Food and Drug Administration. 21 CFR Ch. 1. Part210
advertisement

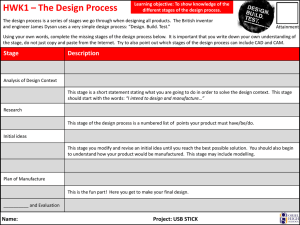
FDA 21 CFR Ch.1, Part 210 – Current Good Manufacturing Practice in Manufacturing, Processing, Packing, or Holding of Drugs; General Questions: 1. This part and parts 211 through 226 of this chapter contains what? 2. Define batch. 3. Define component. 4. Define drug product. 5. Fiber means any particulate contaminant with a length ____________ greater than its width. 6. Non-fiber-releasing filter means any filter, which after any appropriate ______ such as washing or flushing, will/will not release fibers into the component or drug product that is being filtered. All filters composed of asbestos are deemed to be fiber-releasing filters. 7. Define active ingredient. 8. What is medicated feed? 9. What is the difference between theoretical yield and actual yield? 10. What is a representative sample? Answers: 1. The regulations set forth in this part and in parts 211 through 226 of this chapter contain the minimum current good manufacturing practice for methods to be used in, and the facilities or controls to be used for, the manufacture, processing, packing, or holding of a drug to assure that such drug meets the requirements of the act as to safety, and has the identity and strength and meets the quality and purity characteristics that it purports or is represented to possess. 2. Batch means a specific quantity of a drug or other material that is intended to have uniform character and quality, within specified limits, and is produced according to a single manufacturing order during the same cycle of manufacture. 3. Component means any ingredient intended for use in the manufacture of a drug product, including those that may not appear in such drug product. 4. Drug product means a finished dosage form, for example, tablet, capsule, solution, etc., that contains an active drug ingredient generally, but not necessarily, in association with inactive ingredients. The term also includes a finished dosage form that does not contain an active ingredient but is intended to be used as a placebo. 5. Fiber means any particulate contaminant with a length at least three times greater than its width. 6. Non-fiber-releasing filter means any filter, which after any appropriate pretreatment such as washing or flushing, will not release fibers into the component or drug product that is being filtered. All filters composed of asbestos are deemed to be fiber-releasing filters. 7. Active ingredient means any component that is intended to furnish pharmacological activity or other direct effect in the diagnosis, cure, mitigation, treatment, or prevention of disease, or to affect the structure or any function of the body of man or other animals. The term includes those components that may undergo chemical change in the manufacture of the drug product and be present in the drug product in a modified form intended to furnish the specified activity or effect. 8. The term medicated feed means any Type B or Type C medicated feed as defined in § 558.3 of this chapter. The feed contains one or more drugs as defined in section 201(g) of the act. The manufacture of medicated feeds is subject to the requirements of part 225 of this chapter. 9. Theoretical yield means the quantity that would be produced at any appropriate phase of manufacture, processing, or packing of a particular drug product, based upon the quantity of components to be used, in the absence of any loss or error in actual production. Actual yield means the quantity that is actually produced at any appropriate phase of manufacture, processing, or packing of a particular drug product. 10. Representative sample means a sample that consists of a number of units that are drawn based on rational criteria such as random sampling and intended to assure that the sample accurately portrays the material being sampled.