Basic Chemistry XII
advertisement

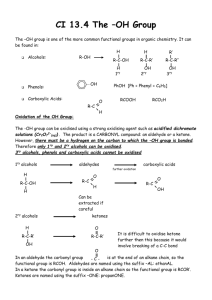
Functional derivatives of carboxylic acids = functional carboxyl group is changed 2.1. SALTS Basic Chemistry XII Vladimíra Kvasnicová R―COO- M+ (M+ = metal cation) = products of neutralization (acid + base → salt + water) • suffix: -ate or -oate -ic acid → -ate / -oic acid → -oate • R―COO- = carboxylate (anion of c.a.) • full name: cation carboxylate (sodium acetate) 2.2. ESTERS R1―O―CO―R2 = products of esterification (acid + alcohol → ester + water) Examples of esters • ethyl formate = ethyl methanoate • the opposite reaction = ester hydrolysis • methyl butyrate • suffix: -ate some esters have a fruity smell: • R1―O― = rest of alcohol • CH3-CH2-CH2-CO-O-CH3 (apple) • R1 = alkyl (from the alcohol name: „alkyl“ alkohol) • CH3-CH2-CH2-CO-O-(CH2)4-CH3 (apricot) • full name: alkyl carboxylate • CH3-CO-O-(CH2)4-CH3 (banana) (methyl acetate = methyl ethanoate) • CH3-CH2-CH2-CO-O-CH2-CH3 (pineapple) • H-CO-O-CH2-CH3 (rum smell) Properties and reactions of esters • lower boiling points than carboxylic acids and alcohols (~ absence of hydrogen bonds) • less soluble in water than carboxylic acids • esters have a fruity smell 2.3. ANHYDRIDES R1―CO―O―CO―R2 • acid → anhydride (acetic acid → acetic anhydride) • organic, organic-inorganic • reactions: ester hydrolysis examples saponification = hydrolysis by a strong base → salt and alcohol → salts of long chain fatty acids are SOAPS • acetic formic anhydride polymerization → polyesters (from difunctional monomers) • „phosphoglycerate“ (= phosphoric glyceric anhydride) 2.4. AMIDES R―CO―NH2 • suffix: -amide (ethanamide) • -ic or –oic acid → -amide (acetamide) • substituted –NH2 group: N-alkyl…amide • glutaric anhydride 2.5. ACYLHALIDES R―CO―X X = halogen • acyl name + halide • e.g. acetyl chloride (= ethanoyl chloride) butyryl bromide (= butanoyl bromide) • substituted amide groups are found in proteins (the peptide bond = „amide bond“) • nitrogen atom contains an unshared pair of electrons → delocalization ⇒ amides are not basic • strong intermolecular H-bonds: amides are solids • low MW amides are soluble in water • carboxylic acid + ammonia (or amine) → amide + water 2.6. NITRILES R―C≡ ≡N • hydrocarbon + suffix: -nitrile or alkyl cyanide CH3CN • e.g ethanenitrile or methyl cyanide butanenitrile or propyl cyanide • mostly toxic liquids CH3CH2CH2CN Exercise • • • • • • • • • • • Exercise H2N-CH2-CH2-CH2-COOH CH3-CO-CH2-COOH CH3-CH(OH)-CH2-COOH CH3-CH2-CH2-CO-O-CH3 CH3-CO-COOH CH3-CH2-O-CH3 CH3-CO-CH2-CH3 CH3-(CH2)14-COOH CH3-CH2-CH2-COONa (CH3-CH2-COO)2Ca CH3-CH2-O-CO-H • • • • • • • • • • • Carbonyl functional group: H2N-CH2-CH2-CH2-COOH CH3-CO-CH2-COOH CH3-CH(OH)-CH2-COOH CH3-CH2-CH2-CO-O-CH3 CH3-CO-COOH CH3-CH2-O-CH3 CH3-CO-CH2-CH3 CH3-(CH2)14-COOH CH3-CH2-CH2-COONa (CH3-CH2-COO)2Ca CH3-CH2-O-CO-H >C=O • • • • • • • • • • • γ-aminobutyric acid acetoacetic acid β-hydroxybutyric acid methyl butyrate pyruvic acid ethyl methyl ether ethyl methyl ketone palmitic acid sodium butyrate calcium propionate ethyl formate Carbonyl compounds • carbonyl- = oxo- group • names: systematic or common (trivial) • polar, planar, 2 nonbonded pairs of electrons (:O:) • aldehydes: substitution principle or name derived from common name of a corresponding acid • it is found in aldehydes and ketones • ALDEHYDES: terminal carbon; -CHO • in addition to a cyclic chain: • prefix: formyl- -al -carbaldehyde (1,2,3-propane tricarbaldehyde) • KETONES: middle carbon; -CO• prefix: oxo- or keto- - one • ketones: substitution principle / radical function principle or common names • properties: formation of H-bonds with water → solubility of low MW carbonyl compounds in water (C1-C4) sharp and irritant odour (↓ MW) – toxic comp., alergy; or pleasant smell (↑ MW) Carbonyl compounds • important aldehydes: Carbonyl compounds - REACTIONS 1. oxidation methanal = formaldehyde • prim. alcohol → aldehyde → carboxylic acid ethanal = acetaldehyde • sec. alcohol → ketone → no more oxidized 2,3-dihydroxypropanal = glyceraldehyde Tollens´ reagent: [Ag(NH3)2]+ → Ag benzaldehyde (bitter almond odour, little toxic) Benedict´s or Fehling´s solution: Cu2+(l) → Cu2O (s) • important ketones: 2. reduction propanone = dimethyl ketone = acetone • aldehyde → primary alcohol 1,3-dihydroxypropanone = dihydroxyacetone • ketone → secondary alcohol ketones are more narcotic than aldehydes Alcohols • contain the hydroxyl –OH functional group Alcohols - properties • primary / secondary / tertiary • low MW alcohols: colourless liquids, specific odour (unpleasant from C4), narcotic effect, toxic • monofunctional / polyfunctional • polyfunctional alcohols: sweet taste • naming: -ol (or alkyl alcohol) -diol (or alkyl glycol) –triol,... • higher alcohols (from C12): solid compounds • -OH attached directly to the benzene ring → phenols (= group name) If the hydroxyl group is not the principle one: hydroxy- • H-bonds → solubility in water, higher boiling points than alkanes • structure: polar functional group nonpolar hydrocarbon chain hydrophobic properties increase with MW Alcohols – important examples • methanol = methyl alcohol • ethanol = ethyl alcohol Alcohols - toxicity Ethylene glycol • toxic: 50 mL, lethal: 100 mL • ethane-1,2-diol = ethylene glycol Methanol • propane-1,2,3-triol = glycerol • 5-10 mL toxic, 30 mL lethal • cyclohexanol, inositols • cholesterol ! add structure formulas ! • loss of eyesight, metabolic acidosis Ethanol • lethal: 6-8 g/kg (∼ 1 L of vodka) • degradation: oxidation of 0,15 g/kg/hour 0,5 L of beer (4%) ∼ 20 mL of ethanol = 16 g 70 kg man: 0,7 x 70 = 49 kg (L) water i.e. 16 g etOH / 49 L = 0,33 g / L = 0,33 ‰ Color Atlas of Biochemistry / J. Koolman, K.H.Röhm. Thieme 1996. ISBN 0-86577-584-2 29,4 kJ/g of ethanol Obrázek převzat z: Color Atlas of Biochemistry / J. Koolman, K.H.Röhm. Thieme 1996. ISBN 0-86577-584-2