Intermolecular Forces and Liquids and Solids
advertisement

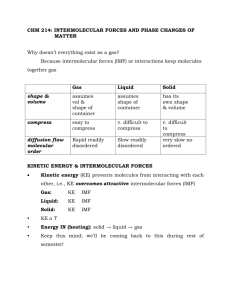
Intermolecular Forces and Liquids and Solids Chapter 11 Copyright © The McGraw­Hill Companies, Inc. Permission required for 1 A phase is a homogeneous part of the system in contact with other parts of the system but separated from them by a well­defined boundary. 2 Phases Solid phase ­ ice Liquid phase ­ water 11.1 2 Intermolecular Forces Intermolecular forces are attractive forces between molecules. Intramolecular forces hold atoms together in a molecule. Intermolecular vs Intramolecular • 41 kJ to vaporize 1 mole of water (inter) • 930 kJ to break all O­H bonds in 1 mole of water (intra) “Measure” of intermolecular force Generally, boiling point intermolecular melting point forces are much ∆Hvap weaker than ∆Hfus intramolecular ∆Hsub forces. 11.2 3 Intermolecular Forces Dipole­Dipole Forces Attractive forces between polar molecules Orientation of Polar Molecules in a Solid Nonpolar molecules do not have dipole dipole forces. 11.2 4 Intermolecular Forces Ion­Dipole Forces Attractive forces between an ion and a polar molecule Pretty strong attractive force. This is the attractive between water and ionic solute. Ion­Dipole Interaction 11.2 5 Intermolecular Forces Dispersion Forces Attractive forces that arise as a result of temporary dipoles induced in atoms or molecules Present in all molecules, but the only intermolecular force in NONPOLAR MOLECULES, or in Gaseous atoms. ion­induced dipole interaction dipole­induced dipole interaction 11.2 He He the temporary dipole induces a temporary dipole in a neighboring He atom both electrons on the same side atom set up a TEMPORARY DIPOLE. 6 Intermolecular Forces Dispersion Forces Continued Polarizability is the ease with which the electron distribution in the atom or molecule can be distorted. Polarizability increases with: • greater number of electrons • more diffuse electron cloud Dispersion forces usually increase with molar mass. 11.2 7 What type(s) of intermolecular forces exist between each of the following molecules? HBr HBr is a polar molecule: dipole­dipole forces. There are also dispersion forces between HBr molecules. CH4 CH4 is nonpolar: dispersion forces. SO2 O S O SO2 is a polar molecule: dipole­dipole forces. There are also dispersion forces between SO2 molecules. 11.2 8 Intermolecular Forces Hydrogen Bond The hydrogen bond is a special dipole­dipole interaction between they hydrogen atom in a polar N­H, O­H, or F­H bond and an electronegative O, N, or F atom. or A H …B A H …A A & B are N, O, or F 11.2 9 Why is the hydrogen bond considered a “special” dipole­dipole interaction? Decreasing molar mass Decreasing boiling point 11.2 10 11 Rank the following in order of increasing boiling point: HBr CH3OH CH2O NaCl CH3CH3 He CH4 12 Properties of Liquids Surface tension is the amount of energy required to stretch or increase the surface of a liquid by a unit area. Strong intermolecular forces High surface tension 11.3 13 Properties of Liquids Cohesion is the intermolecular attraction between like molecules Adhesion is an attraction between unlike molecules Adhesion Cohesion 11.3 14 Properties of Liquids Viscosity is a measure of a fluid’s resistance to flow. Strong intermolecular forces High viscosity 11.3 15 Water is a Unique Substance Maximum Density 40C Density of Water Ice is less dense than water 11.3 16 A crystalline solid possesses rigid and long­range order. In a crystalline solid, atoms, molecules or ions occupy specific (predictable) positions. An amorphous solid does not possess a well­defined arrangement and long­range molecular order. A unit cell is the basic repeating structural unit of a crystalline solid. At lattice points: • Atoms • Molecules • Ions lattice point Unit Cell Unit cells in 3 dimensions 11.4 17 11.4 18 11.4 19 11.4 20 11.4 21 Shared by 8 unit cells Shared by 2 unit cells 11.4 22 1 atom/unit cell (8 x 1/8 = 1) 2 atoms/unit cell (8 x 1/8 + 1 = 2) 4 atoms/unit cell (8 x 1/8 + 6 x 1/2 = 4) 11.4 23 11.4 24 When silver crystallizes, it forms face­centered cubic cells. The unit cell edge length is 409 pm. Calculate the density of silver. m V = a3 = (409 pm)3 = 6.83 x 10­23 cm3 d = V 4 atoms/unit cell in a face­centered cubic cell m = 4 Ag atomsx 1 mole Ag 107.9 g x = 7.17 x 10­22 g 23 mole Ag 6.022 x 10 atoms 7.17 x 10­22 g m = d = = 10.5 g/cm3 ­23 3 V 6.83 x 10 cm 11.4 25 11.5 26 Extra distance = BC + CD =2d sinθ = nλ (Bragg Equation) 11.5 27 X rays of wavelength 0.154 nm are diffracted from a crystal at an angle of 14.170. Assuming that n = 1, what is the distance (in pm) between layers in the crystal? nλ = 2d sin θ d = n = 1 θ = 14.170 λ = 0.154 nm = 154 pm nλ 1 x 154 pm = 77.0 pm 2 x sin14.17 2sinθ = 11.5 28 Types of Crystals Ionic Crystals • Lattice points occupied by cations and anions • Held together by electrostatic attraction • Hard, brittle, high melting point • Poor conductor of heat and electricity CsCl ZnS CaF2 11.6 29 Types of Crystals Covalent Crystals • Lattice points occupied by atoms • Held together by covalent bonds • Hard, high melting point • Poor conductor of heat and electricity carbon atoms diamond graphite 11.6 30 Types of Crystals Molecular Crystals • Lattice points occupied by molecules • Held together by intermolecular forces • Soft, low melting point • Poor conductor of heat and electricity 11.6 31 Types of Crystals Metallic Crystals • Lattice points occupied by metal atoms • Held together by metallic bonds • Soft to hard, low to high melting point • Good conductors of heat and electricity Cross Section of a Metallic Crystal nucleus & inner shell e­ mobile “sea” of e­ 11.6 A metallic bond is the attractive force between the nucleus of the metal atoms and the "sea" of mobile electrons. 32 Types of Crystals 11.6 33 An amorphous solid does not possess a well­defined arrangement and long­range molecular order. A glass is an optically transparent fusion product of inorganic materials that has cooled to a rigid state without crystallizing Crystalline quartz (SiO2) Non­crystalline quartz glass 11.7 34 Least Order Condensation Evaporation T2 > T1 Greatest Order 11.8 35 The equilibrium vapor pressure is the vapor pressure measured when a dynamic equilibrium exists between condensation and evaporation H2O (l) H2O (g) Dynamic Equilibrium Rate of = condensation Rate of evaporation 11.8 36 Before Evaporation At Equilibrium 11.8 37 Molar heat of vaporization (∆Hvap) is the energy required to vaporize 1 mole of a liquid. Clausius­Clapeyron Equation ∆Hvap ln P = ­ + C RT P = (equilibrium) vapor pressure T = temperature (K) R = gas constant (8.314 J/K•mol) 11.8 38 The boiling point is the temperature at which the (equilibrium) vapor pressure of a liquid is equal to the external pressure. The normal boiling point is the temperature at which a liquid boils when the external pressure is 1 atm. 11.8 39 The critical temperature (Tc) is the temperature above which the gas cannot be made to liquefy, no matter how great the applied pressure. The critical pressure (Pc) is the minimum pressure that must be applied to bring about liquefaction at the critical temperature. 11.8 40 H2O (s) H2O (l) The melting point of a solid or the freezing point of a liquid is the temperature at which the solid and liquid phases coexist in equilibrium Freezing Melting 11.8 41 Molar heat of fusion (∆Hfus) is the energy required to melt 1 mole of a solid substance. 11.8 42 11.8 43 H2O (s) H2O (g) Molar heat of sublimation (∆Hsub) is the energy required to Sublimation sublime 1 mole of a solid. Deposition ∆Hsub = ∆Hfus + ∆Hvap ( Hess’s Law) 11.8 44 A phase diagram summarizes the conditions at which a substance exists as a solid, liquid, or gas. Phase Diagram of Water 11.8 45 11.8 46 47