How Matter Emits Light: 2. Line Emission and Spectroscopy
advertisement

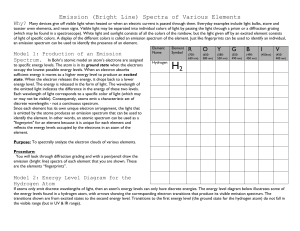
How Matter Emits Light: 2. Line Emission and Spectroscopy Announcements q Homework # 3 is due today, Oct 20th. q Homework # 4 starts today, Thursday October 20th, and is due Thursday October 27th Assigned Reading q Units 24 and 25 Today’s Goals q Discuss and understand the other mechanism, other than Blackbody, through which matter can emit light q Familiarize ourselves with the concept of atomic spectra How do objects make light? n There are two principal mechanisms for producing electromagnetic radiation • Blackbody radiation • Spectral line emission of atoms and molecules Both of these mechanisms result from accelerating/decelerating electrons! I.e., you accelerate an electric charge to create EM radiation How Atoms Emit Light: absorption § The electrons in the atoms first need to absorb energy, either from light (photons) or from the collisions of atoms with each other. § Then the electrons can re-emit the absorbed emission energy. § I.e., they accelerate or decelerate in their orbits Excitation of Atoms (The atoms do not produce light, yet) v To change its energy levels, an electron must either absorb or emit a photon that has the same amount of energy as the difference between the energy levels v E1-E4 = Eph = hν = h c/λ v Larger energy difference means higher frequency. v Different jumps in energy levels means different frequencies of light absorbed. What Happens after the Excitation: absorption emission Now they produce light § The excited electrons will tend to go back to their ground-state (state of minimal energy) § They may choose a path that causes multiple `jumps’ through energy levels of decreasing energy § Each `jump’ produces a photon with energy: E4-E3 = Eph = hν = h c/λ § When there are many identical atoms, light with the same wavelength (single color or line) will be produced Do we see this effect? Spectral Line Emission (photon absorption) If a photon of exactly the right energy is absorbed by an electron in an atom, the electron will gain the energy of the photon and jump to an outer, higher energy orbit. A photon of the same energy is emitted when the electron falls back down to its original orbit. Spectral Line Emission (collision) Collisions (like in a hot gas) can also provide electrons with enough energy to change energy levels. A photon of the same energy is emitted when the electron falls back down to its original orbit. Energy Levels in an Atom or Molecule are Unique! n n The allowed electron energies are specific to the kind of atom (element) or molecule. The amount of energy in the electron’s orbit is related to the average distance of the electron from the nucleus. If we add enough energy, the electron escapes all together from the atom. The atom is then ionized. Also the ionization energy is specific to the atom or molecule. Spectral Lines of Some Elements Argon Helium Mercury Sodium Neon Spectral lines are like a cosmic barcode system for elements. Atoms of different elements have unique spectral lines because each element n has atoms of a unique color n has a unique set of neutrons n has a unique set of electron orbits n has unique photons Light bulb = Blackbody radiation The Solar Spectrum There are similar absorption lines in the other regions of the electromagnetic spectrum. Each line exactly corresponds to chemical elements in the stars. The Sun (a star) is as a blackbody, and the gas in its atmosphere (around it) is made of atoms that absorb its light, and produce dark lines in its spectrum Energy Levels of a Hydrogen Atom Different allowed “orbits” or energy levels in a hydrogen atom. Emission line spectrum Absorption line spectrum Emission nebula Reflection nebula Decomposing an Object’s Spectral Features Visible Spectrum of the Planet Mars As seen through a prism Encoded in an object’s spectrum is information about the emitter/ absorber. This is how we learn what the Universe is made of! Spectra of Galaxies Name that Spectrum! A C Absorption Emission B Continuous Survey Question A gas cloud that has become completely ionized 1) 2) 3) 4) does not have spectral line absorption does not have spectral line emission emits only x-rays emits only radio waves Survey Question The number of electrons lost by an atom in a gas (that is, it’s ionization state) depends primarily on 1) 2) 3) 4) the the the the velocity of the gas temperature of the gas level of the ground state size of the gas cloud The Doppler Effect: other information contained in spectrum n n A moving light or sound source emits a different frequency in the forward direction than in the reverse direction. Think of the approaching (higher pitch) and then receding (lower pitch) police car to figure out how this works. In general … n n n The “native” frequency at which an object is emitting is called the rest frequency. You will see/hear frequencies higher than the rest frequency from objects moving towards you. You will see/hear frequencies lower than the rest frequency from objects moving away from you. Doppler Shift The first crest travels out in circle from the original position of the plane Shorter wavelength (bluer) At a later time, a second crest is emitted from the planes new position, but the old crest keeps moving out in a circle from the planes original position The same thing happens again at a later time Longer wavelength (redder) What we actually see when we do spectroscopy Emission spectrum of hot gas as seen in lab Emission spectrum of hot gas as seen in rapidly moving object Is this object moving towards or away from us? What we do with the Doppler Shift We derive the (approaching or receding) velocity of the source. q The Doppler shift causes the rest wavelength, λr , emitted by an object to become bluer (blue-shift) or redder (red-shift): λo- λr q To derive a velocity, we divide by a time T= 1/νr: v = (λo- λr)/T = (λo- λr) νr = c (λo- λr)/ λr v/c = (λo- λr)/ λr This is the formula used to measured the recession of galaxies in the Universe! The Doppler shift n An object shining red light with λ=656.3 nm is moving at V=5,000,000 m/s away from you. What is the color of the light that you see? n n v/c = (λo- λr)/λr v/c =5x106/3x108 = 1.67x10-2 = (λo- λr)/λr λo = λrx(1+1.67x10-2) = 667.3 nm Survey Question Two identical stars are observed from the Earth. Star A’s emission lines (that are at visible wavelengths in the rest frame) are observed to be at ultraviolet wavelengths. The same emission lines for Star B are observed to be at X-ray wavelengths. From these observations you conclude that: 1) both stars are moving away from the Earth 2) Star A is moving towards the Earth faster than Star B 3) Star B is moving towards the Earth faster than Star A 4) Star B is moving away from the Earth while Star A is moving towards the Earth.