File

Alcohol & Phenol Reactions
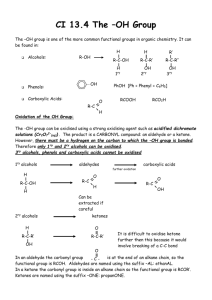
Alcohol Reactions
• 1. Dehydration - elimination of water
• water is eliminated from adjacent carbon atoms and a second bond is formed between the 2 carbon atoms
• uses a large amount of alcohol and a small amount of acid (H
2
SO
• order of reactivity:
4
) as the catalyst
– tertiary > secondary > primary
Dehydration Examples
1. Dehydrate butanol.
2. Dehydrate 2-butanol.
3. Dehydrate 2-methyl-2-butanol.
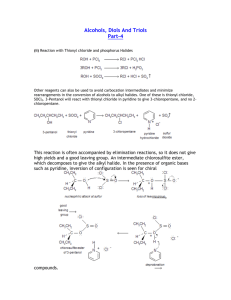
• 2. Halide Substitution alcohols react with concentrated hydrohalogens (HCl, HBr, HF) to be converted into alkyl halides
• competes with the elimination reaction
(dehydration) but this reaction is favored using a large excess of concentrated acid
(HCl and zinc chloride)
• we can distinguish between 1 ° , 2 ° and 3°
• order of reactivity:
– tertiary > secondary > primary alcohols by their different rates of reaction using the Lucas Test
Halide Substitution Examples: The Lucas Test
• 1. Primary alcohols - are not converted to alkyl chlorides at room temp; dissolve completely
• 2. Secondary alcohols – dissolves, but within 5 minutes, the alkyl chloride separates out
• 3. Tertiary alcohols – react immediately; the insoluble tertiary alkyl chloride separates out as a cloudy layer
• 3. Esterification – alcohols react with carboxylic acids to form esters and water
• esters – compounds in which the proton of an acid is replaced by an organic group
• General formula is
• Esters are named as derivatives of BOTH the carboxylic acid and the alcohol
– The 1 st word denotes the alcohol group (R) and is named like a hydrocarbon with a –yl ending
– The 2 nd word is derived from the name of the parent acid, with the “-ic acid” ending changed to an “-ate” ending.
Esterification Examples
• Ex.1 React methanol with acetic acid.
• Ex.2 React propanol with butanoic acid.
• Ex.3 React 2-propanol with butanoic acid.
•
• 4. Oxidation - the most important method of preparing aldehydes, ketones and carboxylic acids from alcohols
aldehyde ketone carboxylic acid
• Uses Jones Reagent in what is called the
Bordwell-Wellman Test
Jones Reagent water with H
2
– made by dissolving CrO
SO
4
to give chromic acid, which is orange in color
3
in
Oxidation Examples
• Primary alcohols - oxidized in a 2-step process to first give an aldehyde, and then an acid
• Ex.; Oxidize ethanol.
• Secondary alcohols - oxidized to form ketones
• Ex.; Oxidize 2-butanol.
• Tertiary alcohols - have no C-H bonds on the hydroxyl carbon, therefore they cannot undergo oxidation.
• Ex.; Oxidize tert-butanol.
Reactions of Phenols
• Phenols DO NOT undergo dehydration or substitution reactions because it is very hard to break the C-O bond when the carbon atom is part of an aromatic ring
• They will form esters because the O-H bond is breaking, not the C-O bond
• 1. Esterification - phenol reacts with an acid anhydride (two acid molecules with a water removed) to form a carboxylic acid and an ester
• Ex.; part a : form acetic anhydride part b : react phenol with acetic anhydride
• 2. Oxidative Coupling - uses potassium ferricyanide, K
3
Fe(CN)
6
, as the catalyst
• -three types of coupling: ortho-ortho, ortho-para, and para-para
• Ex. React phenol with K
3
Fe(CN)
6.
• Ex. React p-methylphenol with K
3
Fe(CN)
6.