is not a mechanism of its own.
Thermal Equilibrium
It is an experimental fact that whenever two objects at different temperatures are brought into contact with
one another, the temperature of the warmer object decreases and the temperature of the cooler object
increases. If the two are left in contact for a period of time, they will eventually reach a common equilibrium
temperature somewhere between the two initial temperatures, and there is no further heat flow between
them. When this occurs we say the two objects are in thermal equilibrium.
Steady State vs. Transient Heat Transfer
In an industrial environment, components and processes may or may not reach a thermally equal state with
the environment or the other parts of the structure to which they are attached to. Even though many of
these components will attempt to become thermally equal with their thermal surroundings, in many
situations the objects we deal with are in a constant state of temperature change. It is very important to
know if they are in a steady or transient state of heat transfer.
For example, an operating electric motor will have a particular thermal signature associated with it as a
result of the work it is doing. When the motor has not been working for a long peiored of time (it is off) it is
in thermal equilibrium with its environment. When the motor is turn on it is in a transient state of heat
transfer as it warms up because of the load that is put on it. As it is coming up to a constant operating
temperature above its previous state of being in thermal equilibrium, it is in a state of transient heat
transfer, that is that it is changing (getting hotter then its environment as it is first started). This transient
state only last as long as the motor is either warming up or cooling down compared to its environment.
When the running motor has reached a conststant temperature , operating at a constant load, and the
ambient temperature does not fluctuate, the heat transfer is then steady and so the temperature of the
motor remains the same. This constitutes steady state heat transfer as long as nothing changes.
Therefore, when you have an object that is receiving a constant and steady flow of thermal energy and this
object is, in turn, transferring the received energy to another part of the object or to the environment at a
constant rate, we call this steady state heat transfer.
Conduction
Conduction is the most common means of heat transfer in a solid, liquid or gas. On a microscopic scale,
conduction occurs as hot, rapidly moving or vibrating atoms and molecules interact with neighboring atoms
and molecules, transferring some of their energy (heat) to these neighboring atoms.
In insulators the heat current is carried almost entirely by phonon vibrations.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 92
The Professional Thermographers Association
The "electron fluid" of a conductive metallic solid conducts nearly all of the heat current through the solid.
(Phonon currents are still there, but carry less than 1% of the energy.) Electrons also conduct electric
current through conductive solids, and the thermal and electrical conductivities of most metals have about
the same ratio. A good electrical conductor, such as copper, usually also conducts heat well.
The Peltier-Seebeck effect exhibits the propensity of electrons to conduct heat through an electrically
conductive solid. Thermoelectricity is caused by the relationship between electrons, heat currents and
electrical currents.
Convection
Convection is usually the dominant form of heat transfer in liquids and gases. This is a term used to
characterize the combined effects of conduction and fluid flow. In convection, enthalpy transfer occurs by
the movement of hot or cold portions of the fluid together with heat transfer by conduction. For example,
when water is heated on a stove, hot water from the bottom of the pan rises, heating the water at the top of
the pan. Two types of convection are commonly distinguished, free convection, in which gravity and
buoyancy forces drive the fluid movement, and forced convection, where a fan, stirrer, or other means is
used to move the fluid. Buoyant convection is due to the effects of gravity, and absent in microgravity
environments.
Radiation
Radiation is a means of heat transfer. Radiative heat transfer is the only form of heat transfer that can
occur in the absence of any form of medium and as such is the only means of heat transfer through a
vacuum. Thermal radiation is a direct result of the movements of atoms and molecules in a material. Since
these atoms and molecules are composed of charged particles (protons and electrons), their movements
result in the emission of electromagnetic radiation, which carries energy away from the surface. At the
same time, the surface is constantly bombarded by radiation from the surroundings, resulting in the transfer
of energy to the surface. Since the amount of emitted radiation increases with increasing temperature, a net
transfer of energy from higher temperatures to lower temperatures results.
For room temperature objects (~300 K), the majority of photons emitted (and involved in radiative heat
transfer) are in the infrared spectrum, but this is by no means the only frequency range involved in
radiation. The frequencies emitted are partially related to black-body radiation. Hotter objects—a campfire
is around 700 K, for instance—transfer heat in the visible spectrum or beyond. Whenever EM radiation is
emitted and then absorbed, heat is transferred. This principle is used in microwave ovens, laser cutting,
and RF hair removal.
Heat transfer features
Latent heat: Transfer of heat through a physical change in the medium such as water-to-ice or
water-to-steam involves significant energy and is exploited in many ways: steam engine,
refrigerator etc. (see latent heat of fusion)
Heat pipe: Using latent heat and capilliary action to move heat, it can carry many times as much
heat as a similar sized copper rod and is starting to have applications in laptop personal
computers.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 93
The Professional Thermographers Association
Heat dissipation
In cold climates, houses with their heating systems form dissipative systems. In spite of efforts to insulate
such houses, to reduce heat losses to their exteriors, considerable heat is lost, or dissipated, from them
which would make their interiors uncomfortably cool or cold. The house is an open system inasmuch as it is
incapable of preventing heat from escaping. Furthermore, the interior of the house must be maintained out
of thermal equilibrium with its exterior for the sake of its inhabitants.
In such a house, a thermostat is a device capable of starting the heating system when the house's interior
falls to a set temperature, and of stopping that same system when another set temperature has been
achieved. Thus the thermostat controls the flow of energy into the house, that energy eventually being
dissipated to the exterior.
Preventing heat transfer
It is often desired to prevent heat transfer.
Weatherization slows convective heat flow in buildings. Insulation slows conductive heat flow. Reflective
barriers or radiant barriers slow radiative heat flow, and often couple directly with insulation.
Thermal barrier coatings (TBCs) can also prevent heat transfer from taking place. These ceramic materials,
which are predominant in zirconia, when coated on say, nuclear reactor coolant pipes can cut down the
heat lost from inside the pipe to the atmosphere.
TBCs are generally used on gas turbine blades and shrouds to prevent them from heating up because of
friction and fuel combustion. This protection increases the working life.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 94
The Professional Thermographers Association
Specific heat capacity
The specific heat capacity (symbol c or s, also called specific heat) of a substance is defined as heat
capacity per unit mass. The SI unit for specific heat capacity is the joule per kilogram kelvin, J·kg-1·K-1 or
J/(kg·K), which is the amount of energy required to raise the temperature of one kilogram of the substance
by one kelvin. Heat capacity can be measured by using calorimetry.
The equivalent definition using cgs units is the amount of energy (measured in ergs) required to raise the
temperature of one gram of the substance by one degree Celsius (erg/(g·°C)). Other units of specific heat
capacity include calories per gram degree Celsius (cal/(g·°C) or cal/(g·K)) and Btu per pound degree
Fahrenheit (Btu/(lb·°F))
The symbol cp is often used to denote specific heat capacity at constant pressure.
Substances with low specific heat such as metals require less input energy to increase their temperature.
Substances with high specific heat such as water require much more energy to increase their temperature.
The specific heat can also be interpreted as a measure of how well a substance preserves its temperature,
i.e. "stores" heat, hence the term "heat capacity".
Factors that influence heat capacity measurements
Temperature: Measuring the heat capacity of water produces different results if the starting point
is 20 °C rather than 60 °C. Therefore the temperature the measurement was conducted at must be
specified for the value to be useful.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 95
The Professional Thermographers Association
Table of specific heat capacities
Substance
Air (dry)
Phase
Specific
heat capacity
J/(kg·K)
gas
1005
Air (100% humidity) gas
≈ 1030
Aluminium
solid
900
Brass
solid
377
Copper
solid
385
Diamond
solid
502
Ethanol
liquid
2460
Gold
solid
129
Graphite
solid
720
Helium
gas
5190
Hydrogen
gas
14300
Iron
solid
444
Lithium
solid
3582
Mercury
liquid
139
Nitrogen
gas
1042
Oil
liquid
Oxygen
gas
920
gas
1850
liquid
4186
solid (0 °C)
2060
Water
≈ 2000
Standard ambient temperature and pressure
used unless otherwise noted.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 96
The Professional Thermographers Association
Heat capacity
Heat capacity (abbreviated Cth or just C, also called thermal capacity) is the ability of matter to store heat.
The heat capacity of a certain amount of matter is the quantity of heat (measured in joules) required to
raise its temperature by one kelvin. The SI unit for heat capacity is J/K (joule per kelvin).
Specific Heat Capacity
The heat capacity is the specific heat capacity multiplied by the mass.
C=m·c
where
C is the heat capacity (in SI units, measured in J/K)
m is mass (measured in kilograms)
c is specific heat capacity (measured in J/(kg·K))
Thermal capacitance
Heat capacity is related to thermal capacitance by the formula
Cth = V · ρ · cp
or, more simply,
Cth = m · cp
where
Cth is the thermal capacitance or heat capacity
V is the volume (measured in m3)
ρ is the density (measured in kg/m3)
cp is the specific heat capacity (measured in J/(kg·K) at constant pressure)
m is the mass (measured in kg)
The product ρ · cp is known as volumetric heat capacity, thermal capacitance or (confusingly) thermal
capacity, and has units of J/(m3·K). Dulong and Petit predicted in 1818 that ρcp would be constant for all
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 97
The Professional Thermographers Association
solids (the Dulong-Petit law). In fact, the quantity varies from about 1.2 to 4.5 J/(m3K). For fluids it is in the
range 1.3 to 1.9, and for ideal monatomic gases it is a constant 0.001 J/(m3K).
Gas Phase Heat Capacities
According to the Equipartition Theorem from classical statistical mechanics, any input of energy into a
closed system composed of N molecules is evenly divided among the degrees of freedom available to each
molecule. It can be shown, for each degree of freedom, that
where
E is the input energy (measured in joules)
T is the temperature (measured in kelvins)
R is the ideal gas constant, (8.314570[70] J K-1mol-1)
In the case of a monatomic gas such as helium under constant volume, if it assumed that no electronic or
nuclear quantum excitations occur, each molecule has only 3 degrees of translational freedom. This is
because there is one for each of the vector components of momentum in the x, y, and z directions. This
leads to the equation
where
n is the number of moles of molecules present in the container
The following table shows experimental molar constant volume heat capacity measurements taken for each
noble monatomic gas (at 1 atm and 25 °C):
Monatomic gas Cv,m (J K-1 mol-1), Cv,m/R
He
12.5
1.50
Ne
12.5
1.50
Ar
12.5
1.50
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 98
The Professional Thermographers Association
Kr
12.5
1.50
Xe
12.5
1.50
It is apparent from the table that the experimental heat capacities of the monatomic noble gases agrees
with this simple application of statistical mechanics to a very high degree. In the somewhat more complex
case of an ideal gas of diatomic molecules, the presence of internal degrees of freedom are apparent. In
addition to the three translational degrees of freedom, there are rotational and vibrational degrees of
freedom. In general, there are three degrees of freedom f per atom in the molecule na
Mathematically, there are a total of three rotational degrees of freedom, one corresponding to rotation
about each of the axes of three dimensional space. However, in practice we shall only consider the
existence of two degrees of rotational freedom for linear molecules. This approximation is valid because the
moment of inertia about the internuclear axis is essentially zero. Quantum mechanically, it can be shown
that the interval between successive rotational energy eigenstates is inversely proportional to the moment
of inertia about that axis. Because the moment of inertia about the internuclear axis is vanishingly small
relative to the other two rotational axes, the energy spacing can be considered so high that no excitations
of the rotational state can possibly occur unless the temperature is extremely high. We can easily calculate
the expected number of vibrational degrees of freedom (or vibrational modes). There are three degrees of
translational freedom, and two degrees of rotational freedom, therefore
Each rotational and translational degree of freedom will contribute R/2 in the total molar heat capacity of the
gas. Each vibrational mode will contribute R in the total molar heat capacity, however. This is because for
each vibrational mode, there is a potential and kinetic energy component. Both the potential and kinetic
components will contribute R/2 to the total molar heat capacity of the gas. Therefore, we expect that a
diatomic molecule would have a constant volume heat capacity of
where the terms originate from the translational, rotational, and vibrational degrees of freedom,
respectively. The following is a table of some constant volume heat capacities of various diatomics
Diatomic gas Cv,m (J K-1 mol-1), Cv,m/R
H2
20.18
2.427
CO
20.2
2.43
N2
19.9
2.39
Cl2
24.1
2.90
Br2
32.0
3.84
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 99
The Professional Thermographers Association
From the above table, clearly there is a problem with the above theory. All of the diatomics examined have
heat capacities that are lower than those predicted by the Equipartition theorem, except Br2. However, as
the atoms composing the molecules become heavier, the heat capacities move closer to their expected
values. One of the reasons for this phenomenon is the quantization of vibrational, and to a lesser extent,
rotational states. In fact, if it is assumed that the molecules remain in their lowest energy vibrational state
because the interlevel energy spacings are large, the predicted constant volume heat capacity for a
diatomic molecule becomes
which is a fairly close approximation of the heat capacities of the lighter molecules in the above table. If the
quantum harmonic oscillator approximation is made, it turns out that the quantum vibrational energy level
spacings are actually inversely proportional to the square root of the reduced mass of the atoms composing
the diatomic molecule. Therefore, in the case of the heavier diatomic molecules, the quantum vibrational
energy level spacings become finer, which allows more excitations into higher vibrational levels at a fixed
temperature.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 100
The Professional Thermographers Association
Latent heat
.
Latent heat describes the amount of energy in the form of heat that is required for a material to undergo a
change of phase. Two latent heats are typically described. One is the latent heat of fusion (melting), and
the other is the latent heat of vaporization (evaporation).
They are so named as to describe the direction of heat flow from one phase to the next:
solid → liquid → gas.
The energy change is endothermic when going from solid to liquid to gas, but exothermic when going in the
opposite direction.
For example, in the
atmosphere when a molecule
of water evaporates off the
surface of any body of water,
heat is transported by the
water molecule into a lower
temperature air parcel that
contains more water vapor
than its surroundings. Because
energy is needed to turn water
into water vapor, water vapor
is a way for a body to release
energy. If the water vapor is
returned to a liquid or solid
phase (by condensation or
sublimation), the stored energy
is released as sensible heat
onto the surface where
condensation (or sublimation)
has occurred.
Latent heats and change of phase temps of common fluids and gases
Substance
Latent Heat
Melting
J/g
Melting
Temp
°C
Latent Heat
Boiling
J/g
Alcohol, ethyl
108
-114
855
Ammonia
339
-75
1369
Carbon Dioxide
184
Boiling
Temp
°C
78.3
574
Helium
21
Hydrogen
58
-259
455
-253
Nitrogen
25.7
-210
200
-196
Oxygen
13.9
-219
213
-183
Toluene
351
Turpentine
293
Water
335
0
2272
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
100
Page: 101
The Professional Thermographers Association
Heat of fusion
Heat of fusion is the amount of heat energy which must be absorbed or lost for 1 gram of a substance to
change states from a solid to a liquid or vice versa. It is also called the latent heat of fusion or the enthalpy
of fusion, and the temperature at which it occurs is called the melting point.
When you withdraw thermal energy from a liquid or solid, the temperature falls. When you add heat energy
the temperature rises. However, at the transition point between solid and liquid (the melting point), extra
energy is required (the heat of fusion). To go from liquid to solid, the molecules of a substance must
become more ordered. For them to maintain the order of a solid, extra heat must be withdrawn. In the other
direction, to create the disorder from the solid crystal to liquid, extra heat must be added.
The heat of fusion can be observed if you measure the temperature of water as it freezes. If you plunge a
closed container of room temperature water into a very cold environment (say −20 °C), you will see the
temperature fall steadily until it drops just below the freezing point (0 °C). The temperature then rebounds
and holds steady while the water crystalizes. Once completely frozen, the temperature will fall steadily
again.
The temperature stops falling at (or just below) the freezing point due to the heat of fusion. The energy of
the heat of fusion must be withdrawn (the liquid must turn to solid) before the temperature can continue to
fall.
The units of heat of fusion are usually expressed as
joules per mole (the SI units)
or
calories per gram (old metric units now little used, except for a different, larger calorie used in
nutritional contexts)
or
British thermal units per pound or Btu per pound-mole
Note: These are not the calories found in food. The calories found in food are kilogram calories,
equal to 1000 small calories. 1000 calories = 1 kilocalorie = 1 food calorie. Food calories are
sometimes abbreviated as kcal as if small calories were being used, while calories are abbreviated
as cal. Another distinguishing method, though often confusing, uses capitalization. A Calorie is a
food calorie, or 1000 calories. So 1 Cal = 1000 cal.
Reference Values
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 102
The Professional Thermographers Association
The heat of fusion of water is
79.72 calories per gram
or 334.5 kilojoules per kilogram
The heat of fusion of a few more common substances:
Heat of fusion Heat of fusion
(cal/g)
(kJ/kg)
methane:
13.96
58.41
ethane:
22.73
95.10
propane:
19.11
79.96
methanol: 23.70
99.16
ethanol:
26.05
108.99
glycerol:
47.95
200.62
formic acid: 66.05
276.35
acetic acid: 45.91
192.09
acetone:
23.42
97.99
benzene:
30.45
127.40
myristic acid: 47.49
198.70
palmitic acid: 39.18
163.93
stearic acid: 47.54
198.91
Substance
These values are from the CRC Handbook of Chemistry and Physics, 62nd .ion.
Application
To heat one kilogram (about 1 liter) of cool water 20 °C from 10 °C to 30 °C requires 20 kcal.
However, to melt ice and raise the resulting water temperature 20 °C requires extra energy. To heat ice
from 0 °C to water at 20 °C requires:
(1) 80 cal/g (heat of fusion of ice) = 80 kcal for 1 kg
PLUS
(2) 1 cal/(g·°C) = 20 kcal for 1 kg to go up 20 °C
= 100 kcal
Heat of vaporization
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 103
The Professional Thermographers Association
The heat of vaporization is a physical property of substances. It is defined as the heat required to
vaporize one mole of a substance at its boiling point under standard pressure (101.325 kPa). The heat of
vaporization is expressed in kJ/mol. The use of kJ/kg is also possible, but less customary. Other units still
in use include Btu/lb.
Because vaporization is the opposite process of condensation, the term heat of condensation is also
used. The latter is defined as the heat released when one mole of the substance condenses at its boiling
point under standard pressure.
The heat of vaporization of water is about 2260 kJ/kg which is equal to 40.8 kJ/mol. This is quite a lot: it is
five times the energy needed for heating the water from 0 to 100 degrees Celsius.
Heats of vaporization of the elements
Heat of
vaporization
(kJ/mol)
Element
Actinium
n/a
Heat of
vaporization
(kJ/mol)
Element
Heat of
vaporization
(kJ/mol)
Element
Hydrogen
0.44936
Radon
16.4
Aluminium 293.4
Indium
231.5
Rhenium
715
Antimony
77.14
Iodine
20.752
Rhodium
493
Argon
6.447
Iridium
604
Rubidium
72.216
Arsenic
34.76
Iron
349.6
Ruthenium 595
Astatine
114
Krypton
9.029
Scandium
314.2
Barium
142
Lanthanum 414
Selenium
26.3
Beryllium
292.40
Lead
177.7
Silicon
384.22
Bismuth
104.8
Lithium
145.92
Silver
250.58
Boron
489.7
Magnesium 127.4
Sodium
96.96
Bromine
15.438
Manganese 226
Strontium
144
Cadmium
100
Mercury
Sulfur
1.7175
Caesium
67.74
Molybdenum 598
Tantalum
743
Calcium
153.6
Neon
1.7326
Technetium 660
Carbon
355.8
Neptunium
n/a
Tellurium
52.55
Cerium
414
Nickel
370.4
Thallium
164.1
Chlorine
10.2
Niobium
696.6
Thorium
514.4
Chromium 344.3
Nitrogen
2.7928
Tin
295.8
Cobalt
376.5
Osmium
627.6
Titanium
421
Copper
300.3
Oxygen
3.4099
Tungsten
824
Fluorine
3.2698
Palladium
357
Vanadium 452
Gallium
258.7
Phosphorus 12.129
59.229
Xenon
12.636
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 104
The Professional Thermographers Association
Germanium 330.9
Platinum
510
Yttrium
363
Gold
334.4
Polonium
60.1
Zinc
115.3
Hafnium
575
Potassium
79.87
Zirconium
58.2
Helium
0.0845
Radium
37
Methanol
37.4
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 105
The Professional Thermographers Association
Heat conduction
.
Heat flow along perfectly insulated wire
Heat conduction is the transmission of heat
across matter.
Heat transfer is always directed from a
higher to a lower temperature. Denser
substances are usually better conductors;
metals are excellent conductors.
The law of heat conduction also know as
Fourier's law states that the time rate of
heat flow Q through a slab (or a portion of a
perfectly insulated wire, as shown in the
figure) is proportional to the gradient of
temperature difference:
A is the transversal surface area, Δ x is the thickness of the body of matter through which the heat is
passing, K is a conductivity constant dependent on the nature of the material and its temperature, and ΔT is
the temperature difference through which the heat is being transferred. This law forms the basis for the
derivation of the heat equation.
Thermal
Conductivity (k)
Material
Metals
Aluminum
Brass
w/m2 K
205
109
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 106
The Professional Thermographers Association
Copper
Lead
Silver - 99.9% pure
Steel - carbon .5%
Building Material
Brick, Red
Concrete
Common brick
Glass - window
Insulating brick
Plywood
Plaster, Gypsum
Insulation Materials
Fibreglass/ Rockwool
Corkboard
Felt
Down
Styrofoam
Refractory Brick
Other
Rubber- soft
Ice
Water
Animal fat
PVC -sheet
Air
Vacuum
380
35
407
54
.6
.8
.65
.78
.6
.12
.48
.038
.043
.052
.02
.01
1.05
0.13
1.88
.56
.17
.175
.024
0
Conductance
Writing
Fourier's law can also be stated as:
where U is the conductance. The reciprocal of conductance is resistance, equal to '
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 107
The Professional Thermographers Association
and it is resistance which is additive when several conducting layers lie between the hot and cool regions,
because A and Q are the same for all layers. In a multilayer partition, the total conductance is related to the
conductance of its layers by:
So, when dealing with a multilayer partition, the following formula is usually used:
When heat is being conducted from one fluid to another through a barrier, it is sometimes important to
consider the conductance of the thin film of fluid which remains stationary next to the barrier. This thin film
of fluid is difficult to quantify, its characteristics depending upon complex conditions of turbulence and
viscosity, but when dealing with thin high-conductance barriers it can sometimes be quite significant.
Newton's law of cooling
Newton's law of cooling, states that the rate of heat loss of a body is proportional to the difference in
temperatures between the body and its surroundings. This form of heat loss principle, however, is not
very precise; a more accurate formulation requires an analysis of heat flow based on the heat equation in
an inhomogeneous medium. The general applicability of this simplification is characterized by the Biot
number.
Nevertheless, it is easy to derive from this principle the exponential decay of temperature of a body. If T is
the temperature of the body, then
where r is some positive constant. From which, it follows that
For example, simplified climate models may use Newtonian cooling instead of a full (and computationally
expensive) radiation code to maintain atmospheric temperatures.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 108
The Professional Thermographers Association
Thermal conductivity
.
In physics, thermal conductivity, λ, is the intensive property of a material which relates its ability to
conduct heat.
Thermal conductivity is the quantity of heat, Q, transmitted through a thickness L, in a direction normal to
a surface of area A, due to a temperature gradient ΔT, under steady state conditions and when the heat
transfer is dependent only on the temperature gradient.
thermal conductivity = heat flow rate × distance / (area × temperature gradient)
λ = Q × L / (A × ΔT)
Examples
In general thermal conductivity tracks electrical conductivity; metals being good thermal conductors. There
are exceptions: the most outstanding is that of diamond which has a high thermal conductivity, between
1000 and 2600 W·m-1·K-1, while its electrical conductivity is low.
The following table was compiled from data available in the CRC Handbook of Chemistry and Physics. All
values are for materials at room temperature (298K), except where indicated.
Thermal conductivity of other common materials:
Thermal conductivity (298K)
W·m-1·K-1
Diamond
895-2300
Silver
429
Copper
401
Gold
317
Aluminium
237
Brass
120
Platinum
71.6
Iron
80.2
Lead
35.3
Quartz (273K) 6.8-12
Glass
1.35
Wood
0.04 (balsa) - 0.35 (fir)
Styrofoam
0.033
Wool
0.04
Silica aerogel 0.017
Air (100 kPa) 0.0262
Material
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 109
The Professional Thermographers Association
Water
Ice (273K)
Mercury
0.6062
2.2
8.514
Thermal conductivity changes with temperature. For most materials it decreases slightly as the temperature
rises.
Since diamond has such a high thermal conductivity, natural blue diamond much higher still, one may test
gems to determine if they are genuine diamonds using a thermal conductance tester, one of the
instruments of gemology. Diamonds of any size are notably cool to the touch because of their high thermal
conductivity, perhaps the origin of the term "ice."
Related terms
The reciprocal of thermal conductivity is thermal resistivity, measured in kelvin-metres per watt (K·m·W-1).
When dealing with a known amount of material, its thermal conductance and the reciprocal property,
thermal resistance, can be described. Unfortunately there are differing definitions for these terms.
First definition (general)
For general scientific use, thermal conductance is the quantity of heat that passes in unit time through a
plate of particular area and thickness when its opposite faces differ in temperature by one degree. For a
plate of thermal conductivity λ, area A and thickness L this is λA/L, measured in W·K-1. This matches the
relationship between electrical conductivity (A·m-1·V-1) and electrical conductance (A·V-1).
There is also a measure known as heat transfer coefficient: the quantity of heat that passes in unit time
through unit area of a plate of particular thickness when its opposite faces differ in temperature by one
degree. The reciprocal is thermal insulance. In summary:
thermal conductance = λA/L, measured in W·K-1
o thermal resistance = L/λA, measured in K·W-1
heat transfer coefficient = λ/L, measured in W·K-1·m-2
o thermal insulance = L/λ, measured in K·m2·W-1.
The heat transfer coefficient is also known as thermal admittance. But thermal admittance may mean other
things.
Second definition (buildings)
When dealing with buildings, thermal resistance or R-value means what is described above as thermal
insulance, and thermal conductance means the reciprocal. For materials in series, these thermal
resistances (unlike conductances) can simply be added to give a thermal resistance for the whole.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 110
The Professional Thermographers Association
A third term, thermal transmittance, incoporates the thermal conductance of a structure along with heat
transfer due to convection and radiation. It is measured in the same units as thermal conductance and is
sometimes known as the composite thermal conductance. The term U value is another synonym.
The term K value is a synonym for thermal conductivity.
In summary, for a plate of thermal conductivity λ, area A and thickness L:
thermal conductance = λ/L, measured in W·K-1·m-2
thermal resistance (R value, thermal resistivity in scientific terms) = L/λ, measured in K·m2·W-1.
thermal transmittance (U value)= 1/(Σ(L/λ)) + convection + radiation, measured in W·K-1·m-2
Molecular origins
The thermal conductivity of a system is determined by how molecules comprising the system interact.
There are no simple but correct expressions for the thermal conductivity. The simplest exact expression
employs one of the Green-Kubo relations. Although this expression is exact, in order to calculate the
thermal conductivity of a dense fluid or solid using this relation requires the use of molecular dynamics
computer simulation.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 111
The Professional Thermographers Association
Convection
Convection is the transfer of heat by the motion of or within a fluid. It may arise from temperature
differences either within the fluid or between the fluid and its boundary, other sources of density variations
(such as variable salinity), or from the application of an external motive force. It is one of the three primary
mechanisms of heat transfer, the others being
conduction and radiation. Convection occurs in
atmospheres, oceans, and planetary mantles.
Free and forced convection
In heat transfer, a distinction is made between
free and forced convection.
Free convection is convection in which motion
of the fluid arises solely due to the temperature
differences existing within the fluid. Example:
hot air rising off the surface of a radiator.
The basic premise behind free convection is
that heated matter becomes more buoyant and
"rises"; while cooler material "sinks". Free
convection occurs in any liquid or gas which
expands or contracts in response to changing
temperatures when it is exposed to multiple
temperatures in an acceleration field such as
gravity or a centrifuge. The local changes in
density results in buoyancy forces that cause
currents in the fluid.
Forced convection happens when motion of
the fluid is imposed externally (such as by a
pump, fan or even the wind). Example: a fanpowered heater, where a fan blows cool air
past a heating element, heating the air. When a
person blows on their food to cool it, he/she is using forced convection.
Convection at a surface
In both of the previous examples, an engineer would often be interested in the rate of heat transfer from the
hot 'source' surface to the fluid medium.
The local convective heat flux of a fluid passing over a surface is expressed as
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 112
The Professional Thermographers Association
q' = h (Ts - T∞);
q' local heat flux (dq/dA)
h local convection coefficient
Ts surface temperature
T∞ quiescent or ambient temperature
The total heat transfer over a surface is then calculated as the integral of q",
q = ∫Asq' dAs
As area of the surface
q total heat transfer rate (units of energy/time)
This then leads to a definition of average convection coefficient, h-bar, defined from
q = h-bar As (Ts - T∞)
Studies of forced convection lead to a close inspection of the flow in the boundary layer of the fluid.
Atmospheric convection
In the case of Earth's atmosphere, solar radiation heats the Earth's surface, and this heat is then
transferred to the air by convection. When a layer of air receives enough heat from the Earth's surface, it
expands, becomes less dense and is pushed upward by buoyancy. Colder, heavier air sinks under it and is
then warmed, expands, and rises. The warm rising air cools as it reaches the higher, cooler regions of the
atmosphere and becomes more dense. Since it cannot sink through the rising air beneath it, it moves
laterally and then begins to sink. When it reaches the surface again it is heated, and is drawn back into the
original rising column. These convection currents cause local breezes, winds, thermals, cyclones and
thunderstorms, and at a larger scale, produce the global atmospheric circulation features.
A single region of air with a rising and falling current is called a convection cell.
Heat is lost from the rising air through radiation into space.
Oceanic convection
Solar radiation also affects the oceans. Warm water from the Equator tends to circulate toward the poles,
while cold polar water heads towards the Equator. Oceanic convection is also frequently driven by density
differences due to varying salinity, known as thermohaline convection, and is of crucial importance in the
global thermohaline circulation. In this case it is quite possible for relatively warm, saline water to sink, and
colder, fresher water to rise, reversing the normal transport of heat.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 113
The Professional Thermographers Association
COMPENSATING FOR WIND EFFECTS – Electrical Systems
During outdoor measurements the cooling effect of wind must be taken into account. The table below is a
hypothetical correction table that has been developed, but it is by no means the rule! Please note that it is
being presented in as a discussion topic only. Where it makes the suggestion that at a wind speed of
greater then 17.9 MPH, measurements are not recommended, would mean that in places like Hawaii,
USA that you could not do IR, because the trade winds typically blow at from 20 to 25 MPG all of the
time. When in point in case, it is very easy to perform, and identify problems and make
measurements, but it should be noted that the wind will have an effect on the cooling of the
problems and that they will be hotter when the wind stops blowing!
An anomaly measured at a wind velocity of 5 m/sec. (10 knots) will be approximately twice as high at 1
m/sec. An excess temperature measured at 8 m/sec. will be 2.5 times as high at 1 m/sec. Normally, it is
not recommended to conduct thermographic inspections at more than 8 m/sec. Of course,
emergency measurements are excluded from this general rule. The table below shows the correction
factor for wind speeds from 1 to 8 m/s.
WIND SPEED
m/s
CORRECTION FACTOR
mph
<1
<2.24
1.00
2
4.48
1.36
3
6.72
1.64
4
8.96
1.86
5
11.2
2.06
6
13.4
2.23
7
15.7
2.40
8
17.9
2.50
9 (OR MORE)
MEASUREMENT NOT RECOMMENDED
_________________________________________________________________
(1 METRE/SECOND = 2 KNOTS = 2.2369 MILES PER HOUR)
(measured overheating) x (correction factor) = (excess temperature at 1 m/s)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 114
The Professional Thermographers Association
Thermal radiation
Thermal radiation, or radiant
heat, is electromagnetic
radiation from an object that is
simply caused by its
temperature. It rapidly
increases in power, and also
increases in frequency, with
increasing temperature.
A black body is an object that
absorbs all electromagnetic
radiation that falls onto it. For
this case there are formulas
for the power as a function of
temperature, etc.
A device specially made for
thermal radiation may have
different purposes:
the radiation itself may be the objective, as in a light bulb
the objective may be cooling
For example, spacecraft may have thermal radiators, also called heat radiators to lose excess heat.
They tend to be reflective to avoid absorption of solar radiation energy. E.g. the Space Shuttle has heat
radiators mounted on the inner surfaces of the payload bay doors, and so these are kept open while the
Shuttle is in orbit to allow the radiators to function.
Radiant energy
Radiant energy is the energy transported by electromagnetic waves. The term is most commonly used in
the fields of solar energy, heating and lighting. The quantity of radiant energy may be calculated by
integrating radiant power with respect to time and, like all forms of energy, its SI unit is the joule.
Analysis
Because electromagnetic (EM) radiation can be considered as consisting of photons, radiant energy can be
viewed as the energy carried by these photons. The radiation emitted by radiating bodies is concentrated in
certain portions of the electromagnetic spectrum. Such radiation bands may be sharply defined, as is seen
in star spectra, or may be more diffuse. EM radiation has three principal components: wavelength and
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 115
The Professional Thermographers Association
frequency, which are strictly inversely proportional, and intensity, which determines the amount of energy
carried by the radiation. Radiant energy may originate in a body or be reflected from another body (in which
case it may be refracted or otherwise modified).
Open systems
Radiant energy is one of the energy sources that can be used to power an open system. Such an open
system can be man-made (such as a solar energy collector), or natural, such as the Earth's atmosphere. In
geophysics, transparent greenhouse gases trap the sun's radiant energy (at certain wavelengths), allowing
it to penetrate deep into the atmosphere or all the way to the Earth's surface, where they are re-emitted as
longer wavelength radiation (chiefly infrared radiation). Radiant energy produced in the sun is a result of
nuclear fusion.
SI radiometry units
SI radiometry units
.
Quantity
SI unit
Symbol
Notes
radiant
energy
Joule
J
energy
radiant flux
Watt
W
radiant energy per unit time, also called
Radiant power
radiant
intensity
Watts per
steradian
W·sr-1
power per unit solid angle
radiance
Watts per
steradian per
square metre
W·sr1·m-2
power per unit solid angle per unit
projected source area
energy flux
density
Watts per square
W·m-2
metre
power per unit area of the receiver
(irradiance) or the emitter (radiant
emittance or radiant exitance)
spectral
radiance
watt per steradian W·srper cubic metre 1·m-3
commonly measured in W·sr-1·m-2·nm-1
spectral
irradiance
Watt per cubic
metre
commonly measured in W·m-2·nm-1
W·m-3
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 116
The Professional Thermographers Association
Electromagnetic radiation
Electromagnetic radiation is a propagating wave in space with electric and magnetic components. These
components oscillate at right angles to each other and to the direction of propagation.
The term electromagnetic radiation is also used as a synonym for electromagnetic waves in general, even if
they are not radiating or traveling in free space. This sense includes, for example, light traveling through an
optical fiber, or electrical energy traveling within a coaxial cable.
Electromagnetic (EM) radiation carries energy and momentum which may be imparted when it interacts
with matter.
Physics
o
Theory
Electromagnetic waves were predicted by Maxwell's equations and subsequently discovered by Heinrich
Hertz. Maxwell derived a wave form of the electric and magnetic equations which made explicit the wave
nature of the electric and magnetic fields. These equations displayed the symmetry of the fields.
According to the theory, a time-varying electric field generates a magnetic field and vice versa. Thus, an
oscillating electric field creates an oscillating magnetic field, which in turn creates an oscillating electric
field, and so on. By this means an EM wave is produced which propagates through space.
Properties
Electric and magnetic fields exhibit the property of superposition. This means that the field due to a
particular particle or time-varying electric or magnetic field adds to the fields due to other causes. (As
magnetic and electric fields are vector fields, this is the vector addition of all the individual electric and
magnetic field vectors.) As a result, EM radiation is influenced by various phenomena such as refraction
and diffraction. For example, a traveling EM wave incident on a particular arrangement of atoms induces
oscillation in the atoms and thus causes them to emit their own EM waves (called wavelets). These
emissions interfere with the impinging wave and alter its form.
In refraction, a wave moving from one medium to another of a different density changes its speed and
direction when it enters the new medium. The ratio of the refractive indices of the media determines the
extent of refraction. Refraction is the mechanism by which light disperses into a spectrum when it is shone
through a prism.
The physics of electromagnetic radiation is electrodynamics, a subfield of electromagnetism.
EM radiation exhibits both wave properties and particle properties (see wave-particle duality). These
characteristics are mutually exclusive and appear separately in different circumstances: the wave
characteristics appear when EM radiation is measured over relatively larger timescales and over larger
distances, and the particle characteristics are evident when measuring smaller distances and timescales.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 117
The Professional Thermographers Association
EM radiation's behaviors as a wave and as a stream of particles have been confirmed by a large number of
experiments.
Wave model
An important aspect of the wave nature of light is frequency. The frequency of a wave is its rate of
oscillation and is measured in hertz, the SI unit of frequency, equal to one oscillation per second. Light
usually comprises a spectrum of frequencies which sum to form the resultant wave. In addition, frequency
affects properties like refraction, in which different frequencies undergo a different level of refraction.
Particle model
In the particle model of EM radiation, EM radiation is quantized as particles called photons. Quantisation of
light represents the discrete packets of energy which constitute the radiation. The frequency of the radiation
determines the magnitude of the energy of the particles. Moreover, these particles are emitted and
absorbed by charged particles, so photons act as transporters of energy.
A photon absorbed by an atom excites an electron and elevates it to a higher energy level. If the energy is
great enough, the electron is liberated from the atom in a process called ionisation. Conversely, an electron
which descends to a lower energy level in an atom emits a photon of light equal to the energy difference.
The energy levels of electrons in atoms are discrete. Therefore, each element has its own characteristic
frequencies.
Together these effects explain the absorption spectra of light. The dark bands in the spectrum are due to
the atoms in the intervening medium which absorb different frequencies of the light. The composition of the
medium through which the light travels determines the nature of the absorption spectrum. For instance, in a
distant star, dark bands in the light it emits are due to the atoms in the atmosphere of the star. These bands
correspond to the allowed energy levels in the atoms. A similar phenomenon occurs for emission. As the
electrons descend to lower energy levels, a spectrum which represents the jumps between the energy
levels of the electrons is exhibited.
Speed of propagation
Any electric charge which accelerates, or any changing magnetic field, produces electromagnetic radiation.
Electromagnetic information about the charge travels at the speed of light. Accurate treatment thus
incorporates a concept known as retarded time (as opposed to advanced time, which is unphysical in light
of causality), which adds to the expressions for the electrodynamic electric field and magnetic field. These
extra terms are responsible for electromagnetic radiation. When any wire (or other conducting object such
as an antenna) conducts alternating current, electromagnetic radiation is propagated at the same frequency
as the electric current. Depending on the circumstances, it may behave as a wave or as particles. As a
wave, it is characterized by a velocity (the speed of light), wavelength, and frequency. When considered as
particles, they are known as photons, and each has an energy related to the frequency of the wave given
by Planck's relation E = hν, where E is the energy of the photon, h = 6.626 × 10-34 J·s is Planck's constant,
and ν is the frequency of the wave.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 118
The Professional Thermographers Association
One rule is always obeyed regardless of the circumstances. EM radiation in a vacuum always travels at the
speed of light, relative to the observer, regardless of the observer's velocity. (This observation led to Albert
Einstein's development of the theory of special relativity.)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 119
The Professional Thermographers Association
Electromagnetic spectrum
Generally, EM radiation is classified by wavelength into gamma rays, X-rays, ultraviolet, the visible region
(we perceive as light), infrared, , microwave , radio and electrical energy.
Electromagnetic Spectrum
Gamma ray | X-ray | Ultraviolet | | Optical spectrum | Infrared | Terahertz radiation| Microwave | Radio waves
Optical (visible) spectrum Violet | Indigo | Blue | Green | Yellow | Orange| Red
The behavior of EM radiation depends on its wavelength. Higher frequencies have shorter wavelengths,
and lower frequencies have longer wavelengths. When EM radiation interacts with single atoms and
molecules, its behavior depends on the amount of energy per quantum it carries.
The electromagnetic
spectrum is the range of all
possible electromagnetic
radiation. Also, the
"electromagnetic spectrum"
(usually just spectrum) of an
object is the range of
electromagnetic radiation that
it emits, reflects, or transmits.
The electromagnetic
spectrum, shown in the table,
extends from frequencies
used in the electric power grid
(at the long-wavelength end)
to gamma radiation (at the
short-wavelength end),
covering wavelengths from
thousands of kilometres down
to fractions of the size of an
atom.
Legend:
γ = Gamma rays
HX = Hard X-rays
SX = Soft X-Rays
EUV = Extreme
ultraviolet
NUV = Near ultraviolet
Visible light
NIR = Near infrared
MIR = Moderate
infrared
FIR = Far infrared
Radio waves:
EHF = Extremely high
frequency
(Microwaves)
Wavelength
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 120
The Professional Thermographers Association
The wavelength is the distance between repeating units of a wave pattern. It is commonly designated by
the Greek letter lambda (λ).
In a sine wave, the wavelength is the
distance between peaks:
The x axis represents distance, and I
would be some varying quantity (for
instance air pressure for a sound wave or
strength of the electric or magnetic field for
light), at a given point in time as a function
of x.
Wavelength λ has an inverse relationship
to frequency f, the number of peaks to
pass a point in a given time. The
wavelength is equal to the speed of the
wave type divided by the frequency of the
wave. When dealing with electromagnetic
radiation in a vacuum, this speed is the speed of light c.
Frequency Wavelength Conversion Table
http://www.unitconversion.org/unit_converter/frequency-wavelength-ex.html
Frequency to Wavelength Conversion Chart and Calculator
http://www.ee.byu.edu/photonics/fwnomograph.phtml
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 121
The Professional Thermographers Association
How big is a Micron?
When dealing with infrared cameras, you will be typically using a micron (µ) at the unit of measurement of
wavelength for the infrared portion of the electromagnetic spectrum that the cameras operate in. It is
important to become familiar with this term.
A metric unit of distance equal to one millionth of a meter. "Micron" is simply a shorter name for the
micrometer. In 1968 the CGPM decided to drop the micron as an approved unit and recommend that
micrometers be used instead. Microns, however, are still in common use.
There are 25,400 microns in one inch.
This dot ( . ) is approximately 1/64 of an inch wide and equals 615 microns.
1 micrometer (micron) is equal to:
Metric
kilometer
1e-09
meter
1e-06
decimeter
1e-05
centimeter
0,0001
millimeter
0,001
nanometer
1000
angstrom
10000
Common Items and their respective particle sizes
Postage Stamp, 1 inch high
25,400 microns
Eye of a Needle
1,230 microns
Human Hair
40 to 300 microns
Oil Smoke
0.03 to 1 micron
Tobacco Smoke
0.01 to 1 microns
Beach Sand
100 to 2000 microns
Pollens
10 to 1000 microns
Typical Atmospheric Dust
0.001 to 30 microns
Mold Spores
10 to 30 microns
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 122
The Professional Thermographers Association
Spectra of objects
Spectroscopy can detect a much wider region of the EM spectrum than the visible range of .4 µm to .7 µm.
A common laboratory spectroscope can detect wavelengths from 2 nm to 2500 nm. More in-depth
information about the physical properties of objects, gases, or even stars can be obtained from this type of
device. It is widely used in astrophysics. For example, many hydrogen atoms emit radio waves which have
a wavelength of 21.12 cm.
Nearly all objects in the universe emit, reflect and/or transmit some light. (In the broadest of terms the word
light is used for Ultraviolet, Visual, and Infrared rather commonly in physics, so don’t think of it as only the
visual spectrum.) The distribution of this light along the electromagnetic spectrum (called the spectrum of
the object) is determined by the object's composition. Several types of spectra can be distinguished
depending upon the nature of the radiation coming from an object:
If the spectrum is composed primarily of thermal radiation emitted by the object itself, an emission
spectrum occurs.
o Some bodies emit light more or less according to the blackbody spectrum.
If the spectrum is composed of background light, parts of which the object transmits and parts of
which it absorbs, an absorption spectrum occurs.
Electromagnetic spectroscopy is the branch of physics that deals with the characterization of matter by its
spectra.
Classification systems
While the classification scheme is generally accurate, in reality there is often some overlap between
neighboring types of electromagnetic energy. For example, SLF radio waves at 60 Hz may be received and
studied by astronomers, or may be ducted along wires as electric power. Also, some low-energy gamma
rays actually have a longer wavelength than some high-energy X-rays. This is possible because "gamma
ray" is the name given to the photons generated from nuclear decay or other nuclear and sub-nuclear
processes, whereas X-rays on the other hand are generated by electronic transitions involving highly
energetic inner electrons. Therefore the distinction between gamma ray and X-ray is related to the radiation
source rather than the radiation wavelength. Generally, nuclear transitions are much more energetic than
electronic transitions, so usually, gamma-rays are more energetic than X-rays. However, there are a few
low-energy nuclear transitions (e.g. the 14.4 keV nuclear transition of Fe-57) that produce gamma rays that
are less energetic than some of the higher energy X-rays.
Use of the radio frequency spectrum is regulated by governments. This is called frequency allocation.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 123
The Professional Thermographers Association
Gamma rays
After hard X-rays come gamma rays. These are the most energetic photons, having no lower limit to their
wavelength. They are useful to astronomers in the study of high-energy objects or regions and find a use
with physicists thanks to their penetrative ability and their production from radioisotopes. The wavelength of
gamma rays can be measured with high accuracy by means of Compton scattering.
Note that there are no defined boundaries between the types of electromagnetic radiation. Some
wavelengths have a mixture of the properties of two regions of the spectrum. For example, red light
resembles infra-red radiation in that it can resonate some chemical bonds.
X-rays
After UV come X-rays. Hard X-rays are of shorter wavelengths than soft X-rays. X-rays are used for seeing
through some things and not others, as well as for high-energy physics and astronomy. Neutron stars and
accretion disks around black holes emit X-rays, which enable us to study them.
Ultraviolet light
Next comes ultraviolet. This is radiation whose wavelength is shorter than the violet end of the visible
spectrum.
Being very energetic, UV can break chemical bonds, make molecules unusually reactive or ionize them, in
general changing their mutual behavior. Sunburn, for example, is caused by the disruptive effects of UV
radiation on skin cells, which can even cause skin cancer, if the radiation damages the complex DNA
molecules in the cells (UV radiation is a proven mutagen). The Sun emits a large amount of UV radiation,
which could quickly turn Earth into a barren desert, but most of it is absorbed by the atmosphere's ozone
layer before reaching the surface.
Visible radiation Light
EM radiation with a wavelength between.4 µm to .7 µm. is detected by the human eye and perceived as
visible light.
If radiation having a frequency in the visible region of the EM spectrum shines on an object, say a bowl of
fruit, this results in our visual perception identifying information from the scene. Our brain's visual system
processes the multitude of reflected frequencies into different shades and hues, and through this notentirely-explained "psychophysical phenomenon," most humans perceive a bowl of fruit.
In the vast majority of cases, however, the information carried by light is not directly apprehensible by
human senses. Natural sources produce EM radiations across the spectrum; so, too, can human
technology manipulate a broad range of wavelengths. Optical fiber transmits light which, although not
suitable for direct viewing, can carry data. Those data can be translated into sound or even into an image.
The coded form of such data is similar to that used with radio waves.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 124
The Professional Thermographers Association
Color
Wavelength interval
Frequency interval
red
~ 625 to 740 nm
~ 480 to 405 THz
orange
~ 590 to 625 nm
~ 510 to 480 THz
yellow
~ 565 to 590 nm
~ 530 to 510 THz
green
~ 520 to 565 nm
~ 580 to 530 THz
cyan
~ 500 to 520 nm
~ 600 to 580 THz
blue
~ 430 to 500 nm
~ 700 to 600 THz
violet
~ 380 to 430 nm
~ 790 to 700 THz
Continuous spectrum The spectrum of visible light Designed for monitors with gamma 1.5.
After infrared comes visible light. This is the range in which the sun and stars similar to it emit most of their
radiation. It is probably not a coincidence that the human eye is sensitive to the wavelengths that the sun
emits most strongly. Visible light (and near-infrared light) is typically absorbed and emitted by electrons in
molecules and atoms that move from one energy level to another. The light we see with our eyes is really a
very small portion of the electromagnetic spectrum. A rainbow shows the optical (visible) part of the
electromagnetic spectrum; infrared (if you could see it) would be located just beyond the red side of the
rainbow with ultraviolet appearing just beyond the violet end.
Infrared radiation
The infrared part of the electromagnetic spectrum covers the range from roughly .75 to 1,000 microns or (
400 THz, 750 nm to 300 GHz, 1 mm) It can be divided into three parts:
Near-infrared, from (2,500 to 750 nm). 120 to 400 THz. Physical processes that are relevant for
this range are similar to those for visible light.
Mid-infrared, from (10 to 2.5 μm) 30 to 120 THz. Hot objects (black-body radiators) can radiate
strongly in this range. It is absorbed by molecular vibrations, that is, when the different atoms in a
molecule vibrate around their equilibrium positions. This range is sometimes called the fingerprint
region since the mid-infrared absorption spectrum of a compound is very specific for that
compound.
Far-infrared, from (1 mm) 300 GHz to (10 μm). 30 THz The lower part of this range may also be
called microwaves. This radiation is typically absorbed by so-called rotational modes in gas-phase
molecules, by molecular motions in liquids, and by phonons in solids. The water in the Earth's
atmosphere absorbs so strongly in this range that it renders the atmosphere effectively opaque.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 125
The Professional Thermographers Association
However, there are certain wavelength ranges ("windows") within the opaque range which allow
partial transmission, and can be used for astronomy. The wavelength range from approximately
200 μm up to a few mm is often referred to as "sub-millimeter" in astronomy, reserving far infrared
for wavelengths below 200 μm.
Terahertz radiation
This is a region of the light spectrum between far infrared and microwaves. Until recently, the range was
rarely studied and few sources existed for microwave energy at the high end of the band (sub-millimeter
waves or so-called terahertz waves), but applications are now appearing. The proposed WiMAX standard
for wireless networking, a long-range enhancement of Wi-Fi, lies within this region.
Microwaves
The super high frequency (SHF) and extremely high frequency (EHF) of Microwaves come next.
Microwaves are waves which are typically short enough to employ tubular metal waveguides of reasonable
diameter. Microwave energy is produced with klystron and magnetron tubes, and with solid state diodes
such as Gunn and IMPATT devices. Microwaves are absorbed by molecules that have a dipole moment in
liquids. In a microwave oven, this effect is used to heat food. Low-intensity microwave radiation is used in
Wi-Fi.
It should be noted that an average microwave oven in active condition is, in close range, powerful enough
to cause interference with poorly shielded electromagnetic fields such as those found in mobile medical
devices and cheap consumer electronics.
Radio frequency
Radio waves
Radio waves carry information by varying amplitude and by varying frequency within a frequency band.
When EM radiation impinges upon a conductor, it couples to the conductor, travels along it, and
induces an electric current on the surface of that conductor by exciting the electrons of the
conducting material. This effect (the skin effect) is used in antennas. EM radiation may also cause
certain molecules to absorb energy and thus to heat up; this is exploited in microwave ovens.
Radio spectrum
ELF
SLF
ULF/VF
VLF
LF/LW
MW
HF/SW
VHF
UHF
SHF
EHF
3 Hz 30 Hz 300 Hz 3 kHz 30 kHz 300 kHz 3 MHz 30 MHz 300 MHz 3 GHz 30 GHz 300 GHz
Plot of Earth's atmospheric transmittance (or opacity) to various wavelengths of electromagnetic radiation.
Main article: radio frequency
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 126
The Professional Thermographers Association
Radio waves generally are utilized by antennas of appropriate size, with wavelengths ranging from
hundreds of meters to about one millimeter. They are used for transmission of data, via modulation.
Television, mobile phones, wireless networking and amateur radio all use radio waves.
Electric energy
Electrical energy covers the low-frequency, long-wavelength end of the spectrum. The radiation is usually
ducted along 2-wire and 3-wire transmission lines and sent to various devices besides antennas. At zero
frequency the energy is emitted by batteries and DC power supplies, while at 50 Hz and 60 Hz it is
produced by rotary magnetic generators and ducted through the international power grids. At frequencies
between 20 Hz to 30 kHz the EM energy is translated to and from acoustic energy and is distributed over
telephone lines, as well as being used to operate loudspeakers for public address or in music systems.
Note that other than its frequency, there is no functional difference between the VHF energy guided along a
television coaxial cable, versus the 60 Hz traveling along the cord leading to a light bulb. When connected
to the appropriate antenna, both will radiate into space.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 127
The Professional Thermographers Association
Infrared
Infrared (IR) radiation is electromagnetic
radiation of a wavelength longer than visible light,
but shorter than microwave radiation. The name
means "below red" (from the Latin infra, "below"),
red being the color of visible light of longest
wavelength. Infrared radiation spans three orders
of magnitude and has wavelengths between
700 nm and 1 mm.
Different regions in the infrared
IR is often subdivided into:
Near infrared NIR, IR-A DIN, 0.7–1.4 µm in wavelength, defined by the water absorption, and
commonly used in fiber optic telecommunication because of low attenuation losses in the SiO2
glass (silica) medium.
Short wavelength IR SWIR, IR-B DIN, 1.4–3 µm, water absorption increases significantly at
1.450 µm
Mid wavelength IR MWIR, IR-C DIN, also intermediate-IR (IIR), 3–8 µm
Long wavelength IR LWIR, IR-C DIN, 8–15 µm
Far infrared FIR, 15–1,000 µm
However, these terms are not precise, and are used differently in various studies i.e. near (0.7–5 µm) / mid
(5–30 µm) / long (30–1000 µm). Especially at the telecom-wavelengths the spectrum is further subdivided
into individual bands, due to limitations of detectors, amplifiers and sources. Infrared radiation is often
linked to heat, since objects at room temperature or above will emit radiation mostly concentrated in the
mid-infrared band.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 128
The Professional Thermographers Association
Plot of atmospheric transmittance in part of the infrared region.
The common nomenclature is justified by the different human response to this radiation (near infrared = the
red you just cannot see, far IR = thermal radiation), other definitions follow different physical mechanisms
(emission peaks, vs. bands, water absorption) and the newest follow technical reasons (The common
silicon detectors are sensitive to about 1050 nm, while InGaAs sensitivity starts around 950 nm and ends
between 1700 and 2200 nm, depending on the specific configuration). Unfortunately the international
standards for these specifications are not currently available.
The Earth as an infrared emitter
The Earth's surface absorbs visible radiation from the sun and re-emits much of the energy as infrared back
to the atmosphere. Certain gases in the atmosphere, chiefly water vapor, but also carbon dioxide,
methane, nitrous oxide, sulfur hexafluoride, and chlorofluorocarbons, absorb this infrared, and reradiate it in all directions including back to Earth. Thus, the greenhouse effect, keeps the atmosphere and
surface much warmer than if the infrared absorbers were absent from the atmosphere.
Applications for Infrared
Night Vision
Infrared is used in night-vision equipment, when there is insufficient visible light to see an object. The
radiation is detected and turned into an image on a screen, hotter objects showing up brighter, enabling the
police and military to acquire thermally significant targets, such as human beings and automobiles.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 129
The Professional Thermographers Association
.
Other Imaging
In infrared photography, infrared filters are used to capture only the infrared spectrum. Digital cameras
often use infrared blockers. Cheaper digital cameras and some camera phones which do not have
appropriate filters can "see" infrared, appearing as a bright white color (try pointing a TV remote at your
digital camera). This is especially pronounced when taking pictures of subjects near bright areas (such as
near a lamp), where the resulting infrared interference can wash out the image.
Thermography
Infrared radiation can be used to remotely determine the temperature of objects (if the emissivity is known).
This is termed thermography, or in the case of very hot objects in the NIR or visible it is termed pyrometry.
Thermography (thermal imaging) is mainly used in military and industrial applications but the technology is
reaching the public market in the form of infrared cameras on cars due to the massively reduced production
costs.
Smoke is more transparent to infrared than to visible light, so fire fighters use infrared imaging equipment
when working in smoke-filled areas
Heating
Infrared radiation is used in Infrared saunas to heat the sauna's occupants and to remove ice from the
wings of aircraft (de-icing).
Communications
IR data transmission is also employed in short-range communication among computer peripherals and
personal digital assistants. These devices usually conform to standards published by IrDA, the Infrared
Data Association. Remote controls and IrDA devices use infrared light-emitting diodes (LEDs) to emit
infrared radiation which is focused by a plastic lens into a narrow beam. The beam is modulated, i.e.
switched on and off, to encode the data. The receiver uses a silicon photodiode to convert the infrared
radiation to an electric current. It responds only to the rapidly pulsing signal created by the transmitter, and
filters out slowly changing infrared radiation from ambient light. Infrared communications are useful for
indoor use in areas of high population density. IR does not penetrate walls and so does not interfere with
other devices in adjoining rooms.
Free space optical communication using infrared lasers can be a relatively inexpensive way to install a
Gigabit/s communications link in urban areas, compared to the cost of burying fibre optic cable.
Infrared lasers are used to provide the light for optical fibre communications systems. Infrared light with a
wavelength around 1330 nm (best transmission) or 1550 nm (least dispersion) are the best choices for
standard silica fibres.
Spectroscopy
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 130
The Professional Thermographers Association
Infrared radiation spectroscopy is the study of the composition of (usually) organic compounds, finding out
a compound's structure and composition based on the percentage transmittance of IR radiation through a
sample. Different frequencies are absorbed by different stretches and bends in the molecular bonds
occurring inside the sample. Carbon dioxide, for example, has an absorption band at 4.2µm.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 131
The Professional Thermographers Association
Stefan-Boltzmann law
Stefan-Boltzmann law (also Stefan's law) states that the total energy radiated per unit surface area of a
black body in unit time (black-body irradiance), (or the energy flux density (radiant flux) or the emissive
power), j* is directly proportional to the fourth power of its thermodynamic temperature T:
The non-fundamental constant of proportionality is called the Stefan-Boltzmann constant or the Stefan's
constant σ. Its value is
Thus at 100 K the energy flux density is 5.67 W/m2,
at 1000 K 56.7 kW/m2, etc.
STEFFAN-BOLTZMAN
STEFFAN-BOLTZMAN LAW:
LAW:
This
This law
law describes:
describes:
•• The
The total
totalenergy
energyradiated
radiatedfrom
fromthe
the
surface
surface of
ofaa object
objectat
ataaspecific
specific
The law was experimentally discovered by Jožef
temperature.
temperature.
Stefan (1835-1893) in 1879 and theoretically
-8
4
q”
q” == 5.678
5.678 XX10
10-8XXAAXXTT4(K)
(K)
derived in the frame of the thermodynamics by
Ludwig Boltzmann (1844-1906) in 1884. Boltzmann
This
Thisformula
formula shows
showsthat:
that:
•• IfIf the
thetemperature
temperatureof
ofan
anobject
objectdoubles,
doubles,
treated a certain ideal heat engine with the light as
4
•• ItIt will
will radiate
radiate infrared
infraredenergy
energyat
ataarate
rateof
of224
–
Or
16
times
more.
This
means
that
– Or 16 times more. This means thataasmall
small
a working matter instead of the gas. This law is the
increase
increaseinintemperature
temperatureequals
equalsaalarge
largeincrease
increase
ininthe
only physical law of nature named after a Slovene
theradiated
radiatedenergy
energy
Graphically
Graphicallyititrepresents
representsthe
thearea
areaunder
underthe
the
physicist. The law is valid only for ideal black
Planck
Planck curve
curvefor
foraaparticular
particulartemperature
temperature
objects, the perfect radiators, called blackbodies.
Stefan published this law on March 20 in the article
Über die Beziehung zwischen der Wärmestrahlung und der Temperatur (On the relationship between
thermal radiation and temperature) in the Bulletins from the sessions of the Vienna Academy of Sciences.
Derivation of the Stefan-Boltzmann law
The Stefan-Boltzmann law can be easily derived by integrating the emitted intensity from the surface of a
black body given by Planck's law of black body radiation over the half-sphere into which it is emitted, and
over all frequencies.
where Ω0 is the half-sphere into which the radiation is emitted, and Iν is the amount of the black body
emitted energy per unit surface per unit time per unit solid angle. The cosine factor is included because the
black body is a perfect Lambertian radiator. Using dΩ= sin(θ) dθdφ and integrating yields:
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 132
The Professional Thermographers Association
Temperature of the Sun
With his law Stefan also determined the temperature of the Sun's surface. He learnt from the data of
Charles Soret (1854–1904) that the energy flux density from the Sun is 29 times greater than the energy
flux density of a warmed metal lamella. A round lamella was placed at such a distance from the measuring
device that it would be seen at the same angle as the Sun. Soret estimated the temperature of the lamella
to be circa 1900 °C to 2000 °C. Stefan surmised that 1/3 of the energy flux from the Sun is absorbed by the
Earth's atmosphere, so he took for the correct Sun's energy flux a value 3/2 times greater, namely 29 × 3/2
= 43.5. Precise measurements of atmospheric absorption were not made until 1888 and 1904. The
temperature Stefan obtained was a median value of previous ones, 1950 °C and the absolute
thermodynamic one 2200 K. As 2.574 = 43.5, it follows from the law that the temperature of the Sun is 2.57
times greater than the temperature of a lamella, so Stefan got a value of 5430 °C or 5700 K (modern value
is 5780 K). This was the first sensible value for the temperature of the Sun. Before this, values from circa
1800 °C to 13,000,000 °C were claimed. The first value of 1800 °C was determined by Claude Servais
Mathias Pouillet (1790-1868) in 1838 using the Dulong-Petit law. Pouilett also took just half the value of the
Sun's correct energy flux. Perhaps this result reminded Stefan that the Dulong-Petit law could break down
at large temperatures. If we collect the Sun's light with a lens, we can warm a solid to much higher
temperature than 1800 °C.
The Stefan-Boltzmann law is an example of a power law.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 133
The Professional Thermographers Association
Gustav Kirchhoff
Gustav Robert Kirchhoff (March 12, 1824 –
October 17, 1887), a German physicist who
contributed to the fundamental understanding of
electrical circuits, spectroscopy, and the emission
of black-body radiation by heated objects. He
coined the term "black body" radiation in 1862,
and two sets of independent concepts in both
circuit theory and thermal emission are named
"Kirchhoff's laws" after him.
Gustav Kirchhoff was born in Königsberg, Prussia
(now Kaliningrad, Russia), the son of Friedrich
Kirchhoff, a lawyer, and Johanna Henriette Wittke.
He graduated from the Albertus University of
Königsberg (now Kaliningrad) in 1847 and married
Clara Richelot, the daughter of his mathematics
professor Friedrich Richelot. In the same year, they
moved to Berlin, where he stayed until he received
a professorship at Breslau (now Wroclaw).
Kirchhoff formulated his circuit laws, which are now
ubiquitous in electrical engineering, in 1845, while still a student. He proposed his law of thermal radiation
in 1859, and gave a proof in 1861. At Breslau, he collaborated in spectroscopic work with Robert Bunsen,
he was a co-discoverer of caesium and rubidium in 1861 while studying the chemical composition of the
Sun via its spectral signature.
Later, he postulated three empirical laws describing the spectral composition of light emitted by
incandescent objects.
1. A hot solid object produces light with a continuous spectrum.
2. A hot tenuous gas produces light with spectral lines at discrete wavelengths (i.e. specific colors)
which depend on the energy levels of the atoms in the gas. (See also: emission spectrum)
3. A hot solid object surrounded by a cool tenuous gas (i.e. cooler than the hot object) produces light
with an almost continuous spectrum which has gaps at discrete wavelengths depending on the
energy levels of the atoms in the gas.
The existence of these discrete lines was later explained by the Bohr model, which helped lead to the
development of quantum mechanics.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 134
The Professional Thermographers Association
Kirchhoff's law of thermal radiation
.
Kirchhoff's law in thermodynamics, also called e.g. Kirchhoff's law of thermal radiation, is a general
statement equating emission and absorption in heated objects, proposed by Gustav Kirchhoff in 1859 (and
proved in 1861), following from general considerations of thermodynamic equilibrium. (See Kirchhoff's laws
for other laws named after Kirchhoff.)
An object at some non-zero temperature radiates electromagnetic energy. If it is a perfect black body,
absorbing all light that strikes it, it radiates energy according to the blackbody radiation formula. More
generally, it radiates with some emissivity multiplied by the blackbody formula. Kirchhoff's law states that:
At thermal equilibrium, the emissivity of a body (or surface) equals its absorptivity.
A = E where A : Absorpitivity and E : Emissivity or Emission
Here, the absorptivity (or absorbance) is the fraction of incident light (power) that is absorbed by the
body/surface. In the most general form of the theorem, this power must be integrated over all wavelengths
and angles. In some cases, however, emissivity and absorption may be defined to depend on wavelength
and angle, as described
below.
A
A black
black body,
body, defined
defined by
by Gustav
Gustav Kirchoff
Kirchoff
Kirchhoff's Law has a
corollary: the emissivity cannot
•• An
An object
object which
which at
at all
alltemperatures
temperatures
absorbs
absorbs completely
completelyall
allthe
theradiation
radiationfalling
falling
exceed one (because the
on
on it.
it.
absorptivity cannot, by
•• Furthermore,
Furthermore, at
at any
anygiven
giventemperature
temperatureitit
emits
emits aa characteristic
characteristic continuous
continuous spectrum
spectrum
conservation of energy), so it
of
of wavelengths
wavelengths of
of radiation
radiation that
thatdepends
depends
is not possible to thermally
only
only on
on the
the temperature
temperature of
of the
theblack
black body
body
and
and not
not what
what it's
it's made
madeof.
of.
radiate more energy than a
•• While
While an
an ideal
ideal black
black body
bodyisis not
notreally
really
blackbody, at equilibrium. This
found
found in
in practice,
practice, itit can
canbe
be approximated
approximated
by
by aa hollow
hollow enclosure,
enclosure, painted
paintedblack
black on
on
has two caveats, however.
the
inside,
with
aa very
small
opening
in
the
inside,
with
very
small
opening
inthe
the
100
Watts
100
Watts
wall.
So,
radiation
entering
the
hole
has
First, if the surface is
wall. So, radiation entering the hole has
little
little chance
chance of
of escaping;
escaping;rather
ratherititisis
diffractive, so that incident
gradually
gradually absorbed
absorbed by
by repeated
repeatedcontact
contact
with
energy at one angle is partially
with the
the internal
internalsurfaces
surfaces of
of the
theobject.
object.
•• Similarly,
Similarly, ifif the
the enclosure
enclosure isis heated,
heated,the
the
reflected to another angle,
radiation
radiation coming
coming from
from the
thehole
hole isis
100
Watts
effectively
100 Watts
then the emissivity at one
effectively black
black body
body radiation.
radiation.
100
Watts
100 Watts
angle can exceed unity, but
An
objects
ability
to
absorb
An objects ability to absorbenergy
energyis
isequal
equal
to
to its
its ability
abilityto
toemit
emitenergy:
energy: Therefore
ThereforeAA==EE
not the emissivity of power
integrated over all angles.
Second, if the object is nonlinear (e.g. fluorescent), so that incident power at one wavelength is re-emitted
at another wavelength, then the emissivity at some wavelengths can exceed unity, but not the emissivity of
power integrated over all wavelengths.
Absorbs
Absorbs50
50watts
watts
Reflects
Reflects50
50watts
watts
Absorbs
Absorbs25
25watts
watts
Reflects 25 watts
Reflects 25 watts
Absorbs
Absorbs6.25
6.25watts
watts
Reflects
Reflects6.25
6.25watts
watts
100
100watts
watts
Absorbs 12.5 watts
Absorbs 12.5 watts
Reflects 12.5 watts
Reflects 12.5 watts
This theorem is sometimes informally stated as a poor reflector is a good emitter, and a good reflector is a
poor emitter. It is why, for example, lightweight emergency thermal blankets are typically made of reflective
material: they lose little heat by radiation.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 135
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 136
The Professional Thermographers Association
Black body
As the temperature decreases, the
peak of the black body radiation
curve moves to lower intensities
and longer wavelengths. The
black-body radiation graph is also
compared with the classical model
that preceded it.
In physics, a black body is an
object that absorbs all
electromagnetic radiation that falls
onto it. No radiation passes
through it and none is reflected.
Despite the name, black bodies
are not actually black as they
radiate energy as well. How much
electromagnetic radiation they give off just depends on their temperature. Black bodies below around
700K produce very little radiation at visible wavelengths and appear black. Black bodies above this
temperature however, start to produce radiation at visible wavelengths starting at red, going through
orange, yellow and white before ending up at blue as the temperature increases.
The term "black body" was introduced by Gustav Kirchhoff in 1862. The light emitted by a black body
is called black-body radiation1.
Details
In the laboratory, the closest thing to black-body radiation is the radiation from a small hole entrance
to a larger cavity. Any light entering the hole would have to reflect off the walls of the cavity multiple
times before it escaped and is almost certain to be absorbed by the walls in the process, regardless
of what they are made of or the wavelength of the radiation (as long as it is small compared to the
hole). The hole then is a close approximation of a theoretical black body and if the cavity is heated,
the spectrum of the hole's radiation (i.e. the amount of light emitted from the hole at each
wavelength) will be continuous, and will not depend on the material in the cavity (compare with
Emission spectrum). By a theorem proved by Kirchhoff, this curve depends only on the temperature
of the cavity walls.
Calculating this curve was a long-standing challenge that was solved in 1900 by Max Planck as
Planck's law of black-body radiation. To calculate the curve, Planck had to assume that
electromagnetic radiation could propagate only in discrete packets, or quanta. This idea was later
used by Einstein in 1905 to explain the photoelectric effect. These theoretical advances eventually
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 137
The Professional Thermographers Association
resulted in the replacement of classical electromagnetism by quantum mechanics. Today, the quanta are
called photons.
It was later realized that the observed spectrum of black-body radiation can not be explained by classical
electromagnetism and statistical mechanics: these predict infinite brightness at high frequencies (i.e. low
wavelength), a prediction often called the ultraviolet
catastrophe.
The temperature of a Pahoehoe lava flow can be
approximated by merely observing its colour. The result
agrees nicely with the measured temperatures of lava
flows at about 1,000 to 1,200 °C (1,273 to 1,473 kelvins).
The wavelength at which the radiation is strongest is
given by Wien's law, and the overall power emitted per unit area is given by the Stefan-Boltzmann law.
So, as temperature increases, the glow color changes from red to yellow to white to blue. Even as the peak
wavelength moves into the ultra-violet enough radiation continues to be emitted in the blue wavelenths that
the body will continue to appear blue. It will never become invisible—indeed, the radiation of visible light
increases monotonically with temperature.
The radiance or observed intensity is not a function of direction. Therefore a black body is a perfect
Lambertian radiator.
Real objects never behave as full-ideal black bodies, and instead the emitted radiation at a given frequency
is a fraction of what the ideal emission would be. The emissivity of a material specifies how well a real
body radiates energy as compared with a black body. This emissivity depends on factors such as
temperature, emission angle, and wavelength.
A typical engineering assumption (and a mistake!) is to assume that a surface's spectral emissivity and
absorptivity do not depend on wavelength, so that the emissivity is a constant. This is known as the grey
body assumption.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 138
The Professional Thermographers Association
The spectrum of an incandescent bulb in a
typical flashlight immediately reveals the
mechanism of incandescent lighting to be
blackbody radiation. Here, the filament
temperature appears to be about 4,600
Kelvins (7,820 F or 4,326 C) due to a
peak emittance of around 630 nanometers
or .63 microns.
When dealing with non-black surfaces, the
deviations from ideal black body behavior
are determined by both the geometrical
structure and the chemical composition,
and follow Kirchhoff's Law: emissivity
equals absorptivity, so that an object that
does not absorb all incident light will also emit less radiation than an ideal black body.
Equations governing black bodies
Planck's law of black-body radiation
where
is the amount of energy per unit surface per unit time per unit solid angle
emitted in the frequency range between ν and ν+dν;
is the temperature of the black body;
is Planck's constant;
is the speed of light; and
is Boltzmann's constant.
Wien's displacement law
The relationship between the temperature T of a black body, and wavelength λmax at which the
intensity of the radiation it produces is at a maximum is:
Stefan-Boltzmann law
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 139
The Professional Thermographers Association
The total energy radiated per unit area per unit time (
and the Stefan-Boltzmann constant σ as follows:
) by a black body is related to its temperature T
Derivation of temperature relation between earth and sun
Assumptions
If we assume the following:
1. That the Earth and the Sun behave like black bodies (which is true to a good approximation), and
2. That the earth is in thermal equilibrium (or in other words, its average temperature isn't changing)
then we can derive a formula for the relationship between the earth's surface temperature and the sun's
surface temperature.
Derivation
To begin, let's use the Stefan-Boltzmann law to find the total power (energy/second) the sun is emitting:
where
is the Stefan-boltzmann constant
is the surface temperature of the sun
is the radius of the sun
The sun emits that power equally in all directions. Because of this, the earth is hit with only a tiny fraction of
it. This is the power from the sun that the earth absorbs:
where
is the radius of the earth
is the distance between the sun and the earth
Even though the earth only absorbs as a circular area πR2, it emits equally in all directions as a sphere:
where TE is the surface temperature of the earth
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 140
The Professional Thermographers Association
Now, in the second assumption the earth is in thermal equlibrium, so the power absorbed must equal the
power emitted:
So plug in equations 1, 2 and 3 into this and we get:
Many factors cancel from both sides and this equation can be greatly simplified.
The result
After canceling of factors, the final result is:
where
is the surface temperature of the sun
is the radius of the sun
is the distance between the sun and the earth
is the average surface temperature of the earth
In other words, the temperature of the earth only depends on the surface temperature of the sun, the radius
of the sun, and the distance between the earth and the sun.
Temperature of the sun
If we plug in the measured values:
We'll find the surface temperature of the sun to be:
Which is roughly 9,900 degrees fahrenheit, or about 5,500 degrees celsius. This isn't too far off from the
actual temperature, 5780 K.
Footnotes
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 141
The Professional Thermographers Association
Note 1: When used as a compound adjective, the term is typically hyphenated, as in "black-body radiation",
or combined into one word, as in "blackbody radiation". The hyphenated and one-word forms should not
generally be used as nouns, however.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 142
The Professional Thermographers Association
Planck's law of black body radiation
.
Black body spectrum as a function of wavelength
In physics, the spectral intensity of electromagnetic radiation from a black body at
temperature T is given by the Planck's law of black body radiation:
where:
I(ν) is the amount of energy per unit time per unit
surface area per unit solid angle per unit frequency.
Units are e.g. [W m-2 Hz-1 sr-1];
ν is the frequency
T is the temperature of the black body
h is Planck's constant,:
c is the speed of light
k is Boltzmann's constant.
The law is sometimes written in terms of the spectral energy
density
1.E+08
1.E+08
Planck
Radiation
Curves
Planck
Radiation
Curves
Planck
PlanckRadiation
RadiationCurves
Curves
00
32
32
°C
°C
67
21
67
21
212
100
100 212
275
275 527
527
525
977
525 977
2600
2600 4,712
4,712
5727
10,340
572710,340
Wien's
Wien'sLaw
Law
1.E+04
1.E+04
1.E+03
1.E+03
1.E+02
1.E+02
IRFilm
Film
IR
1.E+05
1.E+05
1.E+01
1.E+01
LW
LW
1.E+00
1.E+00
1.E-01
1.E-01
SW
SW
Max Planck originally produced this law in 1900
(published in 1901) in an attempt to improve upon an
expression proposed by Wilhelm Wien which fit the
experimental data at short wavelengths but deviated from
it at long wavelengths. He found that the above function
1.E+06
1.E+06
visual
visual
which has units of energy per unit volume per unit
frequency.
EmissivePower
Power(watts
(watts/ /sq.
sq.meter-µm)
meter-µm)
Emissive
1.E+07
1.E+07
1.E-02
1.E-02
1.E-03
1.E-03
0.1
0.1
11
Wavelength
Wavelength(µm)
(µm)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
10
10
Page: 143
100
100
The Professional Thermographers Association
fit the data for all wavelengths remarkably well. In constructing a derivation of this law, he considered the
possible ways of distributing electromagnetic energy over the different modes of charged oscillators in
matter. Planck's law emerges if it is assumed that these oscillators have energy proportional to frequency
.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 144
The Professional Thermographers Association
Wien's displacement law
Wien's displacement law is a law of physics that states that there is an inverse relationship between
the wavelength of the peak of the emission of a black body and its temperature.
where T is the temperature of the blackbody in kelvins (K) and λmax is the peak wavelength in
meters. The 0.002898... is a proportionality constant with units meter-kelvins (m·K). It is sometimes
expressed in cgs units as 0.29 cm·K. (or in Infrared Thermography as 2898 for Mikrons!)
Basically, the hotter an object is, the
shorter the wavelength at which it will
emit radiation. For example, the surface
temperature of the sun is 5780 K, giving a
peak at 500 nm. As can be seen in the
article Color, this is fairly in the middle of
the visual spectrum, due to the spread
resulting in white light. Due to the Rayleigh
scattering of blue light by the atmosphere
this white light is separated somewhat,
resulting in a blue sky and a yellow sun.
A lightbulb has a glowing wire with a
somewhat lower temperature, resulting in
yellow light, and something that is "red hot"
is again a little less hot.
Wien’s
Wien’s Displacement
Displacement Law
Law
Wilhelm
WilhelmWien
Wien 1864
1864 -- 1928
1928
recognized
recognized that
that for
for any
anyone
one
temperature
temperature there
there isis one
one
wavelength
wavelength at
at which
which aa maximum
maximum
amount
amount of
of energy
energy isis emitted
emitted by
byaa
body.
body.
2898
max
2898m
m
max
TT
This
This formula
formula isis useful
useful to
to the
the
thermographer
thermographer in
in determining,
determining,
theoretically,
theoretically, at
at what
what wavelength
wavelength
the
the object
object of
of interest
interest would
would emit
emit aa
maximum
maximum amount
amount of
of energy.
energy.
Although the law was first formulated by Wilhelm Wien, we now derive it from Planck's law of black body
radiation.
This can be turned into an equation for the frequency of the radiation, using the speed of light to get
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 145
The Professional Thermographers Association
Emissivity
The emissivity of a material (usually written e) is the ratio of energy radiated from a object as
compared to energy radiated by a black body at the same temperature and wavelength. It is a
measure of a material's ability to absorb and radiate energy. A true black body would have an e = 1 while
any real object would have e < 1.
This emissivity depends on factors such as temperature, emission angle, and wavelength. However, a
typical engineering assumption is to assume that a surface's spectral emissivity and absorptivity do not
depend on wavelength, so that the emissivity is a constant. This is known as the grey body assumption.
When dealing with non-black surfaces, the deviations from ideal black body behavior are determined by
both the geometrical structure, surface chemical composition influences and wavelignth, and follow
Kirchhoff's Law: emissivity equals absorptivity, so that an object that does not absorb all incident light will
also emit less radiation than an ideal black body.
How to do an emissivity test
To do an emissivity test you will need to know either:
1. What the true temperature of the object is
2. Have a correct emissivity reference on the object.
Reference Emissivity Method
Working from a known emissivity on the object you would want to:
1.
2.
3.
4.
Set the correct emissivity value for the reference area/emissivity that you want to measure.
Measure the temperature of the object
Note what the correct temperature of the object is
Move your measurement tool (Spot, Isotherm, Area, etc.) to the area next to the reference
area/emissivity that you want to calculate.
5. Change your emissivity till the temperature from your reference area/emissivity are the
same.
Known Temperature Method
By obtaining the actual temperature of the surface that you are trying to calculate the emissivity for,
by using a contact thermometer you can calculate the emissivity by:
1. Image the area where the contact measurement was made and place your measurement
tool (Spot, Isotherm, Area, etc.) as close to the same location as the contact thermometer
was at the time of the reading.
2. Change your emissivity setting till the temperature in your camera matches the reading
that you got from your contact temperature device.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 146
The Professional Thermographers Association
Emissivity Table Examples:
(DO NOT USE IN YOUR IR CAMERA) These tables are for calculating the heat transfer from the
surface of objects by engineer doing heat loss calculations for comparisons’ only…never use them
in your IR camera. You will need to develop your own tables based on the object and environments
that you are working in.
Remember that emissivity of an object can change with:
The temperature of the object.
The wavelength of the camera.
The angle of viewing
The shape of the object
MATERIAL
TEMP °F (°C)
e-EMISSIVITY
Aluminium
Unoxidized
77 (25)
.02
Unoxidized
212 (100)
.03
Unoxidized
932 (500)
.06
Oxidized
390 (199)
.11
Oxidized
1110 (599)
.19
Oxidized at 1110¡F (599¡C)
390 (199)
.11
Oxidized at 1110¡F (599¡C)
1110 (599)
.19
Heavily Oxidized
200 (93)
.20
Heavily Oxidized
940 (504)
.31
Highly Polished
212 (100)
.09
Roughly Polished
212 (100)
.18
Commercial Sheet
212 (100)
.09
Highly Polished Plate
440 (227)
.04
Highly Polished Plate
1070 (577)
.06
Bright Rolled Plate
338 (170)
.04
Bright Rolled Plate
932 (500)
.05
Alloy A3003, Oxidized
600 (316)
.40
Alloy A3003, Oxidized
900 (482)
.40
200-800 (93-427)
.05
Alloy 24ST
75 (24)
.09
Alloy 24ST, Polished
75 (24)
.09
Alloy 75ST
75 (24)
.11
Alloy 1100-0
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 147
The Professional Thermographers Association
Alloy 75ST, Polished
MATERIAL
75 (24)
TEMP °F (°C)
.08
e-EMISSIVITY
Copper
Cuprous Oxide
100 (38)
.87
Cuprous Oxide
500 (260)
.83
Cuprous Oxide
1000 (538)
.77
Black, Oxidized
100 (38)
.78
Etched
100 (38)
.09
Matte
100 (38)
.22
Roughly Polished
100 (38)
.07
Polished
100 (38)
.03
Highly Polished
100 (38)
.02
Rolled
100 (38)
.64
Rough
100 (38)
.74
Molten
1000 (538)
.15
Molten
1970 (1077)
.16
Molten
2230 (1221)
.13
100-500 (38-260)
.37
Nickel Plated
MATERIAL
TEMP °F (°C)
e-EMISSIVITY
Steel
Cold Rolled
200 (93)
.75-.85
Ground Sheet
1720-2010 (938-1099)
.55-.61
Polished Sheet
100 (38)
.07
Polished Sheet
500 (260)
.10
Polished Sheet
1000 (538)
.14
Mild Steel, Polished
75 (24)
.10
Mild Steel, Smooth
75 (24)
.12
Mild Steel, Liquid
2910-3270 (1599-1793)
.28
Steel, Unoxidized
212 (100)
.08
77 (25)
.80
Steel Oxidized
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 148
The Professional Thermographers Association
MATERIAL
Wood
TEMP °F (°C)
Low
eEMISSIVITY
.80-.90
Beech, Planed
158 (70)
.94
Oak, Planed
100 (38)
.91
Spruce, Sanded
100 (38)
.89
Emissivity Tables on the Web:
http://www.electro-optical.com/bb_rad/emissivity/matlemisivty.htm
http://www.omega.com/literature/transactions/volume1/emissivity.html
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 149
The Professional Thermographers Association
Glossary of terms
A
A-scope
A graph presentation of signal versus time or signal versus space. A simplified display of the progressive temperature changes in
an object over time or the thermal gradient along a cursor line.
Aberration
Any undesired factor that distorts an image. Typical sources of image aberrations are noise, signal crosstalk, external signal
pickup; optical effects such as chromatic aberration, vignetting, video effects such as barrel distortion, pin cushion; digitizing
effects such as pixel clock crosstalk, video leveling, inadequate sampling, etc.
Absolute Temperature Scale
A temperature measurement scale based on the coldest possible temperature equal to 0. (See Rankine and Kelvin)
Absorptance (Absorptivity)
The ratio between the amount of energy absorbed and the total energy incident (irradiance), usually measured at a surface. The
absorptance of a surface is equal to its emissivity.
Accuracy
A measure of the similarity of an instrument reading to the actual value for that reading. Instrument accuracy is affected by many
things -instrument drift, environment, temperature, time, operator expertise, etc.
Active Element
The area on a detector which responds to radiation by creating a signal output. Many detectors contain both unused (inactive)
elements and unused areas which are not part of the active, or responsive element area.
Active IR system
A system using an (active) IR source to illuminate a scene - much like a visible image scene is illuminated with a floodlight. An
active IR system monitors the illuminated scene in much the same way as a passive system.
Afocal
Usually refers to radiation in image space for a system adjusted for infinity focus. This radiation is fairly telecentric, having a very
long focal length (no focal length).
AGA
AGEMA
Airy Disc
A measurement of the resolving capability of a lens expressing the diameter of the minimum spot size. Depends on lens,
aperture and on the wavelength of passband radiation. (A.D. = 1.2 x lambda / A)
Albedo
The ratio between the amount of energy reflected and the total energy incident (irradiance), usually measured at a surface. The
albedo of a surface is equal to its reflectance.
Ambient temperature
The temperature of the surrounding air and environment considered the baseline measurement for heat transfers around an
object.
Amorphous
Having an irregular structure. Amorphous crystal structures are more useful in optical assemblies since they are not as prone to
fracture along a crystal boundary. Crystals can sometimes be converted from mono-crystalline to poly-crystalline to amorphous
by proper annealing.
Annealing
The process of heating and cooling a material at a controlled rate to improve its internal stresses and modify its internal
crystalline or chemical structure. Annealing can make metals and optical materials more flexible and less brittle.
Antireflective Coating
A thin layer of material applied to the surface of an optical element to increase the transmission of the element by reducing
bascattering due to surface reflections. A simple A/R coating will be comprised of a one-quarter wavelength thick coating of a
material whose refractive index is between the indexes of the air and the element material.
Angstrom
One millimicron; a unit to measure the wavelength of high frequency electromagnetic radiation such as visible light.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 150
The Professional Thermographers Association
Aperture
A hole or opening limits the amount of radiation used by an optical system. The aperture of a system may be set by the size of
the lens or window bounding the cross section of the radiation bundle.
Aperture Stop
The limiting aperture of an optical system, usually where the system energy bundle size is fully determined. Multiple aperture
stops in a system with moving optical elements will usually lead to image aberrations and vignetting.
Apparent Temperature
The temperature determined for an object calculated by a remote infrared sensor based solely on the amount of radiation it emits
assuming the object has Blackbody emission characteristics.
Aspect Ratio
The ratio of the vertical length to the horizontal width. For a television display the aspect ratio is 3:4.
Atmospheric Windows
Spectral radiation regions not absorbed by atmospheric gasses. These windows are transparent to radiation at those
wavelengths. The most obvious window is the visible light window - if the 'smog is not too bad we can see through the
atmosphere forever.
Atmospheric Attenuation Atmospheric Absorption
The amount of signal reduction occurring when IR radiation travels through the atmosphere between the target and imaging
system. The amount of attenuation can be very small on clear days with short transmission paths and can be very large on
foggy, smoggy, dusty days with long transmission paths. Attenuation The reduction in intensity of radiation after processing
through a filter. This can be measured in dB.
Azimuth Angle
The angle measurement of horizontal dimension on an optically generated display. Normally zero azimuth is taken to be the
center of the field of view and dimensions to the right and left are positive and negative azimuth angles.
B
Back Surface Mirror
A mirror who's reflective coating is on the opposite side of the optical material from the incident radiation. Back surface coating is
used often in visible optics to help protect the mirror surface. Back surface coating is normally not used in infrared optics due to
the additional attenuation caused by the passage of radiation through the optical element.
Backscattering
The re-reflection of thermal energy generated on the ground and reflecting off the underside of clouds or inversion layers. Also
can refer to unwanted front surface reflections off of a transparent optical element.
Background Noise
The noise present in a sensor (detector) independent of the signal strength or ambient temperature. Normally caused by thermal,
generant-recombinant characteristics or 1/f effects.
Background Temperature
The average temperature of the environment around an object under study. The background temperature creates the radiation
level available as reflected energy.
Bandpass Filter
A filter that is transparent for a range of wavelengths and opaque (reflective or absorptive) for wavelengths outside this bandpass
region.
Bandwidth
A range of wavelengths or frequencies passed or processed. Typically used in both electronics and optics to designate the
operating range of a filter or electronic component.
Bar
A unit for measuring gas pressure in the cgs system. one bar is equal to 10 6 dynes per square centimeter. (29.52 in. of Mercury)
Bar-Code Reader
An instrument providing an alternate means of entering data into a computer by reading a series of machine printed parallel lines
off of a product label. The ASCII data is encoded in the width of the parallel lines or bars and the spacing between bars.
Barrel Distortion
Image distortion that spatially expands the center of a display and contracts the corners to make rectangular display objects
appear barrel shaped.
Barrier Potential
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 151
The Professional Thermographers Association
In a photonic detector, the width of the energy gap between the conduction band and the valence band. This sets the maximum
wavelength of incident radiation for detector response. Beam A stream or column of particles or rays, a bundle of radiation. The
beam size is usually limited by the aperture of an optical system.
Bias (Supply, Current, Voltage)
Electrical current or voltage generated in a low noise, highly isolated supply applied to a photo-conductive detector through
selected load resistors so that the detector response to photons (a resistance change) can be measured.
Binocular
A display device providing a separate display for each eye.
Biocular
A viewing device made for display images to both eyes simultaneously
Blackbody
An ideal thermal radiator, usually one whose temperature can be controlled, radiating and absorbing the maximum possible
thermal radiation for the set temperature. (Emissivity = 1.0, Reflectance = 0.0, Transmittance = 0.0)
Blur Circle
The smallest size focused spot a lens system can produce at its focal plane.
Bolometer
An temperature measuring instrument using a strip thermistor to achieve higher sensitivity than a simple thermistor. Unlike
thermistors which are used for contact temperature measurements, bolometers have been used to measure radiation levels.
Boresite
The alignment of the sensing axis of a detector to the display axis so that the center of the display screen represents straight
ahead to the imager.
Brightness
A term to indicate the relative amount of light intensity available. Brighter is more light, dimmer is less light. In an infrared system,
the brightness control may affect actual image intensity or it may change the temperature range displayed. Either effect will
change the brightness of a given temperature.
British Thermal Unit (BTU)
The amount of thermal energy required to raise one pound of water one degree Fahrenheit at or near freezing.
Bundle
A defined, limited and usually concentrated beam of radiation rays. The ray bundle represents the radiation available to an
optical system for focusing on to a detector.
C
Calibration
The process of adjusting an instrument to read accurately. or in some cases, accurately constructing a table of errors so that the
instrument reading can be adjusted off-line or post measurement to correlate the reading accurately to the true value.
Calorie
The amount of thermal energy required to raise one gram of water one degree-Celsius at 15'C. Case Hardening A method of
strengthening the surface of metals or optical materials involving heating the surface and quenching rapidly with air or water;
leaving an amorphous layer of material on and just below the surface.
Cavity
Celsius (Centigrade)
A scale for measuring temperature where absolute cold is -273.2'C, the melting point of water (ice point) is 0 C, and the boiling
point of water is 100° C. Centimeter A unit of length measurement in the metric system. one hundred centimeters is equal to one
meter ( 2.54 cm. = 1")
Certified
As in "Certified Infrared Thermographer". A level of competence in understanding the laws of thermal dynamics with regard to
Infrared Thermography. A 40 hour course plus an exam that must be passed.
Channel Valance
See shading.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 152
The Professional Thermographers Association
Chopper
A means of optically blocking and unblocking the detector sensor repetitively. A chopper normally consists of a round, flat,
pancake-like blade rotated by a motor. The blade is cut so that a regular pattern of openings and blockages pass in front of the
detector.
Clear Aperture
The useful area of a lens. The edges of a lens can not be used due to edge effects in coating and due the mounting hardware
interfering with ray transmission. The clear aperture is usually specified to exclude the edges and is usually circular in diameter.
Cold Shield
A means of increasing the sensitivity of a detector by reducing the background noise. A cold shield is built within the detector
package and is cooled to a temperature close to the operating temperature of the detector. It blocks non-signal, high temperature
background photons from the detector and decreases the level of background noise.
Collimated
Traveling together in parallel, as in parallel light rays.
Color Scale
An area of the display screen set aside to display the full range of available colors. Used to demonstrate how each color is
assigned a temperature or radiation level.
Color Thermogram
A display of temperature differences which has been enhanced by the assignment of discrete color values to each of the thermal
intensity levels. This is considered the normal visual display mode for a thermal video system.
Conduction
The transfer of energy through a solid without motion of the conducting solid as a whole. Conduction Band The energy band
above the energy gap in a semiconductor. Electrons with enough energy to reach the conduction band can freely move within
the semiconductor in response to externally applied potential gradients.
Contrast
The relationship between the brightest intensity on a display and the dimmest. In an infrared system this may adjust the
temperature range on the display. Increasing and decreasing the display range of temperature will change the apparent
brightness difference between two different temperature values.
Convection
The transfer of energy through a liquid or gas due to the motion of the medium.
Coolant
A medium, usually gas or liquid that is either held at low temperatures or generates low temperatures when it expands used to
reduce the temperature of another object.
Coulomb
A quantity measurement electricity. one coulomb is equal to 6.3 x 1018 electrons.
Critical Angle
The smallest incident radiation angle at which total internal reflection occurs. This angle is defined using Snell's law, setting the
sine of the refraction angle of the lower index material equal to one.
Crosshair
A graphically-based cursor overlay for identifying locations on an image - represented as the intersection of a vertical and
horizontal line.
Crosstalk
A form of signal interference created when undesired signal radiation or current affects the signal being interpreted. This
interference can be from other currents and signals within the electrical module or can be picked up from radio, television, or
nearby switching.
Cryogen Cryogenics
Pertaining to extreme cold. A cryogen is a material which will create extremely cold conditions. Cryogenics is the study of
extreme cold. In infrared, cryogenics usually refers to the means used to reduce the detector temperature to a useful value.
Temperatures less than 200 K could be considered cryogenic.
Cryostat
A device for cooling detectors to cryogenic temperatures and maintaining these temperatures accurately. Operates on the
principal of Joule-Thompson expansion. Contains a small orifice. Gas under high pressure is put through the orifice and allowed
to expand rapidly. This creates a small field of extreme cold which is placed in the vicinity of the detector array.
Cursor
A means of identifying location on a video display. Cursors are usually supported by separate display fields than either text or
image data displays and can be moved by either graphics input devices or the keyboard. Typical cursor types include a text
cursor (underline or blinking block), graphics cursors (dots or crosshairs), and AOI cursors (circles,, rectangles, polygon shapes).
Cut-off Wavelength
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 153
The Professional Thermographers Association
The wavelength determined when the transmission of a filter drops below 5%.
Cut-on Wavelength
The wavelength of a filter where the transmission first exceeds 5%.
D
D-star (D*)
A measurement of the sensitivity of a detector material in terms of signal to noise ratio. D-star is normally expressed either as a
blackbody D-star or as a peak wavelength D-star within the practical operating frequency of the detector. The units of D-star are
centimeter-square root hertz per watt.
Dead band
The region of signals close enough to the set point of a controlling instrument that they do not initiate a cycle of corrective action.
Similar to the noise level of a monitoring instrument.
Degree
An increment of temperature measurement value. delta-T Small differences of temperature
Depth of Field
The range of object distances which are in focus around a set focus distance value (object space distance).
Depth of Focus
The range of motion of the focus assembly which around a set focus distance which will not defocus an object at that focus
distance (image space distance).
Detectivity
The ability of a detector to sense radiant power - the signal to noise ratio. The inverse of a noise power reading. Detectivity is
related to noise since the signal power must be at least as large as the noise power to be distinguished as signal - not noise.
Detector, IR
A device which converts infrared irradiance into electrical energy.
Detector Element
A small area etched out of a detector material (called substrate) which does the actual photon sensing and conversion. Most of
the rest of the substrate is inactive and either covered with a plating or not activated. Many detector elements can be put on one
substrate and used for sensing IR radiation.
Dewar
A vacuum walled container for thermally isolating the contents from the outside. Dewars are used for holding and transporting
cryogenic liquids.
Diffuse Reflections
Non-specular reflections of defocused, uniform intensity for a wide range of reflection angles. A perfectly diffuse surface is
defined as a Lambertian surface the intensity is constant no matter what the angle of view.
Digitize
Conversion of analog data into digital data. This can be done manually or automatically. In an infrared system an Analog to
Digital converter is used to convert analog radiation signals into digital information which the CPU can process into equivalent
temperature information.
Distance
Doublet, Lens
A compound lens assembly consisting of two lens elements.
Dwell Time
The length of time that the detector is allowed to observe the same location to create its radiation signal. Normally the longer the
dwell time allowed, the less noisy and more sensitive the detector will be.
Duty Cycle
The ratio of active time to total time allowed in repetitive process.
E
Edge Effects (Optical)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 154
The Professional Thermographers Association
Image or data distortions which can occur at the edges and corners of an image due to optical aberrations building up at the
extreme range of the field of view. These aberrations can include geometric distortion (fish eye), vignetting (hot center/dark
corners), ray distortion (apparent corner defocusing).
Electromagnetic Radiation
The field effects given off by accelerating a charged particle in a magnetic field. Depending on field strength and speed of
acceleration, many types of electromagnetic radiation are created.
Electromagnetic Spectrum
A plot of the range of wavelengths and types of electromagnetic radiation found to exist from subsonic waves to cosmic rays.
Emissivity
The ability of an object to radiate and absorb energy from its surroundings measured as a ratio of the actual object emission to
the blackbody equivalent emission.
Endothermic
Pertaining to heat absorption. A chemical reaction is endothermic if it requires heat to complete the reaction.
Entrance pupil
The size of the aperture located on the front surface of the infrared window containing all of the ray bundles required to scan the
object field of view. This is the map of the system aperture stop scanned on the Infrared window.
Equilibrium
A condition where all of the thermal changes in a system have stabilized. To observe true heat flow characteristics, a gradient
must be developed across an object or interface and allowed to stabilize - this is a thermal equilibrium.
Evacuate
To empty. Used in relation to cleaning Dewars or cylinders prior to refilling them with pure gas. Cylinder evacuation is usually
done by drawing a vacuum with a vacuum pump and mildly heating the cylinder to expedite the evacuation. Pure evacuation
cleaning can take up to two days of continuous vacuum so an alternating cycle of purging and evacuating the container is
recommended.
Exothermic
Pertaining to heat generation. A chemical reaction is exothermic if it liberates heat as the reaction proceeds.
F
F-number /#)
The ratio of focal length to aperture for a lens assembly.
Fahrenheit
A temperature measurement scale which defines the ice point of water as 32 F and the boiling point of water as 212 F. Absolute
zero is -459.7 F.
Far Infrared (LWIR)
Infrared radiation whose wavelength is in the range from 8 to 100 microns.
Field of View (FOV)
The total field measured in angle within which objects can be imaged or measured and displayed by an infrared system.
Filter (optical)
An optical device which modifies the characteristics of radiation which is passed through it. Usually filters either attenuate all
wavelengths of radiation a certain controlled amount or modify the optical passband of the radiation - eliminating selected
wavelengths or bands while allowing others to pass.
FLIR
Forward Looking Infrared
Fluorescence
The spectral emission of long wave radiation such as visible or infrared illumination by some materials when stimulated shorter
wave radiation.
Flux
Rate of energy flow expressed as radiant power. Focus The ability of a lens or system to bring image radiation to a point of
convergence within the confines of the active detector sensing area.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 155
The Professional Thermographers Association
Focal Length
The distance from the convergent point for the radiation (focal point) to its affiliated principal plane.
Focal Plane
The plane created by mapping the points of convergence for rays which pass through a lens assembly from an object which is
moved on a plane perpendicular to the optical axis at great distance from the lens assembly (i.e. collimated rays). For a detector
to work properly, all of its active elements must be placed in the same focal plane for such an object. Otherwise, portions of the
object space will appear defocused.
Frequency
The number of cycles an operation occupies per period of time. The normal unit of measurement for frequency is Hertz.
Front Surface Mirror
A mirror whose reflective surface is on the same side as the incident radiation. Front surface mirrors coated for maximum
reflection are normally used in infrared optics to maximize the signal strength of the reflected radiation.
G
Geometric Concentration
The ability of a lens to concentrate radiation on the focal point; the ratio of aperture size to active detector area.
Graybody
A thermal reference source which maintains an output, not at 100% emission (which would be a blackbody) but at a lesser ratio.
Graybody emissions maintain a constant emission ratio over a wide frequency range. This distinguishes them from spectral
radiators.
Gray Scale
An area of the screen reserved for displaying an increasing ramp of image intensities to be used as a calibration aid. Periodic
intensity levels are labeled with the temperature or radiation value they represent.
H
Half Width
The full width of the passband of a filter measured between 50% transmission points.
Heat
Thermal energy.
Heat Capacity
A measurement of an objects ability to store thermal energy. Heat capacity equals the specific heat of an object multiplied by its
density multiplied by its size (or specific heat times mass).
Heatsink
A device for dissipating heat; it absorbs heat by conduction from heat producing devices and dissipates heat by means of
convection.
Heat Transfer
The flow of thermal energy from one object to another. By means of conduction, convection or radiation)
Hertz (Hz)
A unit for measuring frequency. One hertz is one cycle per second.
High-pass Filter
A filter which is blocked at low wavelength, whose transmission spectrum extends from a low cut on wavelength up in
wavelength to the maximum pass wavelength of the detector.
Hue
A term expressing the "color" aspect of a color display; part of the HLS (Hue, Luminosity, Saturation) color model. Without hue,
there are only shades of gray.
I
Ice Point / Ice point reference
The Temperature at which water freezes. A device which creates the ice point temperature very accurately as a reference for
thermocouple temperature measurement.
Image Uniformity
See shading
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 156
The Professional Thermographers Association
Image Enhancement
Techniques used to modify an image to present an observer with more readily accessible information.
Imager
The part of an infrared system which contains the detector, optics and scanning mechanism. The imager must be held and
pointed at whatever scene is to be studied. The word imager is sometimes used instead of viewer to mean an infrared system
which creates relative radiation images without calibration.
Incident Radiant Energy
Total energy impinging on a surface from the surroundings.
Infrared (IR)
Electromagnetic radiation which occupies the band from 0.7 microns to 100 microns. Infrared radiation is between the visible
spectrum and microwave radiation.
Infrared
The region of the electromagnetic spectrum that covers from 1 to 100 microns. The term comes from the Latin base of beyond
the red, as referring to the fact that beyond the red color of the light spectrum there is still energy but it is invisible to the human
eye with out the aid of a infrared imaging instrument. The discipline of viewing objects based on there thermal properties is
called "Infrared Thermography"
Infrared Camera
A optical/electrical device that transfers thermal radiation into a visible image that is displayed on a CRT screen, Monitor, LCD or
other display. Infrared cameras are primarily used Certified Infrared Thermographers in civilian industrial applications and
Research and Development in what is called Infrared Thermography. Infrared imaging systems come in many capabilities and
specific uses. They may only be able to display the infrared image, or they may be able digitally record the image and perform
temperature measurements and analysis at the same. Infrared image cameras primarily work in the 3-5 or the 7-14 micron
range. They should not be confused with Infrared film that is primarily used in photographic camera. Infrared film works in the .7
to 1 micron range and dose not see the thermal energy that is emitted from objects at close to ambient temperatures. Infrared
film see the reflected energy off of objects that is at temperatures of greater that 1,100 deg. F
Infrared Thermography
The science of viewing and understanding objects based on their ability to give off thermal radiation. The use of specialized
infrared imagers that can allow someone to view the object based on the amount of thermal radiation that the object either gives
off or reflects.
Infrared Thermographer
Someone that is trained in the sciences of viewing, measuring and understanding how objects give off their thermal radiation
when viewed through an infrared imaging system. Most Infrared Thermographer today are Certified by certain infrared training
programs that they have passed a criteria of proficiency in the science of Infrared Thermography. Certification alone dose not
indicate a level of proficiency or quality when performing services, but it show that the thermographer has taken the time to
further his level of knowledge. Just as in any trade, the level of education, years of experience, degree of professionalism and
quality of past work must be taken into consideration.
Infrared Film
Photographic media which can image radiation in the SWIR band from about 0.7 microns to 1.1 microns.
Infrared-window
An optical element usually placed on the front of an infrared system that is transparent to infrared radiation but excludes radiation
of other wavelengths and protects the internal sensor components.
Instantaneous Field of view (IFOV)
The angle in milliradians derived by dividing the active detector element's size by the system's effective focal length. An effective
figure of merit for system resolution can be derived by dividing the field of view by the instantaneous field of view.
Insulation
A material to reduce heat transfer by conduction. A good insulator has a large R-value and a large thermal resistance; a low
thermal conductivity.
Interlace
A technique used in visual displays to produce high resolution images at update rates slower than the eye flicker frequency. With
interlace, a fraction of the total frame lines (called a field) is displayed at multiples of the frame rate. These lines are spread out
across the entire display area and with each field update, a different set of lines are displayed. The number of fields required to
reconstruct the entire frame is the interlace factor. The lines are usually spaced apart by the interlace factor. Normal television is
interlaced by a factor of two fields per frame.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 157
The Professional Thermographers Association
Irradiance
The intensity of radiation impinging on a surface, the rate of impact of radiation.
Isoradian
Areas or lines of constant thermal irradiance.
Isotherm
Areas or lines of constant temperature.
Isothermal Windowing
An image processing feature which allows the user, to enhance a range of isotherm or isoradian levels. This feature allows the
user to highlight areas of similar temperature and easily find the hottest and coldest object in an area such as an electrical
distribution panel.
J
Joule
A measurement of thermal energy. one joule is one newton meter, 10 7 ergs, or approx. .737 foot-pounds.
K
Kelvin
The most commonly used absolute temperature scale. The scale is based on the Celsius scale degree increment with 0 K equal
to absolute zero cold.
Kinetic Energy
The energy of motion.
L
Lag (thermal time)
The delay between the construction of a thermal gradient and the achievement of equilibrium.
Lens
An optical component constructed of transparent substance with one or two curved surfaces of different curvature which has the
ability to change the direction of beam travel. Infrared lenses are used for focusing the detector at a distance of interest and for
modifying the size and distance of the focused field of interest.
Level slicing
An image processing function like isotherm windowing. The visual display is modified by changing the image intensity or color of
a region of levels which the user selects. Level slicing is usually implemented by blocking out or whitening out all the levels
above or below the user selected level.
Light
The region of the electromagnetic spectrum which is visible to the human eye. Usually considered as the region from 0.39 (violet)
to 0.77 (red) microns.
Limiting Resolution
The highest spatial frequency which a system can resolve, regardless of target temperature. Ultimately the spatial performance
end of the MRT curve. Usually expressed in milliradians. Line scan An instrument scanning and display mode which scans in
one direction only but displays those lines side-by-side in the perpendicular direction to create a two-dimensional thermal map of
a scene over time or panning motion.
Linear Array
A collection of active detector elements set in a straight line on a plane usually spaced equal distances apart from each other
with a single element in the perpendicular direction.
Low-pass Filter
A filter blocked at higher wavelengths, with a cutoff wavelength in the passband of the detector that passes all wavelengths
shorter than the cutoff wavelength.
Lumen
An SI unit for measuring radiant flux. One lumen is produced per solid angle by a point source with one candela intensity.
Luminance
The portion of a color display which is related to display intensity; how bright a color is perceived by the eye.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 158
The Professional Thermographers Association
M
micron (micrometer))
A measurement of length in the metric system appropriate for measuring infrared radiation wavelengths. 1,000,000 microns
equals one meter.
Mid Infrared (MWIR)
The middle infrared spectrum, usually from 2.4 to 7.0 microns. Milliradians A measure of small angles. Two thousand-pi
milliradians can be measured in a complete circle. There are 17.4 mares per degree of angle.
Monocular
A viewing device made to produce an image for one eye only.
Monotonic
Having the characteristic of steadily increasing. Functions are monotonic if they are continuous, single valued, and steadily
increasing.
Mosaic
See FPA - focal plane array.
N
Narcissus Effect
An optical phenomenon of scanning systems which describes how a detector can look back at itself or view a mixture of active
scene and itself for certain angles of scan. The narcissus effect creates blurry cold areas on screen in an infrared system.
Narrow Bandpass Filter
A multi-layer filter usually based on interference effects whose cut-on and cutoff frequencies are very close to each other. In
infrared, the half width of a narrow bandpass filter will be 0.5 to 1.0 micron.
Near Infrared (SWIR)
The shortest wavelength infrared radiation band - 0.7 to 1.4 microns.
Neutral Density Filter
A filter which attenuates radiation uniformly over a wide range of wavelengths; used in infrared systems as a temperature range
extension filter.
Noise
Unwanted signal interference - usually separated into various forms of signal cross talk and random noise generated by means
internal to the sensor.
O
Object plane
The plane created by tracing ray paths backwards from the active detector elements through the focusing lens assembly,
through the scanning mechanism to project the active field of focused image area in space. The object plane is an optical
description of what the system can focus. Ocular The eyepiece of an optical system.
Ohmmeter
An instrument to measure electrical resistance.
Opaque
The characteristic of not passing any incident radiation (Transmissivity = 0). An optical filter is said to be blocked in a waveband if
it has less than 5% transmission over those wavelengths.
Operating Temperature Range
The range of ambient temperatures over which a system will function accurately. This range can be defined by either a
calibration limit; what range of ambient temperature offsets are allowed in the data calculations or it can be due to a functional
limit in the system; the ability to keep the detector cold or it can be set by a catastrophic limit; batteries may not hold their charge
below certain temperatures and integrated circuits will not function above certain temperatures.
Overlap (optical)
Redundant scanning of the image field which results in multiple sets of data taken from the same image points. If done in the
direction of scan this is usually considered oversampling. If done by laying scan lines adjacent to each other closer than the
optical spot dimension, it is considered scan overlap.
Oversampling
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 159
The Professional Thermographers Association
To take multiple sets of data redundantly. Usually indicates digitization of a signal at a rate faster than the maximum information
frequency of the data. An oversampling rate of at least two is indicated by information theory to avoid aliasing of repetitive
information (moire).
Overscanning
Expanding the beam deflection on a CRT so that the active viewing area is larger than the available CRT faceplate. This cuts off
the edges of the active raster. This technique is used on most television monitors.
P
Panning
A means used to observe an object which is larger than the field of view of the system. The operator moves the system back and
forth across the target to discover thermal areas of interest.
Passive Infrared System
The normal form of infrared system in use commercially. A passive system does not generate a thermal source to illuminate the
scene, it is only acted upon. A passive system monitors the flux of photons already being generated by the scene within its field
of view.
Peltier Effect
The ability for two dissimilar metals and metal/semiconductors to create a thermal difference when current flows across the
dissimilar junction.
Photoconductor
A radiation sensitive resistor whose resistance decreases as it is exposed to radiant energy.
Photon
A single quantum of electromagnetic energy having momentum hf/c and energy hf. (h is Planck's constant, f is the frequency of
the wave and c is the speed of light).
Photovoltaic Detector
A detector which responds to radiant flux be generating current.
Pixel (Picture element)
The smallest location size on a display or in memory. The incremental location of picture information in either horizontal or
vertical direction (also called a Pel).
Principal Plane
A mathematical construct useful in analyzing complex optical assemblies. The principal planes are constructed at the effective
focal length distance away from the lens assembly focus and serve to represent the mathematical position of the lens assembly
assuming it could be constructed as a single thin lens.
Probeye
Registered trademark of Hughes Aircraft Co. assigned to the Hughes line of commercial infrared system products.
Pupil
The map or footprint of the system aperture stop system onto a surface. The pupil represents the collection of all ray paths used
by the system to form an image. Obstruction of the pupil will lead to image vignetting.
Pyrometer
An optical instrument for remote sensing and measurement of spot temperatures or radiation levels
Q
Qualitative Analysis
An analysis of objects or processes which is concerned with deriving structural, material, or relative information. This type of
analysis can be done with imagers, line scanners, and viewers; temperature or radiometric data output is not required.
Quantitative Analysis
An analysis of objects or processes which is concerned with measuring temperatures or radiant energy levels by assigning
numerical values to the characteristics of the displayed scene.
Quantum Efficiency
The ratio of actual detectivity to theoretical detectivity for a given detector material.
Quenching
A technique used for stopping one process with another. In metal or glass formation, crystal formation during cooling can be
stopped by rapid quenching with air or water.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 160
The Professional Thermographers Association
R
Radian
A measurement of angle. There are 2 r radians in a full circle of 360 angular degrees. Radiance The total intensity of thermal
energy (radiant flux) which can be observed from a surface. It is scaled by unit of solid angle of view and by unit of area from the
surface. The flux includes all forms of radiation emission, reflection and transmission.
Radiant Energy
See radiance
Radiation
Heat transfer of energy in the form of electromagnetic waves. Forms of radiation include cosmic rays, gamma rays, x-rays,
ultraviolet radiation, infrared, visible light, radio, audio, and sub sonic. Radiometer An instrument which measures radiation
levels. Such an instrument can be calibrated in power or temperature.
Rankin
A temperature scale based on O*R equal to absolute zero temperature. The increment of one degree Rankin is equal to one
degree Fahrenheit. 459.70 Rankin is equal to O*F.
Rayleigh Criterion
A measure of maximum lens resolution. The smallest possible blur circle a lens can create will depend on the aperture of the
lens and the wavelength of the radiation. for circular optics: R = 1.22 (lambda)/a
Recertification
Retesting of equipment to return it to an original standard or calibration. In infrared, it is spoken of in two ways. If the infrared
system is being used to produce calibrated data, it must be recalibrated to a secondary standard regularly. When high pressure
containers are used to hold the cryogen, they must be recertified to hold the required pressure, either every three or every five
years. See the notice on the cylinder.
Reflectance (Reflectivity)
The amount of total radiance which can be attributed to reflected energy. Usually expressed as a percentage of total energy.
Repeatability
A measure of accuracy and stability for an instrument. The capability of an instrument to duplicate a previous answer to a
previous experiment within a limited margin of error.
Remote Sensing
To obtain a reading or measurement from a distance, without physical contact between the meter and the object to be measured.
Resolution
A measure of the capability of a system to resolve small objects against the viewing field. Measured as a ratio of angles.
Responsivity
A measure of detector performance, measures the value of the detector output change for a given applied thermal power
change.
Rotor
Same as scan wheel. one method of creating a field scanning mechanism using a reflective polygon, spinning on its axis.
S
Saturation
The ratio of pure color to washed out white in the display of a color. 100% saturation is pure color. 0% saturation is pure white, at
whatever intensity is encoded in the color.
Scale Factor, Scaling
A factor used to adjust the gain of an object parameter. Normally used in Temperature range adjustment, the gain of the
amplifiers is scaled until the desired temperature range is displayed in full contrast from black to white. The units associated with
a reading is a temperature scale (F, C, K, R).
Scale Sensitivity
The minimum adjustment allowed in the system per intensity level. A system is normally configured so that the operator can set a
scale sensitivity greater than the system sensitivity. If the NET of a system were 0.5*C, the scale sensitivity might be O.IOC or
.050C to allow enough signal for offline image improvement features such as averaging to reduce the noise level.
Scan conversion
The reformatting of data, usually image data from one standard to another. In the case of infrared, the sensor output is not
compatible in either time nor number of lines nor number of resolution elements to television. The sensor output must be scan
converted into a television format in the image processor.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 161
The Professional Thermographers Association
Scan Efficiency
The ratio of active scan time to total scan time. Can be expressed as a function of the ratio of two angles also. Normal scan
efficiencies with rotating polygons can be from 25% to 60% (with reimaging optics). Normal scan efficiencies with galvanometer
mirrors can be as high as 90%.
Scan Line
One line of infrared data, whether from left to right or top to bottom, as output to the processor for scan conversion. The number
of scan lines read by the scanner is the maximum resolution of the system in one direction. This resolution will be limited further
by any overlap factor used.
Scan Wheel
A mechanical scene scanning method which involves a number of flat facets incorporated into an aluminum wheel., rotating
around an axle through the center of the ring of mirrors.
Seebeck Effect
When a junction of dissimilar metals changes temperature, a current will flow across the junction - creating a measurable emf.@
This is the principal of operation of a thermocouple.
Selective Radiator
A source which radiates thermal energy is specific bandwidths and emits little or no radiation in others. An incandescent light is a
gray body radiator while a quartz iodine light is a selective radiator. Hot pipes are gray body radiators while flames are selective
(spectral) radiators.
Sensitivity
A measure of the minimum amplitude of input signal change to which an instrument will respond. This is a measurement of
thermal resolution.
Sensor
The component which converts radiation into electrical signals. Can be used to refer to the detector specifically, the imager, or
the entire system.
Shading
A measure of the non-uniformity of signal in an infrared system. Shading is measured by exposing the system to a wide area,
constant temperature target and measuring the variations around a mean value. Shading may be identified by source - for
example optical shading will be characterized by strong responses in the center and weak responses in the corners (vignetting).
Channel to channel variations in response can also be a source of shading (Channel balance).
Specific Heat
A property of materials. The specific heat of a material indicates how much thermal energy (in joules) is required to increase a
mass (in grams) of material a small temperature difference (degree C). Specific heat is one of the principal factors in determining
heat capacity, conduction rates in a material ' and the thermal time constant of an object subject to heating.
Spectral
Pertaining to the electromagnetic spectrum, depending on wavelength or frequency, varying based on wavelength.
Spectral absorption
The ability a filter or gas has of transmitting radiation of some wavelength while absorbing materials of other wavelength. The
atmosphere is a spectral absorber for relatively long infrared path lengths.
Spectral emission
A source which radiates thermal energy is specific bandwidths and emits little or no radiation in others. An incandescent light is a
gray body radiator while a quartz iodine light is a selective radiator. Hot pipes are gray body radiators while flames are selective
(spectral) radiators.
Specular
Indicating reflections or a shiny surface.
Spot size
The minimum size of object which can be resolved at a given distance by an optical device. Stability A measure of system
accuracy and reliability. Indicates how little a system reading moves from an original value.
Standards, Primary
The ultimate physical standard used as a basis for measurements. In the United States, all primary standards are maintained by
the National Institute of Standards and Technology. The primary standards are used to calibration secondary standards.
Standards, Secondary
Secondary standards are physical standards and references, which have been calibrated directly off of the NIST primary
standard. These standards are normally maintained by the more sophisticated testing laboratories and used to calibration
reference standards.
Standards, Reference / Standards, Working
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 162
The Professional Thermographers Association
Tertiary standards are the standards that are actually used in plant facilities for the calibration of product. They are periodically
calibrated against the secondary standards. The normal working standards used in the infrared field consist of Black body
sources, thermocouples and thermocouple meters, RTDs , and thermometers.
Sterling Refrigerator
A component used for cooling detectors to cryogenic temperatures. A sterling cycle refrigerator operates on compression and
expansion of gasses like a home refrigerator. However, the refrigerator uses the Sterling cycle and Helium gas instead of freon.
T
Target
An object in the object plane which the system can focus on and analyze.
Television Line (Video line)
A single horizontal line of display data on a television display. NOT the same as scan line which is the data input: a television line
is a single line of output data.
Temperature
An expression of thermal energy density. How hot or cold an object is.
Temperature Range
The maximum to minimum temperature display capability of a system. It should be specified whether the range under
consideration is the range displayed, the total display range capability of the system, or the operating temperature range of the
system.
Thermal Conductivity
A property of materials which measures the ability of a material to conduct heat. It can be expressed as power per degree length
(watts/meter-C). Metals have a high thermal conductivity (conduct well) while air fibers and plastics have much poorer
conductivities.
Thermal Expansion
The change of size which materials undergo as their temperature changes. In tight mechanical tolerance assemblies, the thermal
expansion coefficients must be matched to maintain tolerances over an significant operating temperature range.
Thermal Gradient (or Thermal Profile)
A graph of temperature changes over a distance. A thermal gradient is usually expressed and displayed as a straight line sometimes only vertical or horizontal.
Thermal Radiation
Electromagnetic energy whose natural wavelength fall between .7 and 100 microns.
Thermal Viewer / Thermal Data Viewer
A class of remote temperature sensing systems. This equipment class offers an image of relative radiation levels and a means
for obtaining temperature information from the screen. A common alternative is to have a bulls eye target indicator and an
alphanumeric overlay display of the temperature and the operator selected emissivity.
Thermal Video System
A class of remote temperature sensing systems. This class of equipment is characterized by a fully calibrated display screen,
video output, provision for the standard optical and data handling accessories.
Thermistor
A device which measures temperature. The sensor for the thermistor is a semi-conducting resister whose resistance changes
significantly with temperature.
Thermocouple
A device which can measure temperatures, usually by contacting the device to be measured. The device is made from a junction
of dissimilar metals, as the junction changes temperature, a voltage is created which is read by a previously calibrated meter.
Thermoelectric Cooling
A solid-state device which converts current into a temperature difference between two junctions. It is possible to put
thermoelectric junctions in series or parallel to increase either the overall temperature drop or their power.
Thermogram
A two dimensional hard copy record of the apparent scene temperatures displayed on an IR system (usually a photograph of the
display).
Thermography
The study of remote temperature measurement
Thermometer
A meter for measuring temperature.
Thermopile
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 163
The Professional Thermographers Association
A number of thermocouples whose junctions are assembled in series to magnify the Seebeck voltage and increase the sensitivity
of the reading. Thermopiles have been used in remote sensing pyrometers.
Transmittance (transmissivity)
A measurement of the ability of a material to pass radiation from one side to the other without absorbing or reflecting it.
Transmittance is the ratio of transmitted radiant energy to total irradiance.
Triple Point (of water)
The temperature at which all three phases of the material can exist at equilibrium. 0.01'C for water.
Triplet
A compound lens assembly comprised of three lenses.
U
Undershoot
The amount that a cooling cycle in a thermal feedback control system cools beyond the set point.
V
Valence Band
The energy level which contains molecularly bound electrons which can not move within the crystal lattice. For there to be
conduction in a semi-conductor, valence electrons much be stimulated to overcome the energy gap and enter the conductance
band.
Velocity of Light
The speed at which light travels in a vacuum. 186,280 miles per second.
Velocity of Sound
The speed at which sound travels in air at STP. 12.3 miles per second (nom.).
Vertical Blanking
The time interval during a video signal allowed for the vertical raster to retrace from the bottom of the CRT to the top. During this
interval the CRT is blanked from writing on the phosphor.
Video
A method of storing, generating and reconstructing pictures on monitors based on serial electrical signals and raster scanning of
electroptical cameras and displays. Used in television.
View Factor
A factor which adjusts the radiant energy transfer values between two objects based on their relative surface geometry.
Viewfinder
A small display attached to a camera (or IR sensor) for aiding the operator in adjusting the location and display characteristics of
the equipment. A viewfinder reproduces the finally system display at a reduced size and resolution, usually in black and white to
help with adjustments.
(Infrared) Viewer
A class of remote infrared sensing equipment. This class of-equipment creates an image of relative radiation levels but does not
allow any direct readout of intensity values. This kind of equipment is quite useful for police, fire, and security use. Viewers have
been used effectively for many years in the qualitative analysis of maintenance inspections.
Vignetting
The loss of radiation intensity due to the blocking of a portion of the beam bundle between the focused point on the object plane
and the focused point on the image plane.
Void (thermal)
An area in a surface which displays significantly different thermal impedance from adjacent areas. This can be due to a different
object internal -structure - such as an insulation void in the wall of a building.
W
Watt
A measure of power equal to one joule expended for one second.
Wavelength
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 164
The Professional Thermographers Association
The length of distance between cycles on a repetitive event.
Win's Displacement Algorithm
A statement relating the temperature of an object to the wavelength of maximum radiative output. (They are inversely
proportional.)
Wheatstone Bridge
An electronic circuit for measuring the resistance of an object. This circuit is of specific value in measuring the resistance change
of a thermistor as it tends to linearize the thermistor response.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 165
The Professional Thermographers Association
Appendix: IR The History of Heat / Temperature and Infrared
imaging
The development of non-contact measurement and the physics related to it has a long history. We have
included some of the principle points of this development for your information.
50?
Lucius Annaeus Seneca: Discussed a number of topics that pertained to physics and optic in his
seven books Naturales Quaestiones.
1025? Alhazen: The first historically recorded person to discuss the magnifying effect of a lens.
1606 Galileo Galilei: Invents a thermoscope which was the beginnings of temperature
measurement.
1609 Galileo Galilei: Used an 8 power refracting telescope and paved the way for the use of optical
systems in scientific experiments.
1621 Willebord Snell: Discovers the law of refraction known as Snells law and is used in the manufacture
of lenses.
1622
Galileo Galilei: In his tract the Assayer he discusses atomism.
1666
Sir Isaac Newton: Investigated the refraction of light through a prism.
1668
Sir Isaac Newton: Built the first reflecting telescope.
1669
Sir Isaac Newton: proposed the theories of the fundamental properties of light.
1669
Erasmus Bartholinus: Formulated the principle of the polarization of light in Denmark.
1676
Ole Christensen Roemer: Calculated the velocity of light.
1690
Christian Huygens: Advanced the theory of the refraction of light based on the wave theory of light.
1708 Ole Christensen Roemer: Invented a thermometer with 50 deg as the boiling point of waterh and
7.5 as the melting point of snow. Passed some of his ideas on to Daniel Fahrenheit.
1714
Daniel Fahrenheit: Invented the thermometer scale that bears his name.
1729
James Bradely: Discovered aberrations in light.
1744 Anders Celsius: Invented the thermometer scale that was originally called the Centigrade
scale bnut now bears the Celsius name.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 166
The Professional Thermographers Association
1753 Thomas Melvill: Discovered that various chemicals emitt distinct colors when burned in an alcohol
flame.
1758
John Dollard: Invented the achromatic lens.
1783 William Wollaston: Investegated solar spectral emission by the use of a slit. Also made many
valuable observations on the refraction of light.
1800
Sir William Herschel: discovered the infrared spectrum.
1801
J. Ritter: Discovered the ulttraviolet spectrum
1801
Thomas Young: Formulated the theory of color perception.
1810
Etienne Louis Malus: Presented the teory of double refraction.
1819
Augustin Fresnel: Explained difraction patterns by assuming light to be wave motion.
1822
Thomas Seebeck: discovered the thermoelectric effect.
1828
William Nicol: Invented a prism to produce polarized light.
1830
L Nobile: Invented the thermocouple.
1833 Macedonio Melloni: Improved the thermocouple (thermopile) Made several important discoveries
regarding radiation among which was the fact that rock salt was transparent in the infrared bandwidths.
1839
A. Becquerel: First to discover the photovoltaic effect.
1840
Sir John Herschell: produced the first thermogram using carbon soot and alcohol.
1845
Michael Faraday: Observed a relationship between magnetism and light
1857
Jean Bernard Leon Foucault: Demonstrated that the velocity of light varies in different medias.
1858 Gustav Kirchoff: Kirchoff’s Law - a bodies ability to absorb energy is directly proportional to
its ability to emit energy.
1858
Gustav Kirchoff and Robert Eberhardvon Bunsen: Developed the first practical spectroscope.
1868 A. Becquerel: Demonstrated the phosphorescent and photographic effects of near infrared
radiation.
1872
John Tyndall: Measured the radiant power transferred by one heated body to another.
1873 James Clerk Maxwell: Presented the classical theory of electromagnetic radiation. They were
based on Farradays experiments.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 167
The Professional Thermographers Association
1873
Willoughby Smith: Discovered optical photoconductivity in selenium.
1879 Joseph Stefan: Concluded that the rate at which heat is transferred from one body to
another is proportional to the difference of the fourth power of their absolute temperatures.
1881
Samuel Pierpont Langley: Developed the bolometer.
1884: Ludwig Boltzmann: Showed theoretically that Stefans law of radiation was correct. The Stefan
Boltzmann constant was derived.
1886
Albert Michelson: Measured the velocity of light and developed the interferometer
1887 Heinrich Hertz: Demonstrated that there is no difference between thermally and electrically
produced electromagnetic waves.
1895
Julius Ester and Hans Geitel: Invented the photoelectric cell.
1900 Max Planck: Established the quantum theory in which radiant energy consists of discrete
quantities of energy called photons.
1904
J.C. Bose: Discovered an infrared photovoltaic effect in lead sulfide.
1905 Albert Einstein: Explained the photoelectric effect. Made great contributions validating the quantum
theory.
1917 T.W. Case: Discovered that thallous sulfide was photosensitive in the infrared. Developed the
thalofide cell in 1920.
1919 S.O. Hoffman: Described one of the earliest passive infrared systems that could detect a man at
600 ft.
1919
A.O. Rankine: described voice transmission by light.
1929
M. Czerny: Manufactured the evapograph which was the first working thermal imaging device.
1929
L. Kola: described the first infrared image tube
1930
H. Babcock: Published the first all-infrared photograph
1934
I. Nikion and M. Noskov: Discovered the photoelectromagnetic effect.
1940 G. Hass: Discovered that silicon monoxide could be used as a protective layer on front surfaced
mirrors.
1946
J Becker and W Brattain: Developed the thermister bolometer.
1947
M.J. Golay: developed an improved pneumatic detector (golay cell)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 168
The Professional Thermographers Association
1960 T. Maiman: Constructed the first operating laser.
Appendix: IR Applications
As a Level III Thermographer, you are expected to understand and be capable of directing personnel in the
performance of most IR/T application methods. The knowledge gained in the areas of Thermodynamics as
well as Infrared theory should give you the necessary tools to competently administer programs that are
diverse in nature. This section lists the general areas that you are expected to have knowledge of.
Exothermic or Endothermic Investigations
Power distribution systems
Exposed electrical switch gear
Enclosed electrical switch gear
Exposed electrical busses
Enclosed electrical busses
Transformers
Electric rotating equipment
Overhead power lines
Coils
Capacitors
Circuit breakers
Indoor wiring
Motor control center starters
Lighter arrestors
Chemical process
Foam-in-place insulation
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 169
The Professional Thermographers Association
Fire-fighting
Building investigations
Outside ground base investigations
Outside airborne investigations
Moisture in airframes
Underground investigations
Airborne coal mine fires
Utility locating
Utility pipe leak detection
Void detection
Locating and mapping utilities concealed in structures
Mammal location and monitoring
Ground investigations
Airborne investigations
Sorting mammals according to stress levels
Fracture dynamics
Process heating or cooling
Rate
Uniformity
Heat tracing or channelized cooling
Radiant heating
Electronic components
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 170
The Professional Thermographers Association
Assembled circuit boards
Bare printed circuit boards
Semiconductor microcircuits
Welding
Welding technique parameter
Material parameters
Mapping of energy fields
Electromagnetic fields
Electromagnetic heating processes
Radiant heat flux distribution
Gaseous plums
Monitoring
Mapping
Ground frostline mapping
Friction Investigations
Bearings
Seals
Drive belts
Drive couplings
Exposed gears
Gearboxes
Machining processes
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 171
The Professional Thermographers Association
Aerodynamic heating
Fluid Flow Investigations
Fluid piping
Valves
Heat exchangers
Fin fans
Cooling ponds
Cooling towers
Distillation towers
Packed
Trays
HVAC systems
Lake and ocean current mapping
Mapping civil and industrial outflows into waterways
Locating leaks in pressure systems
Filters
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 172
The Professional Thermographers Association
Thermal resistance (steady state heat flow) investigations
Thermal safety audits
Low temperature insulating systems
Building envelopes
Industrial insulation systems
Refractory systems
semi-transparent walls
Furnace interiors
Disbonds in lined process equipment
Thermal Capacitance Investigations
Tank levels
Rigid injection molding
Thermal laminating processes
Building envelopes
Roof level investigations
Airborne investigations
Underground voids
Bridge deck laminations
Stem traps
Paper manufacturing moisture
profiles
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 173
The Professional Thermographers Association
Subsurface discontinuity detection in materials
Coating disbonds
Structural materials
Subsurface discontinuity detection
Thickness variations
Disbonding
Infrared NDT
Passive
Level 1
Simple technique
The imaging of an object or material with no aggressive interaction on the part of the technician.
Active
Level 2
From simple to extremely complicated.
Deliberate action on the part of the technician to change the condition of the object or material in
question, usually done by heating or cooling.
Active Planning Considerations
1. Object/anomaly characteristics
2. Methodology
3. Active source
Object/Anomaly Characteristics
Composition: single layer, multi-layer, multi-material, coated
Qualitative
Absorbtivity
Conductivity
Resistivity
Quantitative
Absorbtivity
Conductivity
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 174
The Professional Thermographers Association
Resistivity
Diffusivity
Effusivity
Specific Heat
Density
Methodology
Heat or Cool object
Conductive or Resistive Method
Time: duration, control
Computer processing of data
Source operation
o Pulse
o Modulated
o Continuous Step
Source Operation
T
e
m
p
e
r
a
t
u
r
e
T
e
m
p
e
r
a
t
u
r
e
Time
Pulse
T
e
m
p
e
r
a
t
u
r
e
Time
Modulated
Time
Continuous Step
Active Sources
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 175
The Professional Thermographers Association
Source
Laser
Flashlamp
Quartz Lamp
Hot Air Gun
Microwave
Resistive Heating
Induction Heating
Types
Broad Uniform
Localized Spot
Line
Line Array
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 176
The Professional Thermographers Association
Appendix: Building Heat Loss
Heating Systems
An energy efficient home by definition has very little heat loss because of high insulation levels and airtight
construction. This leads to two problems: finding a properly-sized heat source and providing adequate
ventilation to maintain indoor air quality.
Except for extremely cold periods, a properly designed and constructed energy efficient home can
sometimes gain almost enough daily heat from 'waste' sources such as the heat given off by lights, people
and appliances. During sunny, cold days solar energy gains also contribute to reducing the heating load.
These heat sources are often called 'internal gains'.
Modern control systems such as programmable thermostats can further help to reduce heating energy
consumption.
Sizing the Heating System
Heating equipment in a home must be capable of maintaining an interior temperature of 68 to 72° F (20 to
22°) during the heating season. Heating equipment is generally oversized for most homes, but is even
worse if the home is an energy efficient home. This leads to frequent on/off operation reducing both
efficiency of fuel use and service life of heating equipment.
A heat loss calculation is required to determine the heating system required. It should have a capacity of no
more than 10% in excess of the calculated requirement. Installing the smallest capacity heating equipment
to meet the loads will save both energy and money. Calculation of total home heat loss is generally done by
heating contractors. Contractors inexperienced in understanding low energy house design and heat loss,
however, may still result in drastic over sizing. A simple heat loss calculation method is provided in this
section.
Isolating The Heating System
If a fuel burning furnace, boiler and/or hot
water heater is required, building an airtight
enclosure (mechanical room) around the
appliances can help control chimney heat
loss. Separate combustion and fresh air
supplies feed into this room. No previously
heated air is used by the fuel burning
appliances and cold outside air is
prevented from entering other areas of the
home. This isolated room must be
insulated and sealed from the rest of the
home. Water pipes and heat supply ducts
should also be insulated.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 177
The Professional Thermographers Association
Calculating Heat Loss
Calculating the heat loss from a home is
quite simple. The heating requirement will
be highest when the outside temperature is
lowest and there is no solar gain. A cold
winter night is when the heating load will
be greatest. Heat flows out through all the
building surfaces including walls, ceilings,
floors, windows and doors.
Heat loss through each surface can be calculated using the following equation:
Heat is also lost through infiltration and exfiltration - air leakage. This heat loss can be calculated
using:
Note: change the constant 0.36 to 0.018 Imperial units.
There are also a number of sources of heat gain in a typical house. Not only do the occupants give off heat,
appliances and lights contribute significantly to home heating. Each person can provide about 75 watts of
heating energy while 200 or 300 watts are available from appliances (like freezers, ranges, refrigerators,
etc). The average home therefore provides 500 or more watts daily of the total energy required for space
heating.
An example heat loss calculation is shown using Plan 13 from the REED Energy Efficient House Plans
section. Plan 13 is a 1,920 square foot (178 sq. metres) two level, rectangular bungalow. If this house was
to be built in the Red Deer (Alberta, Canada) area, the outside heating design day temperature is - 26° F (33° C). A common inside temperature is 68° F (20° C) - the difference between them is 94° F (53° C).
Design day temperatures and heating degree days information for your locale is usually available from your
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 178
The Professional Thermographers Association
local weather office. A short list is provided as an example of January design temperatures and degree
days for various Canadian cities.
The first set of equations is used to calculate the heat loss from each of the seven surfaces (substitute the
areas, R-values and the temperature difference for each surface from your plan). The air leakage heat loss
is next calculated using the second equation. The building air volume is 16,775 cubic feet (475 cubic
metres) and an air change rate of one-third (1/3 of the house air is replaced every hour with fresh air) can
be used in the equation (a typical rate for a well-built, energy efficient home). Substitute the house volume
from your plan into the equation. Using the same temperature difference, the total air leakage heat loss is
calculated and added to the surface heat loss figures. Subtracting the heat gain average of 500 W results in
a total space heat requirement of 8200 W for this example (8.2 kW or about 28,000 btu/hr).
There are a wide variety of computer software programs available which can be used to more accurately
calculate building heat loss. These programs require a detailed breakdown of each building component and
complete area weather data. Most of the programs available require a considerable learning curve and are
often not practical unless you do a lot of heat loss calculations, are a house designer or are designing a
complex solar building.
The efficiency of the heat source must be taken into account when selecting it. In the example, an 8.2 kW
heat source would be needed (28,000 btu/hr). If one chooses a 100% efficient electric heating source, the
exact figure calculated above can be used to size equipment. Gas furnaces range from 70% to 80%
efficient (measured seasonally - over an entire year of operation). Divide the heat load (8.2kW) by the
system efficiency (0.70) to obtain the 'bonnet' size of 11.7kW (40,000 btu/hr) necessary to provide 8.2kW.
Gas-fired furnace and boiler units with efficiencies of 90% to 95% are also available but are usually
produced in large output sizes and are more expensive.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 179
The Professional Thermographers Association
Heat Load Calculation Worksheet
Building Surface
Plan 13 Example
Your Plan
Ceiling Area
84 m²
............
Ceiling RSI
7
............
Wall Area (less door and window areas)
67
............
Wall RSI
3.5
............
Above Grade Basement Wall Area
25
............
Above Grade Basement Wall RSI
3.5
............
Below Grade Basement Wall Area
76
............
Below Grade Basement Wall RSI
3.5
............
Floor Area
80
............
Floor RSI
1.8
............
Window Area
12
............
Window RSI
0.5
............
Door Area
2
............
Door RSI
1.8
............
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 180
The Professional Thermographers Association
Plan 13 Example Calculations
Your Plan Calculations
84 x 53
Ceiling Heat Loss
x
= 636 W
= .......... W
7
67 x 53
Wall Heat Loss
x
= 1015 W
= .......... W
3.5
Above Grade Basement
Wall Heat Loss
Below Grade Basement
Wall Heat Loss
Floor Heat Loss
25 x 53
x
= 379 W
= .......... W
3.5
76 x 25
x
= 543 W
= .......... W
3.5
80 x 15
x
= 667 W
= .......... W
1.8
Window Heat Loss
12 x 53
x
= 1272 W
= .......... W
0.5
2 x 53
Door Heat Loss
Air Leakage Loss
1.8
(0.36)437 x 0.2 x 53
x
= 59 W
= 1667 W
= .......... W
(0.36) .... x .... x
Total Heat Loss
= 6238 W
Average Heat Gain
-500 W
House Total
= 5738 W (5.7 kW)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
= .......... W
-500W
Page: 181
The Professional Thermographers Association
Compact Building Form Cuts Heat Loss
This article appeared in Energy Source Builder #36, December 1994.
© Copyright 1994 Iris Communications, Inc.
Many architects, designers and builders have gotten in the habit of dealing with heat loss once the design
is complete. Better windows here, more insulation there, and presto, you have cut the house's heat loss (or
heat gain). That approach works, but there's another side to heat transfer. It's surface area.
By trimming the surface area of
the building's thermal envelope,
you can dramatically reduce heat
transfer. Reducing surface area
often cuts construction cost, too.
Surface area is closely related to
the geometry or form of the
building. The shape and
complexity of a house design
affects the surface area and the
overall energy used for heating
and cooling.
These three designs illustrate this
idea. Each design contains 2,000
sq. ft. of conditioned floor area.
The insulating value of all building
components is the same. Windows
are evenly distributed among the
four elevations.
The key difference between the designs is the surface area of the building's thermal envelope. The
compact two-story design has 4,210 sq. ft. of
surface area--30 percent less than the complex,
one-story design and 26 percent less than the
ranch. More surface area means more heat loss.
The complex design requires up to 24 percent
more energy for space heating.
The data for these comparisons was generated
with the Wattsun 5.4 computer program
published by the Washington State Energy
Office. The analysis uses weather data for
Eugene, Oregon (4500 heating degree days).
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 182
The Professional Thermographers Association
Component Tradeoffs
Ceilings generally have more insulation than walls, and the same is often true of floors. In these examples,
ceilings are R-38, floors are R-30 and walls are R-19. A square foot of wall allows twice as much heat loss
as a square foot of ceiling. Trading one square foot of highly insulated ceiling for one square foot of wall
with less insulation would be a mistake. However, the two-story design adds about 500 sq. ft. more wall
area, while it subtracts 2,000 sq. ft. in the floor and ceiling. That's a net reduction of 1,500 sq. ft. The large
difference in surface area more than makes up for the difference in R-value.
As these examples show, building form has a major impact on heat loss. That doesn't mean every house
should be a two-story rectangle. But it does suggest that simplicity can save energy (as well as construction
costs). One good tool for making design decisions is a computer program that estimates energy use.
Several good energy analysis programs are available.
These examples have been kept simple to illustrate a point. Other factors, such as solar heat gain, also
influence energy use. Extending the east-west axis could capture additional solar heat and more than make
up for a the extra surface area.
Design Interest without Complexity
Designs do not need to be highly complex to be interesting or marketable. You could consult an architect or
designer for ideas on getting a good design in a simple shape. In some cases, architectural interest can be
applied to the exterior by adding covered porches, bay windows, roof lines and decks.
There's almost always a way to pack more insulating value into a house once it's been designed. At that
point, cost can become a major obstacle. On the other hand, decisions that affect building form must be
made early in the design process. Fortunately, simplifying building form doesn't have to cost anything, it
often saves money.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 183
The Professional Thermographers Association
Appendix: Home inspections and the Law
You, your IR camera and the law.
Thanks to the Agema Thermovision 210 Thermal Imaging Device, the men
in black helicopters were able to see every joule of heat you and your
valentine produced while getting busy last week.
Supreme court dispatches
Spot Pot Hot Shot
By Dahlia Lithwick
Posted Wednesday, Feb. 21, 2001, at 3:00 AM ET
The Oregon National Guard used that device—and an astronomical utility bill—to deduce that Danny Lee
Kyllo might be cultivating marijuana with grow lights in his Florence, Ore., home in 1992. (Alas, it didn't
reveal anyone in the household getting busy.) Based on the thermal imager's discovery of a pattern of
heat loss against one wall and the garage, the police obtained a search warrant of the Kyllo premises.
The question for the court is whether the police should have gotten a search warrant first before
they ran the thermal scanner.
Here's the quick-and-dirty on police searches under the Fourth Amendment: People have a reasonable
expectation of privacy in their own homes. The state needs a warrant to intrude on that privacy. Unless
you're committing homosexual sodomy in Georgia, you're entitled to privacy from governmental intrusion in
your bedroom. Your right to privacy within your own home ebbs somewhat in front of open windows,
however, and it stops when your garbage hits the curb. In the vast Pushmepullyou of privacy law, dog-sniffs
of your person have been deemed OK, wiretaps require warrants, while aerial photography of your property
is OK. Today's case involves more working out of the details.
Sadly, while more sophisticated thermal imagers can indeed pick up the heat from bodies "commingling,"
as it's euphemized, the whole fruitful line of inquiry as to whether one has an expectation of privacy while
getting busy is nipped in the bud today. Justices Scalia and Kennedy manage to persuade the court that its
only task is to determine the constitutionality of this scan and not the Fourth Amendment consequences of
thermal imagers in general.
Since Kyllo v. United States involves the tension between sophisticated surveillance equipment and the
Fourth Amendment's privacy protections, I ordered a pair of those plastic X-ray glasses from the back of an
Archie comic last week. With virtually unlimited power to see what is happening under the judicial robes, a
whole new world is revealed this morning.
Kenneth Lerner is arguing on behalf of Kyllo, who was sentenced to 63 months in prison for manufacturing
marijuana. After this case made many trips back and forth between the 9 th Circuit and the district court, the
panel ruled that thermal imaging of a home absent a warrant doesn't violate the Fourth Amendment. Lerner
opens by explaining that illegal searches of the home are "the chief evil the Fourth Amendment was
designed to protect against." Justice Souter (wearing a hair shirt under his robe) asks whether a video in
evidence, showing people moving around the interior of a home, is the thermal scan in question today.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 184
The Professional Thermographers Association
Chief Justice Rehnquist asks whether the district court didn't already give full consideration to that tape, but
Justice Ginsburg cuts Lerner off midreply with another question. Rehnquist turns to her and snaps, "I think
he's answering my question." Ginsburg is deflated for the rest of the argument (this despite the fact that
she's sporting a fabulous Victoria's Secret matched set in a snakeskin print).
As is clear from the above ramble through privacy law, one problem underlying this case is that it involves
death by a million analogies. The result turns on whether thermal imaging is more like putting a bug on the
outside of a pay phone to hear what the average policeman's ear cannot detect (a "search" proscribed in
Katz v. United States) or more like using police dogs to sniff what the human nose can't detect (a search
permitted under United States v. Place). If you need to believe there are principled, jurisprudential reasons
for prohibiting phone bugs but allowing dog-sniffs, might I offer a unifying constitutional theory: The Fourth
Amendment would bar Jamie Summers from using her bionic ear, but not Max the bionic dog from using
his supernose. Leaving us to decide today whether Steve Austin's bionic eye can be used without a
warrant.
Unfortunately the whole day degenerates into a game of "Distinguish This!" with Scalia (blue Superman
tights under the robe) asking Lerner how thermal imaging differs from simple police observation that snow
melts faster off drug houses than ordinary homes. Ginsburg wonders what you can determine from thermal
imagery that you can't find out from legally obtained utility bills. Lerner replies that utility bills "vary
depending, frankly, on how many women live in the house," due, he observes, to the fairer sex's
unfortunate tendency to shower and do laundry. In other words, utility bills provide information that's too
general to support a search warrant, whereas thermal imaging distinguishes between grow lights over the
pot plants and the George Foreman grill. In other words, if you're growing pot in your basement and don't
want it reflected in your utility bills, either lose the missus or stop showering.
Justice O'Connor (nylon slip, J.C. Penney) asks how dog-sniffs can be OK if they also enhance human
smell, and Scalia asks if a policeman with 10/10 vision is allowed to look in your windows.
"10/10 vision?" replies Lerner.
"I'm assuming that's better than 20/20."
Justice Stevens asks if this entire case simply revolves around the sophistication of thermal-imaging
equipment. Under his robe, he is wearing those pajamas Max wore in Where the Wild Things Are just
before the wild rumpus starts. Then he confuses everyone by asking whether cops could rent the house
next to Kyllo's, tie thermometers to "long poles," and track the temperature that way. Lerner says
thermometers, watches, and "regular" devices are OK.
Souter, exasperated, wonders whether the day will come when "every schoolchild has a $5 thermal
imager," and then there won't be a constitutional violation. I'd guess that even using the Catamount
Radiance HSX 3-5 micron IR Camera (prices start at $71,837) you'd get a flat-line off Justice Souter. He
does get Lerner to admit that the technological sophistication of thermal imagery is probably not the best
way to distinguish the case from phone-tap cases. At which point, Rehnquist (who under his robe is
sporting another robe, with more stripes on the sleeves) and Scalia take turns plying Lerner with questions
about police use of flashlights.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 185
The Professional Thermographers Association
Michael Dreeben from the solicitor general's office argues on behalf of the law-enforcement community,
arguing that thermal imagers don't monitor objects or activity; they just measure "lost heat." He
differentiates the Katz phone-bugging case by explaining that no one is capturing "words spoken in a
private place" but rather, "heat generated by the use of power." Souter wonders whether "if someone wants
to spend his time lying under sunlamps, isn't that his own business?"
Then Justice Breyer—reminding us that there is indeed a test for illegal searches under the Fourth
Amendment—asks how someone can have a "reasonable expectation of privacy" from a technology no
one has even heard of. "Every bird-watcher has binoculars ... every Boy Scout has a flashlight. Who has a
heat thermal device?"
Breyer wears black bikini briefs, and here's how I know: He continues on, "What I'm doing in my bathroom.
... I happen to like the sauna, I turn on every shower."
"That would steam up the windows," crows Dreeben.
"I don't have windows. It's all modern Finnish wood. ... I usually spend three hours a day in my Finnish
sauna. I don't want people to know that. People think I'm working." Stephen Breyer. The Hugh Hefner of the
federal bench.
Scalia, Souter, Dreeben, and Kennedy engage in a long colloquy about whether it's better or worse that
thermal imagers allow police to draw better inferences about the activities inside a home, without providing
conclusive evidence. Kennedy, by the way, is a boxer man, if ever one were born on this green earth.
The test for illegal police searches has two parts after Katz: Does the individual have a subjective
expectation of privacy in the place searched, and is that expectation objectively reasonable? So, if this case
indeed turns on whether pot-growers expect privacy in their homes, there can be no debate: This is why
pot-growers so rarely tend their crops in kitchen window boxes. But the case can be abstracted away—as it
was today—by asking only if pot-growers have any reasonable expectation of privacy in the heat they emit.
Well, of course not. With the exception of Justice Breyer and his dirty little sauna secrets, none of us knew
our heat emissions would be cause for criminal investigation.
Which brings us back to Danny Kyllo, who may have successfully circumvented the laws of the land but
couldn't manage to get round the laws of physics.
Dahlia Lithwick is a Slate senior editor.
Article URL: http://www.slate.com/id/101221/
Copyright 2006 Washingtonpost.Newsweek Interactive Co. LLC
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 186
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 187
The Professional Thermographers Association
Appendix: 12 STEPS FOR SETTING UP A SUCCESSFUL
CONDITION MONITORING PROGRAM
1. Determine existing maintenance costs - this data will be used for initial justification and a baseline for
measuring the programs success.
2. Justify Implementation - be specific. Do not overstate return on investment.
3. Select the best Infrared Instrument and supplier - longevity and support are important considerations.
4. Get management support - lack of total commitment from management to provide resources to
implement and maintain a program is the single largest reason for failure.
5. Develop a detailed program plan - the plan should include goals, objectives and methods used to
implement, maintain and evaluate program benefits.
6. Dedicated staff.
7. Accountability - the staff must be accountable for failure or success of program.
8. Database - benefits of the program will depend on the accuracy and completeness of the database,
which takes effort.
9. Maintain the program - constantly evaluate and correct problems as they occur.
I0. Communicate - management and other related staff must be continually informed of the programs status
and benefits.
11. Training - develop a comprehensive training program. Staff will need to continue their development
throughout the programs life. Remember, the staff cannot absorb and retain all the knowledge they need
with start-up training.
12.Technical support - the operator cannot know it all. Plan for technical support or engineering assistance
to resolve problems.
Infrared Program Implementation and Management
Establishing An IR Program
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 188
The Professional Thermographers Association
One of the first steps in the process of establishing an infrared program is to determine the direction that
your program will take. Although this might sound like a simple task, there is much more to it than meets
the eye. First you must establish the fact that you need the program and that it will have benefits either
financial, safety related or a combination of these and other benefits.
Once the need has been identified you must get your program off the ground and make it successful. It
needs to be “sold” to all levels of participants. Without the buy in of everyone from management on down
there is a good chance your program will not survive. This entails a careful examination of how the
program will effect each area that will be touched by the new program. It is not enough to simply describe
the program, one must make the benefits of the program part of the goals to be reached. The programs
that seem to achieve the greatest support are those that are sold from the top down.
To get management to accept the new program, a presentation highlighting the financial benefits is usually
the most attractive approach. In a predictive maintenance program, the increased revenue from the
extended uptime an IR program can provide is on the top of the list. To make this a realistic prediction one
must try and locate some of the history records as they relate to the downtime that an IR predictive
maintenance program might have prevented. If this is not possible, there are many articles in various trade
magazines that showcase the savings made in their location by an IR program. It is best to try and use an
article that features a facility that is similar to your own.
When doing a presentation of this type, whether it is a written proposal or a personal presentation, one
must also include the costs of such a program. The cost should detail capital costs as well as ongoing
support costs. Manpower and necessary supplies should be included in your proposal. The time frame for
implementation of the program is an important item from a management point of view. Be realistic as the
figures you present will be the benchmarks for the success of your program should it be accepted.
The items to include in your management presentation could include but are not limited to:
Direct financial benefits
decreased down time, increased uptime
decreased parts costs - not as many catastrophic failures.
lower parts inventory costs, no stocking of unnecessary parts (true predictive capabilities of
program)
decreased labor costs - not as many overtime hours for unscheduled outages
possible quality control issues - increased profits
Safety Issues
Employee satisfaction
lower stress levels, not just “putting out fires”
pride of work, especially when in competitive production situations.
gives employees sense of ownership if they are responsible for the success of the program
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 189
The Professional Thermographers Association
Once the program has been accepted by management it must then be presented to and accepted by both
supervisory personal and general personal. Many of the above issues can be included in these
presentations.
It is extremely important to get each of these groups to buy in to the program. Failure to do so can lead to
unwanted resistance to the program. When addressing the supervisors, the benefits of having better control
of their time due to reliable planning can be a cornerstone. Many of the benefits outlined to management
can also be used in this presentation as well.
When dealing with the operators on the floor, the easiest way to get them to participate in the program is to
involve them from the beginning by asking for their input into the setup of the program. Much of the needed
history can sometimes be obtained from these floor personal. Without their support, the program will not
succeed.
Scope/Mission Statement
One of the important components of your presentation could be a mission statement . This statement could
also be used as a cornerstone for the scope portion of your general written practice which describes your
Infrared Program. This should clearly the direction of your program and the intended benefits.
Written Practices
Documentation is often one of the last details to be addressed in the performance of many tasks. To be in
compliance with many of the new standards arising, documentation such as written practices have an
increased prominence. A good guide to aid in the preparation of these materials is “A Model Written
Practice” distributed by ASNT. There are a number of areas that should be addressed by your written
practices.
The examples below show what documents could cover but should not be limited to:
1. Written Practice for Infrared/Thermal Department Procedures
2. Written Practice for the Qualification and Certification of Personnel in the Infrared Thermal Testing
Method
3. Written Practice for Electrical Inspections
4. Written Practice for Mechanical Inspections
5. Written Practice for Furnace Tube Inspections
6. Written Practice for Preparing and Presenting Inspection Reports.
Written Practice for Calibration Procedures.
These written practices should all have the same general format. They should contain sections which
cover:
1.
2.
3.
4.
Name, author, date, written practice number
Scope
Referenced Documents
Definitions
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 190
The Professional Thermographers Association
5. The body of the document which contains all pertinent
should be itemized in sections.
information and procedures. These
Qualification and Certification of Personnel
The qualification and certification of personnel in your program should be described in detail in your written
practice. The document SNT-TC-1A gives you the best model to follow for this purpose. The document
should have the following sections:
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
Title, Author, Date
Scope
Definitions
References
Levels of Qualification
Education Requirements
Training Requirements - Topical Outlines for Each Level
Experience Requirements
Examination Requirements - Written, Practical
Certification
Recertification
Documentation
These information in these sections should be well thought out as they will affect the quality of not only the
personnel in the department but also the quality of the product produced by your department and ultimately
the success of your department.
Inspection Procedures
One of the first steps in activating your IR/T program is to determine what types of applications will be
addressed and the areas in your location which will be covered. You need to determine inspection
procedures. This includes establishing:
Baselines
routes
priorities/criticality
inspection frequency
inspection report formats
fault reinspection procedures
cost analysis
data bases/equipment histories
The key to many of these items can be found in the proper implementation of a detailed database.
The database should be a relational database. For an electrical database for example, the fields could
contain a variety of information.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 191
The Professional Thermographers Association
Area - general area or building where the equipment is located
Location - specific location room etc.
Equipment - the name or number of the individual piece of equipment
Item - ie starter, disconnect, transformer
Date - inspection date
Manufacturer - who made the product, to be used in future purchase recommendations
Inspection Frequency - how often to inspect
Criticality to operation - critical, essential, nonessential, other
Route
Actual temperature
Fault Temperature rise
Repairs
There are any number of items that could be included depending on the application needs.
The main database or a separate database or spread sheet program must detail the savings generated by
your IR/T program. It should have two categories for each anomaly found, a fixed as found and a run to
failure side. Both should detail the cost of the parts, the cost of the labor to repair and the cost of downtime.
Fixed as Found with IR
Run to Failure
Parts
$0.00
Parts
Labor
$0.00
Labor
Lost Production
$0.00
Lost Production
Totals
$0.00
Savings Realized
$0.00
$0.00
$0.00
$0.00
$0.00
A running total should be kept of these savings. These values could also be a part of each report page
generated.
Monitoring and Promoting Your Program
Proper documentation allows you not only to monitor the success of your program but also to promote your
program by issuing monthly, quarterly and annual reports. The benchmarks used should include not only
the money saved but also the reduction in downtime, the reduction in percentage of faults per items
inspected, recommendations on equipment type, improved practices etc. These reports should be
circulated to management as well as supervisors and operators or line personnel.
If the company has a newsletter or bulletin, try and get some of your larger prominent “saves” published.
Feature articles stimulate interest in your program and often open doors for further application areas.
The key to the program’s success is document, document, document!!!
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 192
The Professional Thermographers Association
Safety
It is beyond the scope of this manual to consider safety issues. The main principle when dealing with safety
is that you must comply to all safety requirements in effect at your location. These encompass OSHA
standards, NFPA rulings, federal and state requirements as well as the safety policies that are in force
within your own organization. The new IR systems have no hazards such as liquid nitrogen. Safety issues
that pertain to each application should be incorporated into your individual written procedures where
possible.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 193
The Professional Thermographers Association
Appendix: Existing IR Related Standards
There are a number of organizations around the world that have established standards that are in some
way related to Infrared Thermography. They are made up of a number of testing specifications as well as
safety standards. This is by no means a complete list but does give you a good general idea of what is
available.
There are also a number of written procedures that are helpful in application considerations. We have also
listed a few of those and included them as examples.
American Society for Testing and Materials
ASTM C 177 Standard Test Method for steady-State Heat Flux Measurements and Thermal Transmission
Properties by Means of the Guarded-Hot-Plate Apparatus
Scope: 1.1 This test method covers the achievement and measurement of steady-state heat flux through
flat-slab specimens using a guarded-hot-plate apparatus. The method encompasses both the single-sided
and the double-sided mode of measurement. Both distributed and line source guarded heating plate
designs are included, in principle, in this test method. The reader should consult the standard practices on
the single-sided mode of operation and on the line source apparatus for further details on these variations
of the method.
ASTM C 236 Standard Test Method for Steady-State Thermal Performance of Building Assemblies by
Means of a Guarded Hot Box
Scope: 1.1 This test method, known as the guarded hot box method, covers the measurement of the
steady-state thermal transfer properties of panels. In distinction to Test Method C 177, which is primarily
applicable to homogeneous samples, the guarded hot box method provides for the evaluation of thermal
performance of building assemblies. This test method is suitable for building construction assemblies,
building panels, and other applications of non-homogeneous specimens.
ASTM C 335 Standard Test Method for Steady-State Heat Transfer Properties of Horizontal Pipe Insulation
Scope: 1.1 This test method covers the measurement of the steady-state heat transfer properties of pipe
insulations installed on a horizontal test pipe operating at temperatures above ambient. Specimens may be
rigid, flexible, or loose fill, may be homogeneous or non-homogeneous, isotropic or non-isotropic, and of
circular or non-circular cross section. Measurement of metallic reflective insulation and mass insulations
with metal jackets or other elements of high axial conductance is included; however, additional precautions
must be taken and specified special procedures must be followed.
ASTM C 351 Standard Test Method for Mean Specific Heat of Thermal Insulation
Scope: 1.1 This test method covers the determination of mean specific heat of thermal insulating materials.
The materials must be essentially homogeneous and composed of matter in the solid state. 1.2 This test
method employs the classical method of mixtures. This provides procedures and apparatus simpler than
those generally used in scientific calorimetry, an accuracy that is adequate for most thermal insulating
purposes, and a degree of precision that is reproducible by laboratory technicians of average skill. While
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 194
The Professional Thermographers Association
this test method was developed for testing thermal insulations, it is easily adaptable to measuring the
specific heat of other materials.
ASTM C 518 Standard Test Method for Steady-State Heat Flux Measurements and Thermal Transmission
Properties by Means of the Heat Flow Meter Apparatus
Scope: 1.1 This test method covers the measurement of steady state thermal transmission through flat slab
specimens using a heat flow meter apparatus. 1.2 This is a comparative (or secondary) method of
measurement since specimens of known thermal transmission properties must be used to calibrate the
apparatus. Properties of the calibration specimens must be traceable to a recognized national standards
laboratory.
ASTM C 680 Standard Practice for Determination of Heat Gain or Loss and the Surface Temperatures of
Insulated Pipe and Equipment Systems by the Use of a Computer Program
Scope: 1.1 This computer programs included in this practice provide a calculational procedure for
predicting the heat loss or gain and surface temperatures of insulated pipe or equipment systems. This
procedure is based upon an assumption of a uniform insulation system structure, that is a straight run of
pipe or flat wall section insulated with a uniform density insulation. Questions of applicability to real
systems should be resolved by qualified personnel familiar with insulation systems design and analysis. In
addition to applicability, calculational accuracy is also limited by the range and quality of the physical
property data for the insulation materials and systems.
ASTM C 745 Standard Test for Heat Flux Through Evacuated Insulatons Using a Guarded Flat Plate Boiloff
Calorimeter
Scope: 1.1 This test method covers the determination, from cryogenic to near room temperatures, of heat
flux through evacuated insulations within the approximate range from 0.3 to 30 W/m2. Heat flux values
obtained using this method apply strictly only to the particular specimens as tested. 1.2 This shall be a
primary test method for measuring heat flux through evacuated insulations, since calibration of the
apparatus depends on measurement standards traceable to the National Bureau of Standards (NBS) for
length, force, temperature, time, etc. Traceable standards are not yet available for heat flux through
standard evacuated reference specimens or transfer standards.
ASTM C 835 Standard Test Method for Total Hemispherical Emittance of Surfaces from 20 to 1 400 C
Scope: 1.1 This calorimetric test method covers the determination of total hemispherical emittance of metal
surfaces from approximately 20 to 1 400 C. The upper-use temperature is limited only by the melting
temperature of the specimen and the design limits of the test facility. This test method has been
demonstrated for use up to 1 400 C.
ASTM C 1041 Standard Practice for In-Situ Measurements of Heat Flux in Industrial Thermal Insulation
Using Heat Flux Transducers
Scope: 1.1 This practice covers the in-situ measurement of heat flux through industrial thermal insulation
using a heat flux transducer (HFT). 1.2 This practice estimates the thermal transport properties of thermal
insulation material in-situ in field applications under pseudo steady-state conditions. It is not intended that
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 195
The Professional Thermographers Association
this practice should be used as a substitute for more precise laboratory procedures such as Test Methods
C 177, C 335, or C 518.
ASTM C 1044 Standard Practice for Using the Guarded-Hot-Plate Apparatus in the One-Sided Mode to
Measure Steady-State Heat Flux and Thermal Transmission Properties
Scope: 1.1 This practice covers the determination of the steady-state thermal transmission properties of
insulating specimens using a guarded hot plate in a one-sided configuration. It represents a supplemental
procedure to Test Method C 177, which applies only to a two-sided configuration. 1.2 This practice is
limited to the difference in the procedure for measuring the heat flow between the two-sided method, Test
Method C 177, and this one-sided practice. For the rest of the practice, Test Method C 177 applies.
ASTM C 1045 Standard Practice for Calculating Thermal Transmission Properties From Steady-State Heat
Flux Measurements
Scope: 1.1 This practice provides requirements and guidelines for the determination of thermal
transmission properties based upon heat flux measurements under a variety of conditions. The practice is
directed particularly toward a description of the heat flux and associated measurements necessary to
obtain useful properties that are applicable to end-use conditions. 1.2 The thermal conductance, apparent
thermal resistivity, and apparent thermal resistance.
ASTM C 1046 Standard Practice for In-Situ Measurement of Heat Flux and Temperature on Building
Envelope Components
Scope: 1.1 This practice details a technique for using heat flux and temperature transducers in
measurements of the dynamic or steady state thermal behavior of opaque building components. The
applications for such data include estimation of R-values or of thermal time constants. However, such uses
are beyond the scope of this practice (for information pertaining to the estimation of R-values, see Practice
C 1155).
ASTM C 1055 Standard Guide for Heated System Surface Conditions That Produce Contact Burn Injuries
Scope: 1.1 This guide establishes a process for the determination of acceptable surface operating
conditions for heated systems. The human burn hazard is defined, and methods are presented for use in
the design or evaluation of heated systems to prevent serious injury from contact with the exposed
surfaces.
ASTM C 1057 Standard Practice for Determination of Skin Contact Temperature from Heated Surfaces
Using a Mathematical Model and Thermesthesiometer
Scope: 1.1 This practice establishes a procedure for evaluating the skin contact temperature for heated
surfaces. Two complimentary procedures are presented. The first is a purely mathematical approximation
that can be used during design or for worst case evaluation. The second method describes the
thermethesiometer, an instrument that analogues the human sensory mechanism and can be used only on
operating systems.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 196
The Professional Thermographers Association
ASTM C 1060 Standard Practice for Thermographic Inspection of Insulation Installations in Envelope
Cavities of Frame Buildings
Scope: 1.1 This practice is a guide to the proper use of infrared imaging systems for conducting qualitative
thermal inspections of building walls, ceilings, roofs, and floors, framed in wood or metal, that may contain
insulation in the spaces between framing members. This procedure allows the detection of cavities where
insulation may be inadequate or missing and allows identification of areas with apparently adequate
insulation.
ASTM C 1061 Standard Test Method for Thermal Transmission Properties of Non-Homogeneous Insulation
Panels Installed Vertically
Scope: 1.1 This test method, known as the high temperature hot box method (calibrated or guarded),
covers the measurement of thermal conductance, thermal transference, and thermal transmittance of
insulation panels from a heated surface temperature, of approximately 40 C (104 F) to the maximum
insulation design temperature, not to exceed 540 C (1000 F). In distinction to Test Method C 177, which is
primarily applicable to homogeneous samples, and test method C 236, which is primarily applicable to
measurements on panels representative of such constructions as walls, roofs, and floors of buildings, the
high-temperature hot box method is designed for measurements on non-homogeneous panels
representative of such constructions as metal reflective insulation panels for nuclear power plants and
prefabricated insulation panels for fossil power plants.
ASTM C 1130 Standard Practice for Calibrating Thin Heat Flux Transducers
Scope: 1.1 This practice establishes an experimental procedure for determining the sensitivity of heat flux
transducers (Huts) that are relatively thin. The term sensitivity in this practice refers to the ratio of HFT
electrical output to heat flux through the HFT. 1.1.1 For the purpose of this standard, the thickness of the
HFT shall be less than 15% of the narrowest planar dimension of the HFT. 1.2 This practice discusses two
methods for determining HFT sensitivity. The first method is the calibration of the HFT in unperturbed heat
flow normal to the surface of the HFT, while the second method is the sensitivity of the HFT in actual use,
or the HFT conversion factor.
ASTM C 1153 Standard Practice for the Location of Wet Insulation in Roofing Systems Using Infrared
Imaging
Scope: 1.1 This practice applies to techniques that employ infrared imaging at night to determine the
location of wet insulation in roofing systems that have insulation above the deck in contact with the
waterproofing. This practice includes ground-based and aerial inspections. 1.2 This practices addresses
criteria for infrared equipment such as minimum resolvable temperature difference, spectral range,
instantaneous field of view, and field of view. 1.3 This practice addresses meteorogical conditions on
infrared inspections should be performed. 1.4 This practice addresses the effect of roof construction,
material differences, and roof conditions on infrared data using invasive test methods. 1.7 The values
stated in SI units are to be regarded as standard.
ASTM C 1155 Standard Practice for Determining Thermal Resistance of Building Envelope Components
from In-Situ Data
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 197
The Professional Thermographers Association
Scope: 1.1 This practice details how to use data obtained from in-situ measurement of temperatures and
heat fluxes on building envelopes to compute thermal resistance. Each thermal resistance calculation
applies to a subsection of the building envelope component that was instrumented. Each calculation
applies to temperature conditions similar to those of the measurement. The calculation applies of thermal
resistance from in-situ data represents actual conditions. However, field measurements of temperature and
heat flux may not achieve the accuracy obtainable in laboratory apparatuses.
ASTM E 220 Standard Method for Calibration of Thermocouples By Comparison Techniques
Scope: 1.1 This method covers the techniques of thermocouple calibration based upon comparisons of
thermocouple indications with those of a reference thermometer, different from method involving the use of
fixed points. The precise evaluation of the electromotive force (emf)-temperature relation of a
thermocouple is accomplished by determining its emf output at each of a series of measured temperatures.
Calibrations are covered over temperature ranges appropriate to the individual types of thermocouples
within an over-all range from about –180 to 1 700 C (-290 to 2 660 F). 1.2 In general, the method is
applicable to bare wire thermocouples. The latter may require special care to control thermal conduction
losses.
ASTM E 230 Standard Temperature-Electromotive Force (EMF) Tables for Standardized Thermocouples
Scope: 1.1 This standard consists of reference tables (Tables 4 to 9) that give temperature-electromotive
force (emf) relationships for Types B, E, K, N, R, S, and T thermocouples. These are the thermocouple
types most commonly used in the industry. 1.2 Also included is a list (Table 1) of standard and special
tolerances on initial values of emf versus temperature (tolerances), and recommendations (Table 2)
covering upper temperature limits for the thermocouple types referred to in 1.1.
ASTM E 344 Terminology Relating to thermometry and Hydrometry (Definitions)
ASTM E 452 Standard Test Method for Calibration of Refractory Metal Thermocouples Using an Optical
Pyrometer
Scope: 1.1 This method cover the calibration of refractory metal thermocouples using an optical pyrometer
as the standard instrument. The method is intended for use with types of thermocouples that cannot be
exposed to an oxidizing atmosphere. These procedures are appropriate for thermocouple calibrations at
temperatures above 800 C (1 472 F).
ASTM E 563 Standard Practice for Preparation and Use of Freezing Point Reference Baths
Scope: 1.1 This practice covers the proper preparation and use of freezing point reference standards,
especially for the ice point, and to outline the likely causes of error. It is intended for the guidance of the
industrial user of ASTM standards that call for the use of freezing point references.
ASTM E 644 Standard Test Methods for Testing Industrial Resistance Thermometers
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 198
The Professional Thermographers Association
These test methods describe the principles, apparatus, and procedures for calibration and testing of
resistance thermometers. 1.2 These test methods cover definitions of terms and tests for insulation
resistance, interchangeability, calibration, immersion error, pressure effects, thermal response time,
vibration effect, mechanical shock, self-heating effect, stability, thermoelectric effect, humidity, and thermal
hysteresis. 1.3 This series of tests is not necessarily intended nor recommended to be performed on every
thermometer. Not all the tests are appropriate for all types of thermometers. The expected limits of results
are given in the test procedures and also tabulated in Appendix X4.
ASTM E 839 Standard Test Methods for Sheathed Thermocouples and Sheathed Thermocouple Material.
Scope: 1.1 These are test methods for sheathed thermocouple material and assemblies. 1.2 The tests are
intended to assure quality control and to evaluate the suitability of sheathed thermocouple material or
assemblies for specific applications. Some alternative test methods to obtain the same information are
given, since in a situation an alternative test method may be more practical. Service conditions are widely
variable and so it is unlikely that all the tests described will be appropriate for a given thermocouple
application. A brief statement is made following each test description to indicate when it might be used.
1.3.4.7 Thermal response time, and 10.9.1 General Test Method—The test thermocouple assembly is
stabilized at room temperature and then immersed rapidly in a flowing fluid, usually water, at a higher
temperature. The voltage from the thermocouple assembly is measured as a function of time until it comes
to the thermal equilibrium. The time from the instant of immersion until the voltage has attained the specific
percentage of its total change is recorded as the thermal response time.
ASTM E 988 Standard Temperature-Electromotive Force (EMF) Tables for Tungsten-Rhenium
Thermocouples
Scope: 1.1 This standard consists of reference tables that give temperature-electromotive force (emf)
relationships for 97% Tungsten 3% Rhenium versus 75% Tungsten 25% Rhenium and 95% Tungsten 5%
Rhenium versus 74% Tungsten 26% Rhenium thermocouples. These are the refractory metal
thermocouple types most commonly used in industry. 1.2 Also included is a list (Table 1) of initial
calibration tolerances for the thermocouple types referred to in 1.1 and their respective compensating
extension wires (Table 2). 1.3 These data are intended for industrial and laboratory use.
ASTM E 1213 Standard Test Method for Minimum Resolvable Temperature Difference for Thermal Imaging
Systems
Scope: 1.1 This test method covers the determination of the minimum resolvable temperature difference
(MRTD) capability of the compound observer-thermal imaging system as a function of spatial frequency.
ASTM E 1256 Standard Test Methods for Radiation Thermometers ( Single Waveband Type)
Scope: 1.1 The following methods describe standard tests that can be performed to determine the basic
operation of a temperature measuring instrument. The test methods described can be utilized to determine
the following parameters:
Section
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 199
The Professional Thermographers Association
Accuracy
Repeatability
Sensitivity
Spectral Response Limits
Target Size
Response Time
Warm-up Time
Long Term Drift
6
7
8
9
10
11
12
13
ASTM E 1311 Standard Test for Minimum Detectable Temperature Difference for Thermal Imaging
Systems
Scope: 1.1 This test method covers the determination of the minimum detectable temperature difference
(MDTD) capability of a compound observer-thermal imaging system as a function of the angle subtended
by the target.
ASTM E 1316 Standard Terminology for Nondestructive Examinations
Scope: 1.1 This standard defines the terminology used in the standards prepared by the E-7 committee on
the Nondestructive testing. These nondestructive testing (NDT) methods include: acoustic emission,
electromagnetic testing, magnetic particle testing, neutron radiography and gauging, ultrasonic testing and
other technical methods.
Note: They also include terms for infrared thermography
Section A defines terms that are common to all NDT methods, and the subsequent sections include the
terms pertaining to a specific NDT method. An alphabetical list of the terms defined in this standard is
given in Appendix X1, which also identifies the section in which each term is defined.
Application Related Standards and Procedures
Public Works Canada: Report #30 - A Generic Method for Thermographic Diagnosis of Building Enclosures
This report describes general considerations that should be considered when performing thermographic
inspections of industrial buildings
BSRIA AG 17/97 Safe Thermal Imaging of Electrical Systems
(1000 V AC)
Provides guidance on how to use this highly effective diagnostic tool in safety. This guidance is the result of
extensive research and industry consultation. It provides an approach to risk assessment for
site/environment, system and activity.
Danish Technological Institute Guidelines for Thermographic Inspection in Electrical Installations
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 200
The Professional Thermographers Association
A general guideline to electrical inspections.
IEEE Various guidelines relating to allowable temperatures for electrical components. (examples included)
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 201
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 202
The Professional Thermographers Association
Appendix: Electrical Inspections
Standard Guide for Examining Electrical Power Distribution
Equipment with Infrared Thermography: DRAFT as of
9/18/2006
This standard is issued under the fixed designation E1934; the number immediately
following the designation indicates the year of original adoption or, in the case of revision,
the year of last revision.
1. Scope
1.1 This guide lists the responsibilities of the owner and the infrared thermographer when examining
electrical power distribution equipment. Such equipment includes:
1.1.1 Panelboards and equipment control panels, switchboards and equipment control panels with
molded case or insulated case circuit breakers; enclosed electrical bus and bus plugs, motor control
centers (MCCs), motors, switchgear (with power circuit breakers or fused switches), substations, and other
electrical Equipment.
1.1.2 While the principles of this Guide apply, additional special techniques and equipment may be
required when conducting examinations of electrical equipment out-of-doors in substations and for
overhead lines.
1.2 This guide outlines specific procedures to conduct and document infrared examinations of
electrical power distribution equipment and control circuits.
1.2.1 Examinations of electrical equipment operating at voltages in excess of 600 volts may have
special requirements, especially for safe access; some of these are addressed in documents NFPA 70E
and NFPA 70B, listed below.
1.3 This guide involves exposure to and the use of equipment in the presence of electrically energized
equipment, which presents potential hazards due to electrocution and arc-flash.
1.3.1 This guide recognizes the inherent hazard associated with examining energized electrical
equipment. It is the responsibility of the owner of this standard to ensure that all personnel are properly
trained and qualified (1.4.1) and that appropriate safety precautions are observed (1.4.2).
1.3.2 The American Society for Nondestructive Testing (ASNT) standard SNT-TC-1A provides specific
recommended guidance for minimum qualifications and training of personnel involved in infrared
thermography. All work should be conducted only by qualified personnel.
1.3.3 The National Fire Protection Association (NFPA) 70E “Standard for electrical safety requirements
for employee workplaces”, provides specific, required guidance regarding examination protocol, electrical
risk hazard assessment and personal protective equipment requirements when working inside arc flash
protection boundaries. This information should guide all examination work.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 203
The Professional Thermographers Association
1.3.4 The National Fire Protection Association (NFPA) 70B ”Recommended practice for electrical
equipment maintenance” provides additional, useful guidance on the maintenance of electrical systems,
including the use of infrared examinations for the maintenance process.
1.3.5 This guide does not purport to address all safety concerns, as many safety concerns are specific
to the circumstances. As such, it is the responsibility of the owner to ensure that all persons involved in this
work are properly trained and qualified to perform this work in the owner’s facility.
1.4 While this guide may also be useful when using non-imaging radiometers to conduct examinations,
it specifically addresses examinations conducted using infrared imaging equipment, either radiometric or
non-radiometric.
2. Referenced Documents
2.1 ASTM Standards:
2.1.1 E1316 Terminology for Nondestructive Examinations
2.2 Code of Federal Regulations 29
2.2.1 Parts 1900 to 1910 (to End) Labor
2.2.2 Part 1926 Safety and Health Regulations for Construction
2.3 National Fire Protection Association: NFPA 70 National Electric Code
2.4 National Fire Protection Association: NFPA 70B
2.5 National Fire Protection Association: NFPA 70E
2.6 American Society for Nondestructive Testing: ASNT SNT-TC-1A
2.7 American Society for Nondestructive Testing: ASNT/ANSI CP-189
3. Terminology
3.1 Anomaly: a system or component that is not functioning within acceptable limits; this may be, but is
not always, indicated by a temperature difference from normal.
3.2 Blackbody: the ideal, perfect emitter and absorber of thermal radiation. It emits radiant energy at
each wavelength at the maximum rate possible as a consequence of its temperature, and absorbs all
incident radiance.
3.3 Blackbody reference: a calibrated, traceable device used to check the calibration of an infrared
imaging radiometer or spot radiometer
3.4 Finding: a component or system, whether anomalous or normal, that has been documented with a
thermal image
3.5 Infrared system: an electronic camera used to detect emitted thermal energy; also called a thermal
imaging system
3.6 Owner: the party responsible for the operation of the electrical systems and components
3.7 Qualified escort: the person assisting the thermographer, typically by locating equipment, opening
enclosures, making load readings, and securing the equipment after the examination, among other
activities
3.8 Qualitative infrared examination: an examination that does not include radiometric temperature
data but does provide thermograms showing thermal relationships
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 204
The Professional Thermographers Association
3.9 Quantitative infrared examination: an examination that records radiometric temperature data
provided by the infrared system
3.10 Radiometric temperature measurement: the measurements made using an infrared system by
correlating the radiation emitted by a surface with its temperature; such data must be corrected for
emissivity, transmissivity and reflected background temperature
3.11 Spot radiometer: a non-imaging device that detects infrared radiation
3.12 Thermogram: the image produced by a thermal imaging system showing the thermal relationships
of the equipment being examined.
3.13 Thermographer: a person who is qualified to conduct an examination using thermal imaging
system.
4. Summary of Practice
4.1 There are two primary benefits of conducting an infrared examination of an electrical system:
4.1.1 To validate that the electrical systems and components are operating within acceptable limits.
4.1.2 To detect abnormal conditions in the electrical system and/or components that might result in
adverse operations, equipment damage, system failures or unscheduled outages.
4.2 A secondary benefit may be to provide quantitative radiometric temperatures of components but it
must be noted that for many electrical systems and components these are often unreliable, difficult to
interpret and trend, or not representative of the actual conditions found.
4.3 The non-contact examination is conducted, typically, on energized, exposed electrical components;
appropriate safe work practices must be used at all times.
4.4 It is essential that certain conditions exist for the results to be considered valid, including, among
others, having the system energized and loaded, having a clear line of sight of the components, and
ensuring that the effect of ambient conditions on heat transfer relationships is fully understood.
5. Significance and Use
5.1 This guide can be used by an owner to specify how an infrared examination of electrical equipment
shall be conducted and who is qualified as an infrared thermographer to perform such examinations.
5.2 The purpose of infrared examinations of electrical equipment is to document the "as-found"
condition of the equipment. When this involves the identification of anomalous components or systems,
further measures or testing are generally required to fully understand the root cause of the anomaly, to
correct problems before failures occur, or when appropriate to "run to failure," to minimize consequences.
5.3 Anomalies in electrical components can be generally grouped in one of two categories:
5.3.1 Abnormal electrical resistance at points of electrical contact, such as connectors and switch
mechanisms, which is typically caused by loose or deteriorated connections, improperly installed
components, corrosion, and metal fatigue.
5.3.1.1 Thermal signatures for these types of anomalies will typically be warmer than normal at the
high-resistance contact point with diminished temperatures as distance increases from that point.
5.3.2 Excessive current flow caused by undersized conductors and abnormal load imbalances.
5.3.2.1 Thermal signatures for these types of anomalies will typically be equally warmer (or
occasionally equally cooler) than normal throughout the entire circuit, phases or portion of the system
affected by the anomalous condition.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 205
The Professional Thermographers Association
5.3.3 In addition, equipment may be located that is anomalous but cooler than normal as a result of
failed or inoperable components.
5.4 Mechanical failures, such as misalignment, unbalanced loads, improper lubrication, corrosion or
excessive wear of parts and components, may also have an impact on the electrical systems associated
with them.
5.5 System or component failures often result in substantially higher repair costs compared to
identifying the anomaly prior to failure and remedying the condition on scheduled downtime rather than
incurring unexpected breakdowns.
5.6 These failures can also result in increased risk of substantially greater loss of plant equipment and
production capabilities as well as putting personnel at risk.
6. Personnel Responsibilities
6.1 The Owner shall
6.1.1 Establish an inventory list of the equipment to be examined.
6.1.2 Provide a qualified escort to accompany the thermographer, for the purpose of, among other
duties:
6.1.2.1 Locating equipment and ensuring its safe examination
6.1.2.2 Opening/Closing electrical panels
6.1.2.3 Removing/replacing protective covers
6.1.2.4 Measuring electrical loads
6.1.2.5 Assisting in assessing the presence and criticality of anomalies and recommended remedial
actions
6.1.3 Meet with the thermographer and the person assigned to escort the thermographer, to review the
equipment scheduled for examination and ensure that all required safety practices have been met and
required personal protective equipment is provided.
6.1.4 Shall issue an “Energized Electrical Work Permit” in accordance with the recommendations of
NFPA 70E when working on energized electrical equipment.
6.1.5 The owner shall assume full responsibility for consequences resulting from actions taken, or not
taken, as a result of data provided by an infrared examination.
6.2 The infrared thermographer shall:
6.2.1 Possess certification of adequate knowledge and training in the practice of infrared
thermography, as defined in ASNT SNT-TC-1A, for the duties they are assigned:
6.2.1.1 Level I thermographers may perform examinations, collect data, and, as appropriate, make
evaluations based on specific, written “pass/fail” criteria.
6.2.1.2 Level II thermographers may perform examinations, collect data, analyze data and prepare
reports. Level II competency is recommended for determination of emissivity and most radiometric
temperature measurements.
6.2.2 Possess certification of adequate knowledge and training in safety practices associated with
working on energized electrical equipment (NFPA 70E), and adequate knowledge and training as required
by the owner for necessary familiarity with their facility, operations and equipment.
6.2.3 Verify the owner’s designation of the flash protection boundary and if the boundary is to be
crossed, use appropriate personal protective equipment.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 206
The Professional Thermographers Association
6.3 Unless the thermographer is qualified, the thermographer shall not:
6.3.1 Cross the restricted approach boundary
6.3.2 Remove or replace panel covers or open or close electrical cabinets containing energized
electrical components
6.3.3 Measure electrical loads
7. Procedure
7.1 Equipment to be examined shall be under adequate load; ideally this is “normal operating” load or,
when appropriate for validation testing, under “worst-case” loading.
7.2 If loading is light and likely to increase in the future, particular care should be given to documenting
any abnormalities; in these conditions, some abnormalities may be below the threshold of detection.
7.3 Equipment shall be externally examined before removing any protective covers to determine the
possible presence of internal heating. If abnormal heating is suspected, the owner or qualified escort
should take appropriate remedial action.
7.4 Enclosures of electrical equipment shall be opened for direct examination wherever practical and
safe.
7.5 As an alternative, consideration may be given to conducting the examination through “windows”
that are transparent to detected infrared radiation or to “ports.” Safety procedures should be modified to
accommodate this task. Care must be taken to insure that all equipment can be seen and special lenses
may be required.
7.6 Thermographic examinations shall be made using an infrared imaging system, producing a
thermogram for documenting, ideally, any components or systems that are not operating normally.
7.6.1 Every effort shall be made to ensure the thermal image is in sharp focus; failure to do so can
render further analysis difficult and may mean radiometric temperatures, if measured, are inaccurate.
7.6.2 Thermal images shall be stored, preferably at bit depths of 12-bit or greater, on electronic media.
7.6.3 When appropriate, corresponding visual images shall also be recorded, either with an visual
camera integral to the infrared system or on a separate visual camera
7.6.3.1 Visual images shall be properly exposed to ensure adequate detail is available
7.6.3.2 Every effort should be made to ensure that electronic visual images can be fully utilized, when
needed, during later analysis. Particular attention should be given to perspective, focus, contrast, resolution
and lighting.
7.6.3.3 Visual images shall align with the thermal image as closely as possible
7.7 Thermograms shall be recorded of all anomalies, and, when deemed necessary, of normally
operating equipment or components as well.
7.8 Spot radiometers are not recommended for locating anomalies but, when properly used, they may
sometimes be employed to measure radiometric temperatures.
7.9 All findings shall be documented by appropriate nameplate data, equipment location, a visual
photograph of the equipment and a corresponding thermogram of the anomaly.
7.9.1 The date and time of the observation shall be recorded, preferably as part of the thermal image.
7.9.2 Where possible or practical, the rated and measured electrical load associated with an observed
finding should be recorded.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 207
The Professional Thermographers Association
7.9.3 Environmental and ambient conditions associated with the finding shall be measured and
recorded. Environmental conditions include:
7.9.3.1 Correction values for surface emissivity and reflected background temperature
7.9.3.2 Ambient air temperature
7.9.3.3 Wind speed and direction, solar loading and other external radiant heat sources
7.9.3.4 Care should be taken to work within the limits of both spatial and measurement resolution for
the infrared system being used. Supplemental telephoto lenses may be required for some kinds of work,
such as out door substations or overhead lines.
7.10 A method to determine a repair priority should be agreed upon by the thermographer or system
owner or, preferably, both together. This should be used to account for any abnormal thermal situations.
Prioritization should take into account any observed thermal differences but should also consider the
impact on personnel safety, the criticality of the equipment, present and future thermal relationships, and
productive operation of the equipment and/or the facility.
7.11 Prioritization should be based first on how close the component is to failure. The thermal data
provided by the thermographer is critical to this decision but other factors must also be included. Once this
determination has been made and communicated, a decision about appropriate action to be taken should
be made by appropriate parties; this typically includes management with input from the thermographer.
7.12 An example approach to prioritizing findings for repair is to ask the following questions. If any of
them are answered in the affirmative, the finding is categorized as a Priority 1, requiring immediate action;
this might include such actions as repair, schedule for repair, reduction of temperature by increased cooling
or load reduction, or further testing to determine root cause, etc.
7.12.1 Is the phase-to-phase temperature difference greater than 24C (59F)1?
7.12.2 Is the absolute temperature greater than 100C (212F)1?
7.12.3 Is convection greater than 24kph (15mph)? 2
7.12.4 Is physical damage, such as melting or discoloration, evident?
7.12.5 Is component shiny flat or tubular bus?
7.12.6 Thermal gradients are large or moderate3
7.12.7 Are loads likely to increase by 3X or more prior to repairs?
7.12.8 Notes:
7.12.8.1 1For measurements that are reliable, i.e. high emissivity surfaces
7.12.8.2 2Convection measured at the component surface
7.12.8.3 3Massive, solid connector, indirect connections such as bus stabs, or any oil-filled or gasfilled equipment
7.13 If none of these questions are answered “yes,” component should be scheduled for further
investigation and/or more frequent thermographic inspection cycle.
8. Report
8.1 The infrared thermographer shall provide a detailed report of the thermographic examinations. This
report shall include:
8.1.1 Complete identification of the equipment examined and its location information.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 208
The Professional Thermographers Association
8.1.2 A comprehensive report for each observed anomaly, including the information collected in
Section 7.
8.1.3 When relevant, the history of any previously found anomalies associated with the particular
component or system (NFPA 70B). Trending of an anomaly over time, while useful, must be done only with
great care as many factors can cause thermal patterns to change.
8.1.4 Suggestions for repairs and corrective actions, as appropriate.
8.2 Anomalies should be re-examined following completion of corrective actions to verify that
operations are within acceptable limits.
8.3 Equipment scheduled for examination but not available shall be noted and re-scheduled for later
examination rather than being passed over until the next inspection cycle occurs.
9. Precision and Bias
9.1 The infrared thermographer shall perform a calibration check on a regular basis, depending on
needs.
9.1.1 For many circumstances daily calibration checks of some type are warranted
9.1.1.1 A simple equipment functionality check can be made by measuring the temperature of a
known, high-emissivity reference.
9.1.1.2 A more rigorous calibration check is often warranted using a calibrated, traceable black body
reference standard.
9.1.1.2.1 Records of calibration checks should be kept
9.1.1.2.2 When the infrared system fails to comply with manufacturer’s specifications for calibration,
consideration should be given to having the manufacturer perform a re-calibration. Calibration shall include
all lenses and filters being used.
9.1.1.2.3 The functionality of infrared systems being returned to service after re-calibration by the
manufacturer shall be checked prior to being used for an examination.
9.1.1.3 Annual calibration by the manufacturer may be warranted, but should not be considered as a
substitute for regular functionality checks.
10. Keywords
10.1 Electrical, infrared, thermography, heat, radiometric, non-radiometric, spot radiometer,
calibration, safety, maintenance, condition monitoring
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 209
The Professional Thermographers Association
ANNEX
(Mandatory Information)
A1.
APPENDIX
(Nonmandatory Information)
X1.
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 210
The Professional Thermographers Association
14900 Interurban Avenue South, Suite 225 Seattle, WA 98168 USA Phone: 206 328-3930 www.ProThermographer.com
Page: 211