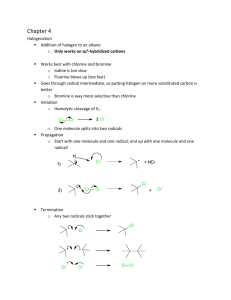
Mass, Moles, Particles, Volume, Gas Laws, Avogadro`s Principle
advertisement

Kinetics and Equilibrium Review Questions 1. Increasing the concentration of the N2 in the reaction N2+3H2 2NH3 _____ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 2. Increasing the temperature of the reaction between N2+3H2 2NH3 _____ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 3. Increasing the surface of the Pt added to the N2+3H2 2NH3 in the first reaction mechanism ___ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 4. What is the sign of the entropy change of the forward reaction between N2(g)+3H2(g) 2NH3(g) a) S = ( + ) because in the forward reaction two different molecules combine to form 1 molecule b) S = ( - ) because in the forward reaction two different molecules combine to form 1 molecule c) S = ( + ) because in the forward reaction four gas molecules combine to form 2 gas molecules d) S = ( - ) because in the forward reaction four gas molecules combine to form 2 gas molecules 5. Since this reaction is reversible the forward reaction should _____ a) release heat and therefore is exothermic creating greater disorder in the surroundings N2(g)+3H2(g) 2NH3(g) + Heat b) release heat and therefore is endothermic creating greater order in the surroundings N2(g)+3H2(g) 2NH3(g) + Heat c) absorb heat and therefore is exothermic creating greater disorder in the surroundings Heat + N2(g)+3H2(g) 2NH3(g) d) absorb heat and therefore is endothermic creating less disorder in the surroundings Heat + N2(g)+3H2(g) 2NH3(g) 6. Which of the following is true given the following reaction that is in equilibrium? ____ N2(g)+3H2(g) 2NH3(g) I. the concentration of the N2, H2,NH3 are constant II. the concentration of the N2 + H2 equal the concentration of the NH3 III. the rate of formation of NH3 equals the rate of decomposition of NH3 a) I only b) II only c) II and III d) I and III 7. Given N2(g)+3H2(g) 2NH3(g) what is the Keq expression: a) Keq = [ N]2 [ H]2 [NH]3 c) Keq = [ NH]3 [ N]2 [ H]3 b) Keq = [ N2] [ H2]3 [NH3]2 d) Keq = [ NH3]2 [ N2] [ H2]3 e) I, II, III only 8. If the value of Keq is 1.8 x 10-5 for this equilibrium at 101.3 KPa and 273 K then ____ a) the equilibrium consists mostly of reactants b) the equilibrium consists of equal amounts of reactants and products c) the equilibrium consists mostly of products d) the equilibrium consists only of reactants because it is such a small value 9. Based on your answer to questions 12 and 13 which of the following are true if the temperature is increased? 4pts ___ ____ ____ ____ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same 10. Which of the following are true if there is an increase in the concentration of the N2? 4pts__ __ __ __ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same 11. Which of the following are true if there is a decrease in the concentration of the NH3?4pts_ __ __ __ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same 12. Which of the following is true if there is an increase in pressure on the system? 4pts__ __ __ __ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same Kinetics and Equilibrium Questions 1. Increasing the concentration of the N2 in the reaction N2+3H2 2NH3 _____ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 2. Increasing the temperature of the reaction between N2+3H2 2NH3 _____ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 3. Increasing the surface of the Pt added to the N2+3H2 2NH3 in the first reaction mechanism ___ a) increases the rate of the reaction because more collisions occur per unit of time b) increases the rate because the collisions are more effective c) increases the rate because the collisions are more frequent and more effective d) will not affect the rate of reaction. 4. What is the sign of the entropy change of the forward reaction between N2(g)+3H2(g) 2NH3(g) a) S = ( + ) because in the forward reaction two different molecules combine to form 1 molecule b) S = ( - ) because in the forward reaction two different molecules combine to form 1 molecule c) S = ( + ) because in the forward reaction four gas molecules combine to form 2 gas molecules d) S = ( - ) because in the forward reaction four gas molecules combine to form 2 gas molecules 5. Since this reaction is reversible the forward reaction should _____ a) release heat and therefore is exothermic creating greater disorder in the surroundings N2(g)+3H2(g) 2NH3(g) + Heat b) release heat and therefore is endothermic creating greater order in the surroundings N2(g)+3H2(g) 2NH3(g) + Heat c) absorb heat and therefore is exothermic creating greater disorder in the surroundings Heat + N2(g)+3H2(g) 2NH3(g) d) absorb heat and therefore is endothermic creating less disorder in the surroundings Heat + N2(g)+3H2(g) 2NH3(g) 6. Which of the following is true given the following reaction that is in equilibrium? ____ N2(g)+3H2(g) 2NH3(g) I. the concentration of the N2, H2,NH3 are constant II. the concentration of the N2 + H2 equal the concentration of the NH3 III. the rate of formation of NH3 equals the rate of decomposition of NH3 a) I only b) II only c) II and III d) I and III e) I, II, III only 7. Given N2(g)+3H2(g) 2NH3(g) what is the Keq expression: a) Keq = [ N]2 [ H]2 [NH]3 c) Keq = [ NH]3 [ N]2 [ H]3 b) Keq = [ N2] [ H2]3 [NH3]2 d) Keq = [ NH3]2 [ N2] [ H2]3 8. If the value of Keq is 1.8 x 10-5 for this equilibrium at 101.3 KPa and 273 K then ____ a) the equilibrium consists mostly of reactants b) the equilibrium consists of equal amounts of reactants and products c) the equilibrium consists mostly of products d) the equilibrium consists only of reactants because it is such a small value 9. Based on your answer to questions 12 and 13 which of the following are true if the temperature is increased? 4pts ___ ____ ____ ____ N2(g)+3H2(g) 2NH3(g) + heat a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same 10. Which of the following are true if there is an increase in the concentration of the N2? 4pts__ __ __ __ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same 11. Which of the following are true if there is a decrease in the concentration of the NH3?4pts_ __ __ __ N2(g)+3H2(g) 2NH3(g) a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same Only changes in temperature or potentially pressure change Keq values 12. Which of the following is true if there is an increase in pressure on the system? 4pts__ __ __ __ 4 N2(g)+3H2(g) 2NH3(g) to 2 An increase in pressure of a system containing gas molecules favors the production of fewer gas molecules a) shifts to the right b) shifts to the left c) the concentration of the H2 increases d) the concentration of the H2 decreases e) the concentration of the NH3 increases f) the concentration of the NH3 decreases g) the value of Keq will increase h) the value of Keq will decrease i) Keq will remain the same