Forensics Exam Prep: Chromatography, Fingerprints, DNA & More
advertisement

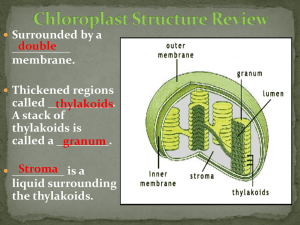
Rf values help? Rf = (dist. moved by solute)/(dist moved by solvent). How do you know how to identify the components of mixtures by means of their Rf values? please drop down some answres thx for read ing and answering my qustiion thank you once again Rf values pertain to certain components of a mixture that you want to separate by chromatography. Let's say you put a drop of a mixture A onto a paper chromatography sheet and dip it into the separating mixture. As the mixture moves up the paper, different components of A have different affinities to the paper and the solution. Lets say component 1 has more affinity to the paper than component 2. Therefore Component 2 is going to move further up the paper in the same space of time as componenet 1. Let's say that component 1 moves a distance "C1" and component 2 moves a distance "C2". The separating mixture moves a distance S which is (obviously) greater than both C1 and C2 because it's going to move those components. The Rf value for component 1 is C1/S and the Rf for component 2 is C2/S. These values are constant for each component in a given separating mixture, pressure, T and chromatography paper type and are given in literature. By comparing the Rf values that you get for unknown components you have and the Rf values in literature (under the same conditions), you can find what your unknown components are. http://answers.yahoo.com/question/index?qid=20070922133354AANUoRB Re: Where can I find tables of Rf values? Date: Mon Jul 10 17:09:29 2000 Posted By: Dan Berger, Faculty Chemistry/Science, Bluffton College Area of science: Chemistry ID: 963260691.Ch Message: There is no such thing as a table of Rf values. Chromatographic retention times are sensitively and unpredictably dependent on the conditions; if you run chromatography on the same sample three consecutive times (using the "same conditions") you will get different retention values (Rf or whatever) for the same substance each time! That's not to say that the values will be wildly different; in sensitive techniques such as gas chromatography you get almost the same values for the same substance on consecutive runs. But with TLC the only way to unambiguously identify something is to run an authentic sample on the same plate as the substance to be identified. Not "another plate, right afterwards." The same plate. That's why you will never find a table of Rf values. Or of GC retention times, for that matter. Dan Berger Bluffton College cs.bluffton.edu/~berger http://www.madsci.org/posts/archives/2000-07/963425409.Ch.r.html Christina03-23-2006, 04:40 PM Would anyone happen to know where I could find the standard Rf values (for Chlorophyll a, Chlorophyll b, Xanthophyll, and Carotene) if varsol is the solvent? I'm trying to identify those plant pigments via Paper Chromatography, and I'd like to compare my results to the standards. The problem is, I've been looking and can't find it. ankankakka 03-30-2007, 07:21 AM This is what you can use: "Rf values will be unique for each solvent. However, the general order of the Rf values should be the same because the more nonpolar pigments move farther in nonpolar organic solvents. A recent plant physiology manual (Reiss 1994) identifies six pigments from spinach leaves extracted with hexane and chromatographed with petroleum ether-acetonechloroform (3:1:1) on silica-gl chromatography. The pigments and their Rf's were: carotene - 0.98 chlorophyll a - 0.59 chlorophyll b - 0.42 pheophytin - 0.81 xanthophyll 1 - 0.28 xanthophyll 2 - 0.15 The color of the bands can be a general guide to identify the pigments. Carotene is orange. Chorophylls are green. Chlorophyll a is a blue-green. Chlorophyll b is a yellowgreen. Xanthophylls are yellow. Phaeophytin is chlorophyll lacking the central magnesium ion. Pheophytin is an olive-green." Source: http://madsci.org/posts/archives/2001-12/1008377272.Bt.r.html (Original source cited: Reiss, Carol 1994. Experiments in Plant Physiology. Englewood Cliffs, NJ: Prentice Hall.) http://physicsforums.com/archive/index.php/t-115321.html