HONORS: UNIT 2B For each unit you are to complete Vocabulary
advertisement
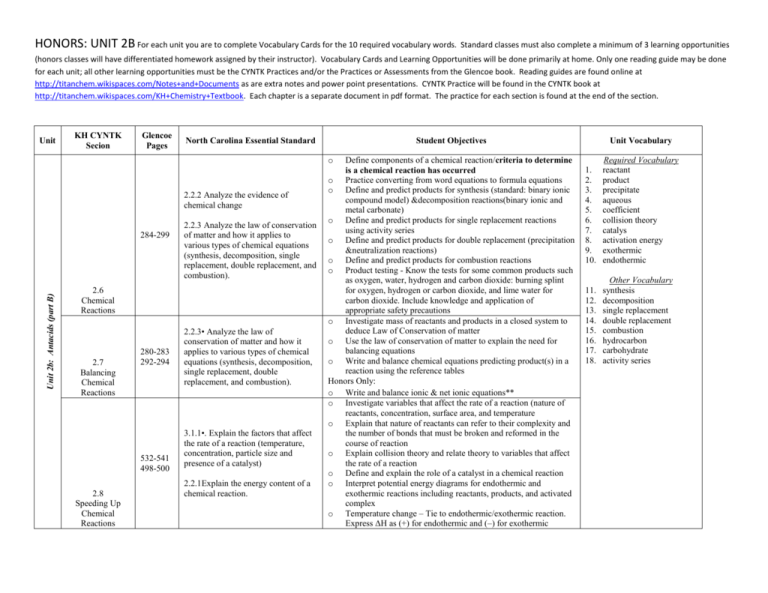
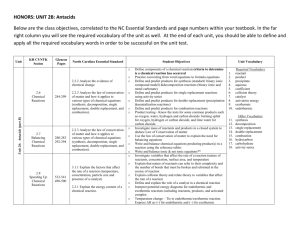
HONORS: UNIT 2B For each unit you are to complete Vocabulary Cards for the 10 required vocabulary words. Standard classes must also complete a minimum of 3 learning opportunities (honors classes will have differentiated homework assigned by their instructor). Vocabulary Cards and Learning Opportunities will be done primarily at home. Only one reading guide may be done for each unit; all other learning opportunities must be the CYNTK Practices and/or the Practices or Assessments from the Glencoe book. Reading guides are found online at http://titanchem.wikispaces.com/Notes+and+Documents as are extra notes and power point presentations. CYNTK Practice will be found in the CYNTK book at http://titanchem.wikispaces.com/KH+Chemistry+Textbook. Each chapter is a separate document in pdf format. The practice for each section is found at the end of the section. Unit KH CYNTK Secion Glencoe Pages North Carolina Essential Standard Student Objectives Unit Vocabulary o 2.2.2 Analyze the evidence of chemical change Unit 2b: Antacids (part B) 284-299 2.2.3 Analyze the law of conservation of matter and how it applies to various types of chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion). 2.6 Chemical Reactions 2.7 Balancing Chemical Reactions 280-283 292-294 532-541 498-500 2.8 Speeding Up Chemical Reactions 2.2.3• Analyze the law of conservation of matter and how it applies to various types of chemical equations (synthesis, decomposition, single replacement, double replacement, and combustion). 3.1.1•. Explain the factors that affect the rate of a reaction (temperature, concentration, particle size and presence of a catalyst) 2.2.1Explain the energy content of a chemical reaction. Define components of a chemical reaction/criteria to determine is a chemical reaction has occurred o Practice converting from word equations to formula equations o Define and predict products for synthesis (standard: binary ionic compound model) &decomposition reactions(binary ionic and metal carbonate) o Define and predict products for single replacement reactions using activity series o Define and predict products for double replacement (precipitation &neutralization reactions) o Define and predict products for combustion reactions o Product testing - Know the tests for some common products such as oxygen, water, hydrogen and carbon dioxide: burning splint for oxygen, hydrogen or carbon dioxide, and lime water for carbon dioxide. Include knowledge and application of appropriate safety precautions o Investigate mass of reactants and products in a closed system to deduce Law of Conservation of matter o Use the law of conservation of matter to explain the need for balancing equations o Write and balance chemical equations predicting product(s) in a reaction using the reference tables Honors Only: o Write and balance ionic & net ionic equations** o Investigate variables that affect the rate of a reaction (nature of reactants, concentration, surface area, and temperature o Explain that nature of reactants can refer to their complexity and the number of bonds that must be broken and reformed in the course of reaction o Explain collision theory and relate theory to variables that affect the rate of a reaction o Define and explain the role of a catalyst in a chemical reaction o Interpret potential energy diagrams for endothermic and exothermic reactions including reactants, products, and activated complex o Temperature change – Tie to endothermic/exothermic reaction. Express ΔH as (+) for endothermic and (–) for exothermic 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Required Vocabulary reactant product precipitate aqueous coefficient collision theory catalys activation energy exothermic endothermic 11. 12. 13. 14. 15. 16. 17. 18. Other Vocabulary synthesis decomposition single replacement double replacement combustion hydrocarbon carbohydrate activity series