Lab Report for Experiment 12H Part C
advertisement

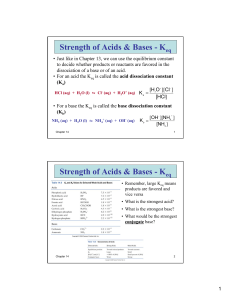
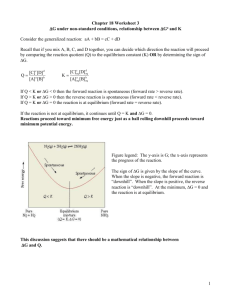
Names: _________________________ ____________________________ Experiment 12H Part C (Due Thursday 3-24) This document includes everything you need for the lab report. Show your work clearly and put the correct unit on EVERY number! READ THE PROCEDURE IN THE LAB TO FAMILIARIZE YOURSELF WITH THIS EXPERIMENT! In this experiment, you measure the equilibrium constant for the following reaction at various temperatures. Fe3+ (aq) + SCN− (aq) = FeSCN2+ (aq) A plot of ln Keq vs. 1/T allows you to determine Ho, So, and Go for the reaction. The reactants have no color and the product is red so the amount of product present at equilibrium is measured by reading the absorbance at 460 nm. Data for this experiment is given below. DATA: = 4925 M-1cm-1 l = 1.00 cm [Fe3+]init = 1.00 x 10-2 M [SCN−]init = 1.00 x 10-4 M Corrected T (oC) 5.9 8.9 12.8 15.9 19.6 22.1 A 0.309 0.305 0.300 0.296 0.292 0.288 [FeSCN2+]eq Keq T (K) 1/T (K-1) ln Keq The temperature was corrected based on the measured temperature of ice-water. Using Beer’s law (A = cl), you can calculate the equilibrium concentration of the product. (The values for and l are given above.) From the given initial concentrations of reactants and the measured equilibrium concentration of the product, you can set up an ICE table and calculate the equilibrium constant at each temperature. 1 1. Reproduce and complete the table on the previous page in Excel. Use Excel formulas to do all of the calculations except for Keq. Use ICE tables to calculate Keq. Show your calculation of [FeSCN2+]eq , the ICE table, and your calculation of Keq for 5.9oC below. 2. Based on how the equilibrium constant changes with temperature, determine whether the reaction is endothermic or exothermic. Explain your answer. 3. Based on the nature of the reaction, does your answer to question 2 make sense? Explain. (Consider bond breaking and bond forming.) 2 4. Use Excel to make a graph of ln Keq vs. 1/T. Add a trend line and show the equation and R2 value. 5. Use the equation of the line to calculate Ho and So (see ws18.3). Does the sign of Ho match your answer to question 2? 6. Calculate Go at 25oC. (See ws18.2. Assume that Ho and So are independent of temperature.) What does the sign of Go tell you about this reaction? 7. Calculate the value of the equilibrium constant at 25oC (see ws18.3). Does your answer make sense based on your measured values of Keq at lower temperatures? Does your answer make sense based on the sign of Go? 3 8. You can also calculate the equilibrium constant at 25oC using the equation of the line. Do this and compare your answer to your answer to question 7. 9. Based on the balanced equation for the reaction, predict the sign of So. Explain your answer. 10. If this was a gas phase reaction, your prediction in question 9 would have been correct but it turns out that this reaction actually has the opposite sign of So. (If you got the same sign as your prediction, you’ve done something wrong!) Can you figure out why this is? [Hint: Remember that all of the ions are hydrated (surrounded by water). What must be happening to the water?] 11. You have determined the value of G at 25oC when Q = 1 (Go). Calculate G at 25oC when Q = 200.0 (see ws18.3). Does the sign of G make sense based on your calculated value of Keq at 25oC? 4








![2 [B]](http://s2.studylib.net/store/data/009861274_1-797e0e32cb4f69343088b2a992994675-300x300.png)
